Abstract
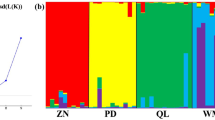
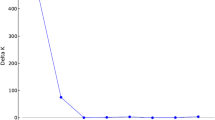
To estimate genetic diversity of the residual northern populations of Oryza rufipogon, a total of 232 individuals from six populations were analyzed using microsatellites (SSRs). The O. rufipogon populations with different status included three from Dongxiang (Jiangxi Province) and three from Chaling (Hunan Province) in China. The 23 rice SSR primer pairs selected from the RiceGenes Database detected a total of 115 alleles, indicating that all the SSR loci were polymorphic in this study. The total gene diversity was 0.919 in the six O. rufipogon populations, and the Donxiang populations showed higher diversity than the Chaling populations. More significant genetic differentiation and less gene flow were found among the Dongxiang populations than those from Chaling. The two putative introgressed populations showed relatively high genetic variation. One in situ conserved population from Dongxiang had the lowest level of genetic diversity. The re-introduced population from Chaling restored about 90% of the genetic variation, compared with the original source population. It is concluded from these results that a relatively high level of genetic variation resided in the northern O. rufipogon populations and continued efforts of conservation of these populations are needed; and that the conservation of some Chaling and Dongxiang populations has been effective in preventing gene flow from cultivated rice. Introgression of cultivated rice demonstrated significant impacts on genetic variability of the O. rufipogon populations, and should be carefully considered in conserving this wild rice. This study also suggested that re-introduction to its original habitats is an effective approach to restore O. rufipogon populations.


Similar content being viewed by others
References
Akagi H, Yokozaki Y, Inagaki A, Fujimura T (1997) Highly polymorphic microsatellites of rice consist of AT repeats, and a classification of closely related cultivars with these microsatellite loci. Theor Appl Genet 94:61–67
Bellon MR, Brar DS, Lu BR, Pham JL (1998) Rice genetic resources. In: Dwoling NG, Greenfield SM, Fischer KS (eds) Sustainability of rice in the global food system. Chapter 16, Davis, California (USA). Pacific Basin Study Center and IRRI, Manila, pp 251–283
Byrne M, Marquez-Garcia MI, Uren T, Smith DS, Moran GF (1996) Conservation and genetic diversity of microsatellite loci in the genus Eucalyptus. Aus J Bot 44:331–341
Chase MR, Moller C, Kessell R, Bawa KS (1996) Distant gene flow in tropical trees. Nature 383:397–398
Cho YG, Ishii T, Temnykh S, Chen X, Lipovich L, McCouch SR, Park WD, Ayres N, Cartinhour S (2000) Diversity of microsatellites derived from genomic libraries and GeneBank sequences in rice (Oryza sativa L.). Theor Appl Genet 100:713–722
Ellstrand NC, Prentice HC, Hancock JF (1999) Gene flow and introgression from domesticated plants into their wild relatives. Annu Rev Ecol Syst 30:539–564
Gao LZ, Hong DY, Ge S (2000a) Allozyme variation and population genetic structure of common wild rice Oryza rufipogon Griff. in China. Theor Appl Genet 101:494–502
Gao LZ, Chen W, Jiang WZ, Ge S, Hong DY, Wang XQ (2000b) Genetic erosion in the Northern marginal population of the common wild rice Oryza rufipogon Griff., and its conservation, revealed by the change of population genetic structure. Hereditas 133:47–53
Gao LZ, Wei CL, Yang QJ (2001) Intra-population genetic structure of Oryza rufipogon Griff. in Yunnan, China. J Plant Res 114:107–113
Ge S, Oliveira GCX, Schaal BA, Gao LZ, Hong DY (1999) RAPD variation within and between natural populations of the wild rice Oryza rufipogon from China and Brazil. Heredity 82:638-644
Hong DY (1995) Rescue the wild rice resources (in Chinese). Bull Chinese Acad Sciences 4:325–326
Innan H, Terauchi, Miyashita NT (1997) Microsatellite polymorphism in natural populations of the wild plant Arabidopsis thaliana. Genetics 146:1441–1452
Morishima H, Barbier P (1990) Mating system and genetic structure of natural populations in wild rice, Oryza rufipogon. Plant Species Biol 5:31–39
Morishima H, Sano Y, Oka HI (1984) Differentiation of perennial and annual types due to habitat conditions in the wild rice Oryza perennis. Plant Syst Evol 144:119–135
McCouch SR, Chen X, Panaud O, Temnykh S, Xu Y, Cho YG, Huang N, Ishii T, Blair M (1997) Microsatellite marker development, mapping and applications in rice genetics and breeding. Plant Mol Biol 35:89–99
Nei M (1978) Estimation of average heterozygosity and genetic distance from a small number of individuals. Genetics 89:583–590
Nei M (1987) Molecular evolutionary genetics. Columbia University Press, New York
Oka HI (1988) Origin of cultivated rice. Japanese Scientific Societies Press, Tokyo
Oka HI, Morishima H (1967) Variation in the breeding systems of a wild rice, Oryza perennis. Evolution 21:249–258
Roa AC, Chavarriaga-Aguirre P, Duque MC, Maya MM, Bonierbale MW, Iglesias C, Tohme J (2000) Cross-species amplification of Cassava (Manihot esculenta) (Euphorbicaceae) microsatellites: allelic poplymorphism and degree of relationship. Am J Bot 87:1647–1655
Rohlf FJ (1994) NTSYS-pc, numerical taxonomy and multivariate analysis system, ver. 1.80. Exeter Software, New York
Slatkin M (1987) Gene flow and the geographic structure of natural populations. Science 236:787–792
Song ZP, Lu BR, Zhu YG, Chen JK (2001) Changes of genetic structure in common wild rice (Oryza rufipogon) populations as referred to by RAPD markers. J Genet Mol Biol 12:78–84
Song ZP, Lu BR, Zhu YG, Chen JK (2002) Pollen competition between cultivated and wild rice species (Oryza sativa and O. rufipogon). New Phytol 153:289–296
Wang ZS, Zhu LH, Liu ZY, Wang XK (1996) Genetic diversity of natural wild rice populations detected by RFLP markers (in Chinese). Agric Biotechnol 4:111-117
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:1358-1370
Wu KS, Tanksley SD (1993) Abundance, polymorphism and genetic mapping of microsatellites in rice. Mol Gen Genet 241:225–235
Xiao JH, Grandillo S, Ahn SN, McCouch SR, Tanksley SD, Li JM, Yuan LP (1996) Genes from wild rice improve yield. Nature 384:223–224.
Xie ZW, Ge S, Hong DY (1999) Preparation of DNA from silica gel a dried mini-amount of leaves of Oryza rufipogon for RAPD study and total DNA bank construction. Acta Bot Sinica 41:807–812
Xie ZW, Lu YQ, Ge S, Hong DY, Li FZ (2001) Clonality in wild rice (Oryza rufipogon, Poaceae) and its implications for conservation management. Am J Bot 88:1058–1064
Wright S (1978) Variability within and among natural populations. Vol. 4. The University Of Chicago Press, Chicago
Yeh FC, Yang RC, Boyle T (1999) Microsoft Window-based freeware for population genetic analysis (POPGENE), ver.1.31. ftp://ftp.microsoft.com/softlib/MSLFILES/HPGL.EXE
Yuan LP, Virmani SS, Mao CX (1989) Hybrid rice: achievements and further outlook. In: Progress in irrigated rice research. International Rice Research Institute, Manila, The Philippines, pp 219–223
Zhou J (1995) Studies on conservation biology of three Northern populations of common wild rice (Oryza rufipogon) (in Chinese). PhD dissertation, Wuhan University, Wuhan, China
Acknowledgements
We thank Mr. Guihua Liu and Ji Qian for their assistants in sample collection, and to Mr. M. Reagon of the Ohio State University for his comments on this manuscript. This research was supported by the National Natural Science Foundation of China for Distinguished Young Scholars (Grant No. 30125029), the 863 Program of Ministry of Science and Technology of China (Grant No. 2001AA227141), and Shanghai Commission of Science and Technology (Grant No. 02JC140022).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J.S. Heslop-Harrison
Rights and permissions
About this article
Cite this article
Song, Z.P., Xu, X., Wang, B. et al. Genetic diversity in the northernmost Oryza rufipogon populations estimated by SSR markers. Theor Appl Genet 107, 1492–1499 (2003). https://doi.org/10.1007/s00122-003-1380-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-003-1380-3