Abstract
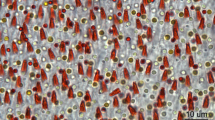
A putative photoreceptor organ is described in the carabid beetle, Pachymorpha sexguttata. The elongated structure, about 20–40 μm wide and more than 300 μm long, is situated within the optic lobe at the fronto-dorsal rim of the lamina. It lies, deep in the head capsule, in front of the compound eyes and beneath window-like thinnings of the cuticle. The organ is composed of two types of cells: (1) clear sheath cells and (2) well-organized inner receptor cells that appear in a horseshoe-like or circular array in cross-section. Common histological features of all inner cells include a distal trunk ending in microvilli that form a rhabdom-like structure, an axon at the proximal end of the cell, lamellar and multivesicular bodies within the trunk, and clusters of small mitochondria. The organ has no shielding pigment. It is connected by thin axons to a circumscribed neuropil that parallels the organ, and thence via a fiber tract to the medulla accessoria, a possible site of the circadian pacemaker in insects. Immunoreactivity to anti-per s, an antibody recognizing the Drosophila period (per) protein that plays a central role in the function of the circadian pacemaker in fruit flies, is demonstratable in thin efferent terminals within the organ, in the associated neuropil and in its fiber connection to the medulla. A second receptor organ displaying the same fine structure lies near the second optic chiasm. This set of putative photoreceptors also occurs in the tenebrionid beetle, Zophobas morio, and its pupa. The possible function of these receptor organs is discussed with respect to former chronobiological data and some recently described types of extraretinal photoreceptors in arthropods.
Similar content being viewed by others
References
Bennett MF (1979) Extraocular light receptors and circadian rhythms. In: Autrum H (ed) Comparative physiology and evolution of vision in invertebrates. A. Invertebrate photoreceptors. Springer, Berlin Heidelberg New York, pp 641–663
Bernhard GD (1975) Physiological optics of the fused rhabdom. In: Snyder AW, Menzel R (eds) Photoreceptor optics, Springer, Berlin Heidelberg New York, pp 78–97
Block GD, Wallace S (1982) Localization of a circadian pacemaker in the eye of a mollusc, Bulla. Science 217:155–157
Bolwig N (1945/46) Senses and sense organs of the anterior end of the house fly larvae. Videnk Medd Dan Naturhist Foren Khobenhavn 109:81–217
Brandes-Frisch B, Fleissner G, Fleissner G, Hall J (1990) “per”-Reactive neurons in the CNS of scorpion and beetle. Verh Dtsch Zool Ges 83:630–631
Calman BG, Lauerman MA, Andrews A, Schmidt M, Batelle BA (1991) Central projections of Limulus photoreceptor cells revealed by a photoreceptor-specific monoclonal antibody. J Comp Neurol 313:553–562
Colot HV, Hall JC, Rosbash M (1988) Interspecific comparison of the period gene of Drosophila reveals large blocks of nonconserved coding DNA. EMBO J 7:3929–3937
Ewer J, Hamblen-Coyle M, Rosbash M, Hall JC (1990) Requirement for period gene expression in the adult and not during development for locomotor activity rhythms of imaginal Drosophila melanogaster. J Neurogenet 7:31–73
Ewer J, Frisch B, Hamblen-Coyle MJ, Rosbash M, Hall JC (1992) Expression of the period clock gene within different cell types in the brain of Drosophila adults and mosaic analysis of these cells' influence on circadian behavioral rhythms. J Neurosci 12:3321–3349
Fleissner G (1982) Isolation of an insect clock. J Comp Physiol [A] 149:311–316
Fleissner G, Fleissner G (1992) A putative new extraretinal photoreceptor in the ground-beetle, Pachymorpha sexguttata. In: Elsner N, Richter DW (eds) Rhythmogenesis in neurons and networks. Thieme, Stuttgart New York, pp 294–295
Garcia-Fernández JM, Siwicki KK, Lawrence MD, Foster RG (1993) Localization of Drosophila per- like protein in the CNS of Xenopus, Anolis and wild-type and tau mutant golden hamsters. J Biol Rhythms (in press)
Goodman LY (1981) Organisation and physiology of the insect dorsal ocellar system. In: Autrum H (ed) Comparative physiology and evolution of vision in invertebrates. C. Invertebrates visual centers and behavior II. Springer, Berlin Heidelberg New York, pp 201–286
Hagberg M (1986) Ocellar and extraocular photoreceptors and associated interneurons in insects. Thesis, University of Lund, Sweden
Hall JC (1990) Genetics of circadian rhythms. Annu Rev Genet 24:659–697
Helfrich C, Engelmann W (1983) Circadian rhythm of the locomotor activity in Drosophila melanogaster mutants “sine oculis” and “small optic lobes”. Physiol Entomol 8:257–272
Hofbauer A, Buchner E (1989) Does Drosophila have seven eyes? Naturwissenschaften 76:335–336
Koehler WK, Fleissner G (1978) Internal desynchronisation of bilaterally organised circadian oscillators in the visual system of insects. Nature 274:708–710
Konopka RJ, Benzer S (1971) Clock mutants in Drosophila melanogaster. Proc Natl Acad Sci USA 68:2112–2116
Meissl H, Dodt E (1981) Comparative physiology of pineal photoreceptor organs. Rev Endocrinol 14:61–80
Meissl H, Ekström P (1988) Photoreceptor responses to light in the isolated pineal organ of the trout, Salmo gairdneri. Neuroscience 25:1071–1076
Menaker M (1976) Symposium on extraretinal photoreception in circadian rhythms and related phenomena. Photochem Photobiol 23:213–306
Mischke U (1986) Stemmata: Innere “Augen” der Insekten. Verh Dtsch Zool Ges 79:227–228
Müller B, Peichl L, Grip J de, Gery I, Korf HW (1989) Opsin- and S-antigen-like immunoreactions in photoreceptors in the tree shrew retina. Invest Ophthalmol Vis Sci 30:530–535
Nässel DR, Holmquist MH, Hardie RC, Håkanson R, Sundler F (1988) Histamine-like immunoreactivity in photoreceptors of the compound eyes and ocelli of the flies, Calliphora erythrocephala and Musca domestica. Cell Tissue Res 253:639–646
Nishiitsutsuji-Uwo J, Pittendrigh CS (1968) Central nervous system control of circadian rhythmicity of the cockroach. Z Vergl Physiol 58:1–13
Oksche A, Hartwig HG (1979) Pineal sense organs — components of photoneuroendocrine systems. Prog Brain Res 52:113–130
Page TL (1982) Transplantation of the cockroach circadian pacemaker. Science 216:73–75
Page TL (1985) Clocks and circadian rhythms. Compr Insect Physiol Biochem Pharmacol 6:577–652
Pollack I, Hofbauer A (1991) Histamine-like immunoreactivity in the visual system and brain of Drosophila melanogaster. Cell Tissue Res 266:391–398
Rosbash M, Hall JC (1989) The molecular biology of circadian rhythms. Neuron 3:337–398
Rosbash M, Hall JC (1989) The molecular biology of circadian rhythms. Neuron 3:337–398
Sandemann DC, Sandeman RE, Couet HG (1990) Extraretinal photoreceptors in the brain of the crayfish, Cherax destructor. J Neurobiol 21:619–629
Scharrer E (1964) Photo-neuro-endocrine systems: general concepts. Ann NY Acad Sci 117:13–22
Schultz WD, Schlüter U, Seifert G (1984) Extraocular photoreceptors in the brain of Epilachna varivestis (Coleoptera, Coccinellidae). Cell Tissue Res 236:317–320
Schwemer J (1986) Turnover of photoreceptor membrane and visual pigment in invertebrates. In: Stieve H (ed) The molecular mechanism of photoreception. Dahlem Konferenzen. Springer, Berlin Heidelberg New York, pp 303–326
Shaw SR (1978) The extracellular space and blood-brain barrier in an insect retina: an ultrastructural study. Cell Tissue Res 188:35–61
Siwicki KK, Eastman C, Petersen G, Rosbash M, Hall JC (1988) Antibodies to the period gene product of Drosophila reveal diverse tissue distribution and rhythmic changes in the visual system. Neuron 1:141–150
Siwicki KK, Strack ST, Rosbash M, Hall JC, Jacklet J (1989) An antibody to the Drosophila period protein recognizes circadian pacemker neurons in Aplysia and Bulla. Neuron 3:51–58
Siwicki KK, Schwartz J, Hall JC (1992) An antibody to the Drosophila period protein labels antigens in the suprachiasmatic nucleus of the rat. J Neurogenet 8:33–92
Smith DP, Stamnes MA, Zuker CS (1991) Signal transduction in the visual system of Drosophila. Annu Rev Cell Biol 7:161–190
Snyder A (1975) Photoreceptor optics — theoretical principles. In: Snyder A, Menzel R (eds) Photoreceptor optics. Springer, Berlin Heidelberg New York, pp 38–55
Strausfeld NJ (1976) Atlas of an insect brain. Springer, Berlin Heidelberg New York
Thompson K, Bacon JP (1991) The vasopressin-like immunoreactive (VPLI) neurons of the locust, Locusta migratoria. II. Physiology. J Comp Physiol [A] 168:619–630
Thompson K, Tyrer NM, May ST, Bacon JP (1991) The vasopressin-like immunoreactive (VPLI) neurons of the locust, Locusta migratoria. I. Anatomy. J Comp Physiol [A] 168:605–617
Tomioka K, Chiba Y (1982) Persistence of circadian ERG-rhythm in the cricket with optic lobes severed. Naturwissenschaften 20:542
Truman JW (1976) Extraretinal photoreception in insects. Photochem Photobiol 23:215–225
Veen TH van, Elofsson R, Hartwig HG, Gery I, Mochizuki M, Cena V, Klein DC (1986) Retinal S-antigen: immunocytochemical and immunochemical studies on distribution in animal photoreceptors and pineal organs. Exp Biol 45:15–25
Yoshida M (1981) Extraocular photoreception. In: Autrum HJ (ed) Comparative physiology and evolution of vision in invertebrates. A. Invertebrate photoreceptors. Springer, Berlin Heidelberg New York, pp 582–640
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fleissner, G., Fleissner, G. & Frisch, B. A new type of putative non-visual photoreceptors in the optic lobe of beetles. Cell Tissue Res 273, 435–445 (1993). https://doi.org/10.1007/BF00333698
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00333698