Abstract
Summary
The International Osteoporosis Foundation (IOF) and the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) recommend that a marker of bone formation (serum procollagen type I N propeptide, s-PINP) and a marker of bone resorption (serum C-terminal telopeptide of type I collagen, s-CTX) are used as reference analytes for bone turnover markers in clinical studies.
Introduction
Bone turnover markers (BTM) predict fracture risk, and treatment-induced changes in specific markers account for a substantial proportion of fracture risk reduction. The aims of this report were to determine their clinical potential in the prediction of fracture risk and for monitoring the treatment of osteoporosis and to set an appropriate research agenda.
Methods
Evidence from prospective studies was gathered through literature review of the PUBMED database between the years 2000 and 2010 and the systematic review of the Agency for Healthcare Research and Quality up to 2001.
Results
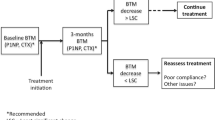
High levels of BTMs may predict fracture risk independently from bone mineral density in postmenopausal women. They have been used for this purpose in clinical practice for many years, but there is still a need for stronger evidence on which to base practice. BTMs provide pharmacodynamic information on the response to osteoporosis treatment, and as a result, they are widely used for monitoring treatment in the individual. However, their clinical value for monitoring is limited by inadequate appreciation of the sources of variability, by limited data for comparison of treatments using the same BTM and by inadequate quality control. IOF/IFCC recommend one bone formation marker (s-PINP) and one bone resorption marker (s-CTX) to be used as reference markers and measured by standardised assays in observational and intervention studies in order to compare the performance of alternatives and to enlarge the international experience of the application of markers to clinical medicine.
Conclusion
BTM hold promise in fracture risk prediction and for monitoring treatment. Uncertainties over their clinical use can be in part resolved by adopting international reference standards.






Similar content being viewed by others
References
Consensus Development Conference (1993) Diagnosis, prophylaxis and treatment of osteoporosis. Am J Med 94:646–650
Kanis JA, Johnell O (2005) Requirements for DXA for the management of osteoporosis in Europe. Osteoporos Int 16:229–238
Burge R, Dawson-Hughes B, Solomon D, Wong J, King A, Tosteson A (2007) Incidence and economic burden of osteoporosis-related fractures in the United States, 2005–2025. J Bone Miner Res 22:465–475
Kanis JA, McCloskey EV, Johansson H, Oden A, Melton LJ, Khaltaev N (2008) A reference standard for the description of osteoporosis. Bone 42:467–475
Kanis JA, Melton LJ, Christiansen C, Johnston C, Khaltaev N (1994) The diagnosis of osteoporosis. J Bone Miner Res 9:1137–1141
Delmas PD (2002) Treatment of postmenopausal osteoporosis. Lancet 359:2018–2026
MacLean C, Newberry S, Maglione M, McMahon M, Ranganath V, Suttorp M, Mojica W, Timmer M, Alexander A et al (2008) Systematic review: comparative effectiveness of treatments to prevent fractures in men and women with low bone density or osteoporosis. Ann Intern Med 148:197–213
Kanis JA, Burlet N, Cooper C, Delmas PD, Reginster J-Y, Borgstrom F, Rizzoli R, on behalf of the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) (2008) European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int 19:399–428
Bergmann P, Body JJ, Boonen S, Boutsen Y, Devogelaer JP, Goemaere S, Kaufman JM, Reginster JY, Gangji V (2009) Evidence-based guidelines for the use of biochemical markers of bone turnover in the selection and monitoring of bisphosphonate treatment in osteoporosis: a consensus document of the Belgian Bone Club. Int J Clin Pract 63:19–26
Vesper H, Cosman F, Endres DB, Garnero P, Hoyle NR, Kleerekoper M, Mallinak NJ (2004) Application of biochemical markers of bone turnover in the assessment and monitoring of bone diseases—approved guidelines. NCCLS document C48-A. ISBN 1-56238-539-9
Brown JP, Albert C, Nassar BA, Adachi JD, Cole D, Davison KS, Dooley KC, Don-Wauchope A, Douville P et al (2009) Bone turnover markers in the management of postmenopausal osteoporosis. Clin Biochem 42:929–942
Szulc P, Delmas P (2008) Biochemical markers of bone turnover: potential use in the investigation and management of postmenopausal osteoporosis. Osteoporos Int 19:1683–1704
Vasikaran SD (2008) Utility of biochemical markers of bone turnover and bone mineral density in management of osteoporosis. Crit Rev Clin Lab Sci 45:221–258
Hannon R, Eastell R (2000) Preanalytical variability of biochemical markers of bone turnover. Osteoporos Int 11:S30–S44
Seibel MJ, Lang M, Geilenkeuser WJ (2001) Interlaboratory variation of biochemical markers of bone turnover. Clin Chem 47:1443–1450
Eisman J, Ebeling P, Ewald D, Flicker L, Holborow B, Nash P, Sambrook P, Seibel M, Stenmark J, Winzenberg T, Herjandono J (2010) Clinical guideline for the prevention and treatment of osteoporosis in postmenopausal women and older men. The Royal Australian College of General Practitioners. www.racgp.org.au. Accessed March 2010
Sociedad Iberoamericana de Osteología y Metabolismo Mineral (SIBOMM) (2009) Ibero-American consensus on osteoporosis (Osteoporosis: Prevención, Diagnóstico y Tratamiento). www.aaomm.org.ar. Accessed March 2010
Lorenc R, Głuszko P, Karczmarewicz E, Księżopolska-Orłowska K, Misiorowski W, Franek E, Horst-Sikorska W, Kaleta M, Męczekalski B et al (2007) Recommendations on the diagnosis and treatment of osteoporosis. Reducing the incidence of fractures through effective prevention and treatment. Terapia 9:11–39
Singapore Ministry of Health (2008) Clinical practice guidelines for osteoporosis. Ministry of Health, Singapore. www.moh.gov.sg/cpg. Accessed March 2010
National Osteoporosis Guideline Group (2008) Osteoporosis—clinical guideline for prevention and treatment—executive summary. www.shef.ac.uk/NOGG. Accessed March 2010
Dawson-Hughes B, Lindsay R, Khosla S, Melton LJ, Tosteson AN, Favus MJ, Baim S (2008) Clinician’s guide to prevention and treatment of osteoporosis. National Osteoporosis Foundation. www.nof.org. Accessed March 2010
Nelson HD, Morris CD, Kraemer DF, Mahon S, Carney N, Nygren PM, Helfand M (2001) Osteoporosis in postmenopausal women: diagnosis and monitoring. Evidence report/technology assessment no. 28. Agency for Healthcare Research and Quality, Rockville
Civitelli R, Armamento-Villareal R, Napoli N (2009) Bone turnover markers: understanding their value in clinical trials and clinical practice. Osteoporos Int 20:843–851
Leeming D, Alexandersen P, Karsdal M, Qvist P, Schaller S, Tankó L (2006) An update on biomarkers of bone turnover and their utility in biomedical research and clinical practice. Eur J Clin Pharmacol 62:781–792
Wickham C, Walsh K, Cooper C, Barker D, Margetts B, Morris J, Bruce S (1989) Dietary calcium, physical activity, and risk of hip fracture: a prospective study. Br Med J Clin Res Ed 299:889–892
Huopio J, Kröger H, Honkanen R, Saarikoski S, Alhava E (2000) Risk factors for perimenopausal fractures: a prospective study. Osteoporos Int 11:219–227
Hochberg MC, Greenspan S, Wasnich RD, Miller P, Thompson DE, Ross PD (2002) Changes in bone density and turnover explain the reductions in incidence of nonvertebral fractures that occur during treatment with antiresorptive agents. J Clin Endocrinol Metab 87:1586–1592
Darby AJ, Meunier PJ (1981) Mean wall thickness and formation periods of trabecular bone packets in idiopathic osteoporosis. Calcif Tissue Int 33:199–204
Jilka RL (2003) Biology of the basic multicellular unit and the pathophysiology of osteoporosis. Med Pediatr Oncol 41:182–185
Garnero P, Sornay-Rendu E, Capuy MC, Delmas PD (1996) Increased bone turnover in late postmenopausal women is a major determinant of osteoporosis. J Bone Miner Res 11:337–349
Chesnut CH, Bell NH, Clark GS, Drinkwater BL, English SC, Johnson CCJ, Notelovitz M, Rosen C, Cain DF, Flessland KA, Mallinak NJ (1997) Hormone replacement therapy in postmenopausal women: urinary N-telopeptide of type I collagen monitors therapeutic effect and predicts response of bone mineral density. Am J Med 102:29–37
Peichl P, Griesmacher A, Pointinger P, Marteau R, Hartl W, Gruber W, Bröll H (1998) Association between female sex hormones and biochemical markers of bone turnover in peri- and postmenopausal women. Calcif Tissue Int 62:388–394
Christiansen C, Riis BJ, Rodbro P (1987) Prediction of rapid bone loss in postmenopausal women. Lancet 1:1105–1108
Christiansen C, Riis BJ, Rodbro P (1990) Screening procedures for women at risk of developing postmenopausal osteoporosis. Osteoporos Int 1:35–40
Cosman F, Nieves J, Wilkinson C, Schnering D, Shen V, Lindsay R (1996) Bone density change and biochemical indices of skeletal turnover. Calcif Tissue Int 58:236–243
Mole PA, Walkinshaw MH, Robins SP, Paterson CR (1992) Can urinary pyridinium crosslinks and urinary oestrogens predict bone mass and rate of bone loss after the menopause? Eur J Clin Investig 22:767–771
Reeve J, Pearson J, Mitchell A, Green J, Nicholls A, Justice J, Hudson E, Klenerman L (1995) Evolution of spinal bone loss and biochemical markers of bone remodelling after menopause in normal women. Calcif Tissue Int 57:105–110
Rosen CJ, Chesnut CH, Mallinak NJ (1997) The predictive value of biochemical markers of bone turnover for bone mineral density in early postmenopausal women treated with hormone replacement therapy or calcium supplementation. J Clin Endocrinol Metab 82:1904–1910
Ross PD, Knowlton W (1998) Rapid bone loss is associated with increased levels of biochemical markers. J Bone Miner Res 13:297–302
Riis BJ, Hansen MA, Jensen AM, Overgaard K, Christiansen C (1996) Low bone mass and fast rate of bone loss at menopause: equal risk factors for future fracture: a 15-year follow-up study. Bone 19:9–12
Finigan J, Greenfield D, Blumsohn A, Hannon R, Peel N, Jiang G, Eastell R (2008) Risk factors for vertebral and nonvertebral fracture over 10 years: a population-based study in women. J Bone Miner Res 23:75–85
Dempster DW (2000) The contribution of trabecular architecture to cancellous bone quality. J Bone Miner Res 15:20–23
Follet H, Boivin G, Rumelhart C, Meunier PJ (2004) The degree of mineralization is a determinant of bone strength: a study on human calcanei. Bone 34:783–789
Banse X, Sims TJ, Bailey AJ (2002) Mechanical properties of adult vertebral cancellous bone: correlation with collagen intermolecular cross-links. J Bone Miner Res 17:1621–1628
Viguet-Carin S, Roux JP, Arlot ME, Merabet Z, Leeming DJ, Byrjalsen I, Delmas PD, Bouxsein ML (2006) Contribution of the advanced glycation end product pentosidine and of maturation of type I collagen to compressive biomechanical properties of human lumbar vertebrae. Bone 39:1073–1079
Bauer DC (2001) Biochemical markers of bone turnover: the Study of Osteoporotic Fracture. In: Eastell R, Baumann M, Hoyle N, Wieczorek L (eds) Bone markers—biochemical and clinical perspectives. Martin Dunitz, London, pp 219–223
Dobnig H, Piswanger-Solkner JC, Obermayer-Pietsch B, Tiran A, Strele A, Maier E, Maritschnegg P, Riedmuller G, Brueck C, Fahrleitner-Pammer A (2007) Hip and nonvertebral fracture prediction in nursing home patients: role of bone ultrasound and bone marker measurements. J Clin Endocrinol Metab 92:1678–1686
Tromp AM, Ooms ME, Popp-Snijders C, Roos JC, Lips P (2000) Predictors of fractures in elderly women. Osteoporos Int 11:134–140
Akesson K, Ljunghall S, Jonsson B, Sernbo I, Johnell O, Gärdsell P, Obrant KJ (1995) Assessment of biochemical markers of bone metabolism in relation to the occurrence of fracture: a retrospective and prospective population-based study of women. J Bone Miner Res 10:1823–1829
Garnero P, Hausherr E, Chapuy M-C, Marcelli C, Grandjean H, Muller C, Cormier C, Breart G, Meunier PJ, Delmas PD (1996) Markers of bone resorption predict hip fracture in elderly women: the EPIDOS prospective study. J Bone Miner Res 11:1531–1538
Gerdhem P, Ivaska KK, Alatalo SL, Halleen JM, Hellman J, Isaksson A, Pettersson K, Vaananen HK, Akesson K, Obrant KJ (2004) Biochemical markers of bone metabolism and prediction of fracture in elderly women. J Bone Miner Res 19:386–393
Ross PD, Kress BC, Parson RE, Wasnich RD, Armour KA, Mizrahi IA (2000) Serum bone alkaline phosphatase and calcaneus bone density predict fractures: a prospective study. Osteoporos Int 11:76–82
Sornay-Rendu E, Munoz F, Garnero P, Duboeuf F, Delmas PD (2005) Identification of osteopenic women at high risk of fracture: the OFELY study. J Bone Miner Res 20:1813–1819
van Daele PL, Seibel MJ, Burger H, Hofman A, Grobbee DE, van Leeuwen JP, Birkenhager JC, Pols HA (1996) Case–control analysis of bone resorption markers, disability, and hip fracture risk: the Rotterdam study. BMJ 312:482–483
Vergnaud P, Garnero P, Meunier PJ, Breart G, Kamihagi K, Delmas PD (1997) Undercarboxylated osteocalcin measured with a specific immunoassay predicts hip fracture in elderly women: the EPIDOS study. J Clin Endocrinol Metab 82:719–724
Szulc P, Chapuy MC, Meunier PJ, Delmas PD (1993) Serum undercarboxylated osteocalcin is a marker of the risk of hip fracture in elderly women. J Clin Invest 91:1769–1774
Chapurlat RD, Garnero P, Bréart G, Meunier PJ, Delmas PD (2000) Serum type I collagen breakdown product (serum CTX) predicts hip fracture risk in elderly women: the EPIDOS study. Bone 27:283–286
Luukinen H, Kakonen S-M, Pettersson K, Koski K, Laippala P, Lovgren T, Kivela S-L, Vaananen HK (2000) Strong prediction of fractures among older adults by the ratio of carboxylated to total serum osteocalcin. J Bone Miner Res 15:2473–2478
Garnero P, Sornay-Rendu E, Claustrat B, Delmas PD (2000) Biochemical markers of bone turnover, endogenous hormones and the risk of fractures in postmenopausal women: the OFELY study. J Bone Miner Res 15:1526–1536
Greenfield DM, Hannon RA, Eastell R (2001) The association between bone turnover and fracture risk (Sheffield Osteoporosis study). In: Eastell R, Baumann M, Hoyle N, Wieczorek L (eds) Bone markers—biochemical and clinical perspectives. Martin Dunitz, London, pp 225–236
Garnero P, Cloos P, Sornay-Rendu E, Qvist PER, Delmas PD (2002) Type I collagen racemization and isomerization and the risk of fracture in postmenopausal women: the OFELY prospective study. J Bone Miner Res 17:826–833
Bruyère O, Collette J, Delmas PD, Rouillon A, Roux C, Seidel L, Richy F, Reginster J-Y (2003) Interest of biochemical markers of bone turnover for long-term prediction of new vertebral fracture in postmenopausal women. Maturitas 44:259–265
Melton LJ III, Crowson CS, O’Fallon WM, Wahner HW, Riggs BL (2003) Relative contributions of bone density, bone turnover, and clinical risk factors to long-term fracture prediction. J Bone Miner Res 18:312–318
Meier C, Nguyen TV, Center JR, Seibel MJ, Eisman JA (2005) Bone resorption and osteoporotic fractures in elderly men: the dubbo osteoporosis epidemiology study. J Bone Miner Res 20:579–587
Ivaska KK, Gerdhem P, Vaananen HK, Akesson K, Obrant KJ (2010) Bone turnover markers and prediction of fracture: a prospective follow-up study of 1040 elderly women for a mean of 9 years. J Bone Miner Res 25:393–403
Bauer DC, Garnero P, Harrison SL, Cauley JA, Eastell R, Ensrud KE, Orwoll E, Osteoporotic Fractures in Men (MrOS) Research Group (2009) Biochemical markers of bone turnover, hip bone loss, and fracture in older men: the MrOS study. J Bone Miner Res 24:2032–2038
Johnell O, Oden A, De Laet C, Garnero P, Delmas PD, Kanis JA (2002) Biochemical indices of bone turnover and the assessment of fracture probability. Osteoporos Int 13:523–526
Bauer DC, Sklarin PM, Stone KL, Black DM, Nevitt MC, Ensrud KE, Arnaud CD, Genant HK, Garnero P, Delmas PD, Lawaetz H, Cummings SR (1999) Biochemical markers of bone turnover and prediction of hip bone loss in older women: the study of osteoporotic fractures. J Bone Miner Res 14:1404–1410
Khosla S, Melton LJ, Atkinson EJ, O’Fallon WM, Klee GG, Riggs BL (1998) Relationship of serum sex steroid levels and bone turnover markers with bone mineral density in men and women: a key role for bioavailable estrogen. J Clin Endocrinol Metab 83:2266–2274
Marcus R, Holloway L, Wells B, Greendale G, James MK, Wasilauskas C, Kelaghan J (1999) The relationship of biochemical markers of bone turnover to bone density changes in postmenopausal women: results from the Postmenopausal Estrogen/Progestin Interventions (PEPI) trial. J Bone Miner Res 14:1583–1595
Stepan JJ, Pospichal J, Presl J, Pacovsky V (1987) Bone loss and biochemical indices of bone remodeling in surgically induced postmenopausal women. Bone 8:279–284
Kanis JA, on behalf of the World Health Organization Scientific Group (2008) Assessment of osteoporosis at the primary health-care level. Technical report, Sheffield, UK. WHO Collaborating Center, University of Sheffield. Available at http://www.shef.ac.uk/FRAX/index.htm. Accessed July 2009
Berry SD, Kiel DP, Donaldson MG, Cummings SR, Kanis JA, Johansson H, Samelson EJ (2010) Application of the National Osteoporosis Foundation Guidelines to postmenopausal women and men: the Framingham Osteoporosis Study. Osteoporos Int 21:53–60
Compston J, Cooper A, Cooper C, Francis R, Kanis JA, Marsh D, McCloskey EV, Reid DM, Selby P, Wilkins M, for the National Osteoporosis Guideline Group (2009) Guidelines for the diagnosis and management of osteoporosis in postmenopausal women and men from the age of 50 years in the UK. Maturitas 62:105–108
Fujiwara S, Nakamura T, Orimo H, Hosoi T, Gorai I, Oden A, Johansson H, Kanis JA (2008) Development and application of a Japanese model of the WHO fracture risk assessment tool (FRAX (TM)). Osteoporos Int 19:429–435
Kanis JA, Black D, Cooper C, Dargent P, Dawson-Hughes B, De Laet C, Delmas PD, Eisman J, Johnell O, on behalf of the International Osteoporosis Foundation and the National Osteoporosis Foundation, USA et al (2002) A new approach to the development of assessment guidelines for osteoporosis. Osteoporos Int 13:527–536
Lippuner K, Johansson H, Kanis JA, Rizzoli R (2010) FRAX (R) assessment of osteoporotic fracture probability in Switzerland. Osteoporos Int 21:381–389
Riggs BL, Parfitt AM (2005) Drugs used to treat osteoporosis: the critical need for a uniform nomenclature based on their action on bone remodeling. J Bone Miner Res 20:177–184
Arlot M, Meunier PJ, Boivin G, Haddock L, Tamayo J, Correa-Rotter R, Jasqui S, Donley DW, Dalsky GP, Martin JS, Eriksen EF (2005) Differential effects of teriparatide and alendronate on bone remodeling in postmenopausal women assessed by histomorphometric parameters. J Bone Miner Res 20:1244–1253
McClung M, Lewiecki E, Cohen S, Bolognese M, Woodson G, Moffett A, Peacock M, Miller P, Lederman S et al (2006) Denosumab in postmenopausal women with low bone mineral density. N Engl J Med 354:821–831
Vasikaran SD, Khan S, McCloskey EV, Kanis JA (1995) Sustained response to intravenous alendronate in postmenopausal osteoporosis. Bone Miner 17:517–520
Saag K, Lindsay R, Kriegman A, Beamer E, Zhou W (2007) A single zoledronic acid infusion reduces bone resorption markers more rapidly than weekly oral alendronate in postmenopausal women with low bone mineral density. Bone 40:1238–1243
Chesnut CH, Silverman S, Andriano K, Genant H, Gimona A, Harris S, Kiel D, LeBoff M, Maricic M et al (2000) A randomized trial of nasal spray salmon calcitonin in postmenopausal women with established osteoporosis: the prevent recurrence of osteoporotic fractures study. Am J Med 109:267–276
Meunier PJ, Roux C, Seeman E, Ortolani S, Badurski JE, Spector TD, Cannata J, Balogh A, Lemmel E-M et al (2004) The effects of strontium ranelate on the risk of vertebral fracture in women with postmenopausal osteoporosis. N Engl J Med 350:459–468
Bruyère O, Collette J, Rizzoli R, Decock C, Ortolani S, Cormier C, Detilleux J, Reginster J-Y (2010) Relationship between 3-month changes in biochemical markers of bone remodelling and changes in bone mineral density and fracture incidence in patients treated with strontium ranelate for 3 years. Osteoporos Int 21:1031–1036
Naylor K, Clowes J, Finigan J, Paggiosi M, Peel N, Eastell R (2010) The effect of cessation of raloxifene treatment on bone turnover in postmenopausal women. Bone 46:592–597
Ettinger B, Black DM, Mitlak BH, Knickerbocker RK, Nickelsen T, Genant HK, Christiansen C, Delmas PD, Zanchetta JR et al (1999) Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene: results from a 3-year randomized clinical trial. JAMA 282:637–645
Prestwood K, Gunness M, Muchmore D, Lu Y, Wong M, Raisz L (2000) A comparison of the effects of raloxifene and estrogen on bone in postmenopausal women. J Clin Endocrinol Metab 85:2197–2202
Pereda C, Hannon R, Naylor K, Eastell R (2002) The impact of subcutaneous oestradiol implants on biochemical markers of bone turnover and bone mineral density in postmenopausal women. Br J Obstet Gynaecol 109:812–820
Harris ST, Watts NB, Genant HK, McKeever CD, Hangartner T, Keller M, Chesnut Iii CH, Brown J, Eriksen EF et al (1999) Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. JAMA 282:1344–1352
Rosen C, Hochberg M, Bonnick S, McClung M, Miller P, Broy S, Kagan R, Chen E, Petruschke R, Thompson D, de Papp A (2005) Treatment with once-weekly alendronate 70 mg compared with once-weekly risedronate 35 mg in women with postmenopausal osteoporosis: a randomized double-blind study. J Bone Miner Res 20:141–151
Hannon R, Clowes J, Eagleton A, Al H, Eastell R, Blumsohn A (2004) Clinical performance of immunoreactive tartrate-resistant acid phosphatase isoform 5b as a marker of bone resorption. Bone 34:187–194
Emkey R, Delmas P, Bolognese M, Borges J, Cosman F, Ragi-Eis S, Recknor C, Zerbini C, Neate C, Sedarati F, Epstein S (2009) Efficacy and tolerability of once-monthly oral ibandronate (150 mg) and once-weekly oral alendronate (70 mg): additional results from the Monthly Oral Therapy With Ibandronate For Osteoporosis Intervention (MOTION) study. Clin Ther 31:751–761
Delmas PD, Recker RR, Chesnut CH, Skag A, Stakkestad JA, Emkey R, Gilbride J, Schimmer RC, Christiansen C (2004) Daily and intermittent oral ibandronate normalize bone turnover and provide significant reduction in vertebral fracture risk: results from the BONE study. Osteoporos Int 15:792–798
Delmas P, Adami S, Strugala C, Stakkestad J, Reginster J, Felsenberg D, Christiansen C, Civitelli R, Drezner M et al (2006) Intravenous ibandronate injections in postmenopausal women with osteoporosis: one-year results from the dosing intravenous administration study. Arthritis Rheum 54:1838–1846
Miller P, McClung M, Macovei L, Stakkestad J, Luckey M, Bonvoisin B, Reginster J, Recker R, Hughes C et al (2005) Monthly oral ibandronate therapy in postmenopausal osteoporosis: 1-year results from the MOBILE study. J Bone Miner Res 20:1315–1322
Black DM, Delmas PD, Eastell R, Reid IR, Boonen S, Cauley JA, Cosman F, Lakatos P, Leung PC et al (2007) Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med 356:1809–1822
Cummings SR, San MJ, McClung MR, Siris ES, Eastell R, Reid IR, Delmas PD, Zoog HB, Austin M et al (2009) Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med 361:756–765
Bone HG, Bolognese MA, Yuen CK, Kendler DL, Wang H, Liu Y, San Martin J (2008) Effects of denosumab on bone mineral density and bone turnover in postmenopausal women. J Clin Endocrinol Metab 93:2149–2157
Lewiecki E, Miller P, McClung M, Cohen S, Bolognese M, Liu Y, Wang A, Siddhanti S, Fitzpatrick L (2007) Two-year treatment with denosumab (AMG 162) in a randomized phase 2 study of postmenopausal women with low BMD. J Bone Miner Res 22:1832–1841
Glover S, Eastell R, McCloskey E, Rogers A, Garnero P, Lowery J, Belleli R, Wright T, John M (2009) Rapid and robust response of biochemical markers of bone formation to teriparatide therapy. Bone Miner 45:1053–1058
Greenspan S, Bone H, Ettinger M, Hanley D, Lindsay R, Zanchetta J, Blosch C, Mathisen A, Morris S, Marriott T (2007) Effect of recombinant human parathyroid hormone (1–84) on vertebral fracture and bone mineral density in postmenopausal women with osteoporosis: a randomized trial. Ann Intern Med 146:326–339
Black D, Bouxsein M, Palermo L, McGowan J, Newitt D, Rosen E, Majumdar S, Rosen C (2008) Randomized trial of once-weekly parathyroid hormone (1–84) on bone mineral density and remodeling. J Clin Endocrinol Metab 93:2166–2172
Black D, Bilezikian J, Ensrud K, Greenspan S, Palermo L, Hue T, Lang T, McGowan J, Rosen C (2005) One year of alendronate after one year of parathyroid hormone (1–84) for osteoporosis. N Engl J Med 353:555–565
Blake G, Compston J, Fogelman I (2009) Could strontium ranelate have a synergistic role in the treatment of osteoporosis? J Bone Miner Res 24:1354–1357
Bone H, McClung M, Roux C, Recker R, Eisman J, Verbruggen N, Hustad C, DaSilva C, Santora A, Ince B (2010) Odanacatib, a cathepsin-K inhibitor for osteoporosis: a two-year study in postmenopausal women with low bone density. J Bone Miner Res 25:937–947
Garnero P, Shih W, Gineyts E, Karpf D, Delmas P (1994) Comparison of new biochemical markers of bone turnover in late postmenopausal osteoporotic women in response to alendronate treatment. J Clin Endocrinol Metab 79:1693–1700
Glover SJ, Gall M, Schoenborn-Kellenberger O, Wagener M, Garnero P, Boonen S, Cauley JA, Black DM, Delmas PD, Eastell R (2009) Establishing a reference interval for bone turnover markers in 637 healthy, young, premenopausal women from the United Kingdom, France, Belgium, and the United States. J Bone Miner Res 24:389–397
Glover SJ, Gamero P, Naylor K, Rogers A, Eastell R (2008) Establishing a reference range for bone turnover markers in young, healthy women. Bone 42:623–630
de Papp AE, Bone HG, Caulfield MP, Kagan R, Buinewicz A, Chen E, Rosenberg E, Reitz RE (2007) A cross-sectional study of bone turnover markers in healthy premenopausal women. Bone 40:1222–1230
Adami S, Bianchi G, Brandi ML, Giannini S, Ortolani S, DiMunno O, Frediani B, Rossini M, on behalf of the BONTURNO study group (2008) Determinants of bone turnover markers in healthy premenopausal women. Calcif Tissue Int 82:341–347
Garnero P, Borel O, Delmas PD (2001) Evaluation of a fully automated serum assay for C-terminal cross-linking telopeptide of type I collagen in osteoporosis. Clin Chem Lab Med 47:694–702
Garnero P, Vergnaud P, Hoyle N (2008) Evaluation of a fully automated serum assay for total N-terminal propeptide of type I collagen in postmenopausal osteoporosis. Clin Chem 54:188–196
Rogers A, Glover SJ, Eastell R (2009) A randomised, double-blinded, placebo-controlled, trial to determine the individual response in bone turnover markers to lasofoxifene therapy. Bone 45:1044–1052
Hannon R, Blumsohn A, Naylor K, Eastell R (1998) Response of biochemical markers of bone turnover to hormone replacement therapy: impact of biological variability. J Bone Miner Res 13:1124–1133
Eastell R, Krege JH, Chen P, Glass EV, Reginster J-Y (2006) Development of an algorithm for using PINP to monitor treatment of patients with teriparatide. Curr Med Res Opin 22:61–66
Greenspan S, Resnick N, Parker R (2005) Early changes in biochemical markers of bone turnover are associated with long-term changes in bone mineral density in elderly women on alendronate, hormone replacement therapy, or combination therapy: a three-year, double-blind, placebo-controlled, randomized clinical trial. J Clin Endocrinol Metab 90:2762–2767
Chen P, Satterwhite JH, Licata AA, Lewiecki EM, Sipos AA, Misurski DM, Wagman RB (2005) Early changes in biochemical markers of bone formation predict BMD response to teriparatide in postmenopausal women with osteoporosis. J Bone Miner Res 20:962–970
Cummings SR, Karpf DB, Harris F, Genant HK, Ensrud K, LaCroix AZ, Black DM (2002) Improvement in spine bone density and reduction in risk of vertebral fractures during treatment with antiresorptive drugs. Am J Med 112:281–289
Watts NB, Cooper C, Lindsay R, Eastell R, Manhart MD, Barton IP, van Staa TP, Adachi JD (2004) Relationship between changes in bone mineral density and vertebral fracture risk associated with risedronate: greater increases in bone mineral density do not relate to greater decreases in fracture risk. J Clin Densitom 7:255–261
Sarkar S, Mitlak BH, Wong M, Stock JL, Black DM, Harper KD (2002) Relationships between bone mineral density and incident vertebral fracture risk with raloxifene therapy. J Bone Miner Res 17:1–10
Delmas PD, Seeman E (2004) Changes in bone mineral density explain little of the reduction in vertebral or nonvertebral fracture risk with anti-resorptive therapy. Bone 34:599–604
Eastell R, Barton I, Hannon RA, Chines A, Garnero P, Delmas PD (2003) Relationship of early changes in bone resorption to the reduction in fracture risk with risedronate. J Bone Miner Res 18:1051–1056
Reginster J-Y, Sarkar S, Zegels B, Henrotin Y, Bruyere O, Agnusdei D, Collette J (2004) Reduction in PINP, a marker of bone metabolism, with raloxifene treatment and its relationship with vertebral fracture risk. Bone 34:344–351
Sarkar S, Reginster J-Y, Crans GG, Diez-Perez A, Pinette KV, Delmas PD (2004) Relationship between changes in biochemical markers of bone turnover and BMD to predict vertebral fracture risk. J Bone Miner Res 19:394–401
Bauer DC, Black DM, Garnero P, Hochberg M, Ott S, Orloff J, Thompson DE, Ewing SK, Delmas PD (2004) Change in bone turnover and hip, non-spine, and vertebral fracture in alendronate-treated women: the fracture intervention trial. J Bone Miner Res 19:1250–1258
Delmas PD, Munoz F, Black DM, Cosman F, Boonen S, Watts NB, Kendler D, Eriksen EF, Mesenbrink PG, Eastell R, The HORIZON-PFT Research Group (2009) Effects of yearly zoledronic acid 5 mg on bone turnover markers and relation of PINP with fracture reduction in postmenopausal women with osteoporosis. J Bone Miner Res 24:1544–1551
Bjarnason NH, Sarkar S, Duong T, Mitlak B, Delmas PD, Christiansen C (2001) Six and twelve month changes in bone turnover are related to reduction in vertebral fracture risk during 3 years of raloxifene treatment in postmenopausal osteoporosis. Osteoporos Int 12:922–930
Li Z, Meredith MP, Hoseyni MS (2002) A method to assess the proportion of treatment effect explained by a surrogate endpoint. Stat Med 20:7531–7588
Freedman LS, Graubard BI, Schatzkin A (1992) Statistical validation of intermediate endpoints for chronic diseases. Stat Med 11:167–178
Eastell R, Hannon RA, Garnero P, Campbell MJ, Delmas PD (2007) Relationship of early changes in bone resorption to the reduction in fracture risk with risedronate: review of statistical analysis. J Bone Miner Res 22:1656–1660
Bauer DC, Garnero P, Hochberg MC, Santora A, Delmas P, Ewing SK, Black DM (2006) Pretreatment levels of bone turnover and the antifracture efficacy of alendronate: the fracture intervention trial. J Bone Miner Res 21:292–299
Veitch SW, Findlay SC, Hamer AJ, Blumsohn A, Eastell R, Ingle BM (2006) Changes in bone mass and bone turnover following tibial shaft fracture. Osteoporos Int 17:364–372
Hansen M, Overgaard K, Riis B, Christiansen C (1991) Potential risk factors for development of postmenopausal osteoporosis—examined over a 12-year period. Osteoporos Int 1:95–102
Delmas PD, Eastell R, Garnero P, Seibel MJ, Stepan J (2000) The use of biochemical markers of bone turnover in osteoporosis. Osteoporos Int 11:S2–S17
McKiernan FE, Berg RL, Linneman JG (2010) The utility of BMD Z-score diagnostic thresholds for secondary causes of osteoporosis. Osteoporos Int. doi:10.1007/s00198-00010-01307-00191
Seibel MJ, Naganathan V, Barton I, Grauer A (2004) Relationship between pretreatment bone resorption and vertebral fracture incidence in postmenopausal osteoporotic women treated with risedronate. J Bone Miner Res 19:323–329
Delmas P, Licata A, Reginster J, Crans G, Chen P, Misurski D, Wagman R, Mitlak B (2006) Fracture risk reduction during treatment with teriparatide is independent of pretreatment bone turnover. Bone Miner 39:237–243
Collette J, Bruyère O, Kaufman J, Lorenc R, Felsenberg D, Spector T, Diaz-Curiel M, Boonen S, Reginster J (2010) Vertebral anti-fracture efficacy of strontium ranelate according to pre-treatment bone turnover. Osteoporos Int 21:233–241
Blouin J, Dragomir A, Ste-Marie LG, Fernandes JC, Perreault S (2007) Discontinuation of antiresorptive therapies: a comparison between 1998–2001 and 2002–2004 among osteoporotic women. J Clin Endocrinol Metab 92:887–894
Blouin J, Dragomir A, Moride Y, Ste-Marie LG, Fernandes JC, Perreault S (2008) Impact of noncompliance with alendronate and risedronate on the incidence of nonvertebral osteoporotic fractures in elderly women. Br J Clin Pharmacol 66:117–127
Siris ES, Harris ST, Rosen CJ, Barr CE, Arvesen JN, Abbott TA, Silverman S (2006) Adherence to bisphosphonate therapy and fracture rates in osteoporotic women: relationship to vertebral and nonvertebral fractures from two US claims databases. Mayo Clin Proc 81:1013–1022
Clowes JA, Peel NF, Eastell R (2004) The impact of monitoring on adherence and persistence with antiresorptive treatment for postmenopausal osteoporosis: a randomized controlled trial. J Clin Endocrinol Metab 89:1117–1123
Delmas PD, Vrijens B, Eastell R, Roux C, Pols HA, Ringe JD, Grauer A, Cahall D, Watts NB, Improving Measurements of Persistence on Actonel Treatment (IMPACT) Investigators (2007) Effect of monitoring bone turnover markers on persistence with risedronate treatment of postmenopausal osteoporosis. J Clin Endocrinol Metab 92:1296–1304
Foged N, Delaissé J, Hou P, Lou H, Sato T, Winding B, Bonde M (1996) Quantification of the collagenolytic activity of isolated osteoclasts by enzyme-linked immunosorbent assay. J Bone Miner Res 11:226–237
Panteghini M (2007) Traceability, reference systems and result comparability. Clin Biochem Rev 28:97–104
Müller MM (2000) Implementation of reference systems in laboratory medicine. Clin Chem 46:1907–1909
Hoelzel W, Weykamp C, Jeppsson JO, Miedema K, Barr JR, Goodall I, Hoshino T, John GW, Kobold U, on behalf of the IFCC Working Group on HbA1c Standardization et al (2004) IFCC reference system for measurement of hemoglobin A1c in human blood and the national standardization schemes in the United States, Japan, and Sweden: a method-comparison study. Clin Chem 50:166–174
Panteghini M, Bunk D, Christenson R, Katrukha A, Porter R, Schimmel H, Wang L, Tate J, The IFCC Working Group on Standardization of Troponin I (2008) Standardization of troponin I measurements: an update. Clin Chem Lab Med 46:1501–1506
Stamey TA, Chen Z, Prestigiacomo AF (1998) Reference material for PSA: the IFCC standardization study. Clin Biochem 31:473–481
Stenman U (2004) Standardization of assays for human chorionic gonadotropin. Clin Chem 50:798–800
Thienpont LM, van Uytfanghe K, Beastall G, Faix JD, Ieiri T, Miller WG, Nelson JC, Ronin C, Ross HA, Thijssen JH, Toussaint B, for the IFCC Working Group on Standardization of Thyroid Function Tests (2010) Report of the IFCC Working Group for Standardization of Thyroid Function Tests; part 1: thyroid-stimulating hormone. Clin Chem 56:902–911
Thienpont LM, van Uytfanghe K, De Leenheer AP (2002) Reference measurement systems in clinical chemistry. Clin Chim Acta 323:73–87
Morris HA (2009) Traceability and standardization of immunoassays: a major challenge. Clin Biochem 42:241–245
Panteghini M (2009) Traceability as a unique tool to improve standardization in laboratory medicine. Clin Biochem 42:236–240
Christenson RH, Duh SH, Apple FS, Bodor GS, Bunk DM, Panteghini M, Welch MJ, Wu AHB, Kahn SE, for the American Association for Clinical Chemistry Cardiac Troponin I Standardization Committee (2006) Toward standardization of cardiac troponin I measurements part II: assessing commutability of candidate reference materials and harmonization of cardiac troponin I assays. Clin Chem 52:1685–1692
Klee GG (2010) Harmonization and standardization of thyroid function tests. Clin Chem 56:879–880
Acknowledgements
This review was undertaken through grants from the International Osteoporosis Foundation and the International Federation of Clinical Chemistry and Laboratory Medicine. It is endorsed by the Committee of Scientific Advisors of the IOF and the Education and Management Division of IFCC. None of the authors were compensated for their contribution.
Conflicts of interest
JAK, CC, OB, AJF, MMcC and MS have no competing interests with regard to markers of bone turnover. AG, SDV, TT and DAW declare no competing interests. HAM has participated in speakers bureaux for Roche Diagnostics Australia and Abbott Diagnostics with the provision of travel expenses. RE has received research funding and/or consultancy funding from Inverness Medical, Unipath, Roche Diagnostics and IDS.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Vasikaran, S., Eastell, R., Bruyère, O. et al. Markers of bone turnover for the prediction of fracture risk and monitoring of osteoporosis treatment: a need for international reference standards. Osteoporos Int 22, 391–420 (2011). https://doi.org/10.1007/s00198-010-1501-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-010-1501-1