Abstract
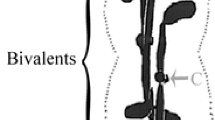
Kinetochores in rat kangaroo (PtK2) cells in prophase of mitosis are finely fibrillar, globular bodies, 5000–8000 Å in diameter. Sister kinetochores are attached to opposite lateral faces in the primary constriction of chromosomes. No microtubules (MTs) occur in prophase nuclei. During prometaphase the ball-shaped kinetochores differentiate into trilaminar plaques. An outer kinetochore layer, less electron dense than chromatin, appears first in the fibrillar matrix. The inner layer, continuous with, but more electron dense than the chromosome, is formed later. Kinetochore-spindle MT interaction is evident at the very beginning of prometaphase. As a result, kinetochore shape is very variable, but three types of kinetochores can be distinguished by fine structure analysis. A comparison of kinetochore structure and chromosome position in the mitotic spindle yielded clues regarding initial orientation and congression. At the time the nuclear envelope (NE) breaks down chromosomes near asters orient first. Chromosomes approximately equidistant from the two spindle poles amphi-orient immediately. Chromosomes closer to one pole probably achieve mono-orientation first, then amphi-orient and congress. In normal metaphase all the chromosomes lie at or near the spindle equator and kinetochores are structurally uniform. Paraxial and para-equatorial sections revealed that they are trilaminar, roughly circular plaques of 4000–6000 Å diameter. Inner and outer layers are 400 Å, and the electron translucent middle layer which separates them is 270 Å thick. From 16 to 40 MTs are anchored in the outer layer. In cold-treated cells the kinetochores are trilaminar, but in colcemid-treated cells the inner layer is lacking. Both kinetochores and their MTs are disorganized beginning in late anaphase. In telophase the inner layer persists for some time as an electron dense patch apposed to the NE, while the outer layer disintegrates.
Similar content being viewed by others
References
Barnicot, N. A., Huxley, H. E.: Electron microscope observations on mitotic chromosomes. Quart. J. micr. Sci. 106, 197–214 (1965).
Bauer, H., Dietz, R., Röbbelen, C.: Die Spermatocytenteilungen der Tipuliden. III. Mitt. Das Bewegungsverhalten der Chromosomen in Translokationsheterozygoten von Tipula oleracea. Chromosoma (Berl.) 12, 116–189 (1961).
Brinkley, B. R., Cartwright, J., Jr.: Organization of microtubules in the mitotic spindle: Differential effects of cold shock on microtubule stability. J. Cell Biol. 47, 25a (1970) (abstr.).
Brinkley, B. R., Cartwright, J., Jr.: Ultrastructural analysis of mitotic spindle elongation in mammalian cells in vitro. Direct microtubule counts. J. Cell Biol. 50, 416–431 (1971).
Brinkley, B. R., Murphy, P., Richardson, L. C.: Procedure for embedding in situ selected cells cultured in vitro. J. Cell Biol. 35, 279–283 (1967).
Brinkley, B. R., Stubblefield, E.: The fine structure of the kinetochore of a mammalian cell in vitro. Chromosoma (Berl.) 19, 28–43 (1966).
Brinkley, B. R., Stubblefield, E.: Ultrastructure and interaction of the kinetochore and centriole in mitosis and meiosis. Advanc. Cell Biol. 1, 119–185 (1970).
Comings, D. E., Okada, T. A.: Fine structure of kinetochore in Indian muntjac. Exp. Cell Res. 67, 97–110 (1971).
George, P., Journey, L. J., Goldstein, M. N.: Effect of vincristine on the fine structure of HeLa cells during mitosis. J. nat. Cancer Inst. (Wash.) 35, 355–375 (1965).
Jokelainen, P. T.: The differentiation of sister kinetochores during metakinesis. J. Cell Biol. 27, 48a (1965) (abstr.).
Jokelainen, P. T.: The ultrastructure and spatial organization of the metaphase kinetochore in mitotic rat cells. J. Ultrastruct. Res. 19, 19–44 (1967).
Journey, L. J., Whaley, A.: Kinetochore ultrastructure in vincristine-treated mammalian cells. J. Cell Sci. 7, 49–54 (1970).
Krishan, A.: Fine structure of the kinetochores in vinblastine sulfate-treated cells. J. Ultrastruct. Res. 23, 134–143 (1968).
Krishan, A., Buck, R. C.: Structure of the mitotic spindle in L strain fibroblasts. J. Cell Biol. 24, 433–444 (1965).
Lambert, A. M.: Étude de structures cinétiques en rapport avec la rupture de la membrane nucléaire, en début de méiose chez Mnium hornum L. Organisation des centromères. C. R. Acad. Sci. (Paris) D 270, 481–484 (1970).
Luft, J. H.: Improvements in epoxy resin embedding methods. J. biophys. biochem. Cytol. 9, 409–414 (1961).
Luykx, P.: The structure of the kinetochore in meiosis and mitosis in Urechis eggs. Exp. Cell Res. 39, 643–657 (1965).
McIntosh, J. R., Landis, S. C.: The distribution of spindle microtubules during mitosis in cultured human cells. J. Cell Biol. 49, 468–497 (1971).
Millonig, G.: Advantages of a phosphate buffer for OsO4 solutions in fixation. J. appl. Phys. 32, 1637 (abstr.) (1961).
Nebel, B. R., Coulon, E. M.: The fine structure of chromosomes in pigeon spermatocytes. Chromosoma (Berl.) 13, 272–291 (1962).
Nicklas, R. B.: Chromosome micromanipulation. II. Induced reorientation and the experimental control of segregation in meiosis. Chromosoma (Berl.) 21, 17–50 (1967).
Nicklas, R. B.: Mitosis. Advanc. Cell Biol. 2, 225–297 (1971).
Östergren, G.: The mechanism of co-orientation in bivalents and multivalents. The theory of orientation by pulling. Hereditas (Lund) 37, 85–156 (1951).
Pickett-Heaps, J. D., Fowke, L. C.: Cell division in Oedogonium. I. Mitosis, cytokinesis, and cell elongation. Aust. J. biol. Sci. 22, 857–894 (1969).
Reynolds, E. S.: The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J. Cell Biol. 17, 208–212 (1963).
Robbins, E., Gonatas, N. K.: The ultrastructure of a mammalian cell during the mitotic cycle. J. Cell Biol. 21, 429–463 (1964).
Roos, U.-P.: Normal and abnormal mitosis in a mammalian cell in vitro. A light and electron microscopic study. 275 p. Doctoral Dissertation, University of Florida 1971.
Roos, U.-P.: Light and electron microscopy of rat kangaroo cells in mitosis. I. Formation and breakdown of the mitotic apparatus. Chromosoma (Berl.) 40, 43–82 (1973).
Roth, L. E.: Electron microscopy of mitosis in amebae. III. Cold and urea treatments: A basis for tests of direct effects of mitotic inhibitors on microtubule formation. J. Cell Biol. 34, 47–60 (1967).
Watson, M. L.: Staining of tissue sections for electron microscopy with heavy metals. J. biophys. biochem. Cytol. 4, 475–478 (1958).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Roos, U.P. Light and electron microscopy of rat kangaroo cells in mitosis. Chromosoma 41, 195–220 (1973). https://doi.org/10.1007/BF00319696
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00319696