Summary
The pineal complex of the teleost, Phoxinus phoxinus L., was studied light-microscopically by the use of the indirect immunocytochemical antiopsin reaction and the histochemical acetylcholinersterase (AChE) method.
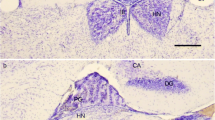
Opsin-immunoreactive outer segments of photoreceptor cells were demonstrated in large numbers in all divisions of the pineal end-vesicle and in the pineal stalk. Moreover, they were found in the roof of the third ventricle, adjacent to the orifice of the pineal recess as well as scattered in the parapineal organ. These immunocytochemical observations provide direct evidence of the presence of an opsin associated with a photopigment in the photosensory cells of the pineal and parapineal organs of Phoxinus.
By means of the AChE reaction (Karnovsky and Roots 1964) inner segments of pineal photoreceptors, intrinsic nerve cells, several intrapineal bundles of nerve fibers, and a prominent pineal tract were specifically marked. The pineal neurons can be divided into two types: one is located near the pineal lumen, the other near the basal lamina. The latter perikarya bear stained processes directed toward the photoreceptor layer. A rostral aggregation of two types of AChE-positive nerve cells occurs in the ventral wall of the pineal end-vesicle. The main portion of the AChE-positive pineal tract, which lies within the dorsal wall of the pineal stalk, can be followed to the posterior commissure where some of the nerve fibers course laterally. A few AChE-positive pineal nerve fibers run toward the lateral habenular nucleus via the habenular commissure. In the region of the subcommissural organ single AChE-positive neurons accompany the pineal tract. The nerve cells of the parapineal organ exhibit a moderate AChE activity.
These findings extend the structural basis for the remarkable light-dependent activity of the pineal organ of Phoxinus phoxinus.
Similar content being viewed by others
References
Ali MA, Wagner HJ (1975) Visual pigments: physiology and ecology. In: Ali MA (ed) Vision in fishes. Plenum Publ Corp, New York, pp 481–516
Benoit J (1970) Étude de l'action des radiations visibles sur la gonadostimulation et de leur pénétration intracranienne chez les Oiseaux et les Mammifères. CNRS (ed) La photorégulation de la reproduction chez les Oiseaux et les Mammifères. CNRS, Paris, pp 121–149
Breucker H, Horstmann E (1965) Elektronenmikroskopische Untersuchungen am Pinealorgan der Regenbogenforelle (Salmo irideus). Prog Brain Res 10:259–269
Converse CA, Papermaster DS (1975) Membrane protein analysis by two-dimensional immunoelectrophoresis. Science (Wash DC) 189:469–472
Crescitelli F (1972) The visual cells and visual pigments of the vertebrate eye. In: Dartnall HJA (ed) Photochemistry of vision, Handbook of sensory physiology VII/1. Springer, Berlin Heidelberg New York, pp 245–363
Dartnell HJA (1972) Photochemistry of vision. Handbook of sensory physiology VII/1. Springer, Berlin, pp 122–145
Dartnall HJA, Lander MR, Munz FW (1961) Periodic changes in the visual pigment of a fish. In: Christensen BChr, Buchmann B (eds) Progress in photobiology. Elsevier, Amsterdam, pp 203–213
Dodt E (1963) Photosensitivity of the pineal organ in the teleost, Salmo irideus (Gibbons). Experientia (Basel) 19:642
Dodt E (1973) The parietal eye (pineal and parapineal organs) of lower vertebrates. In: Jung R (ed) Handbook of sensory physiology VII/3B. Springer, Berlin Heidelberg New York, pp 113–140
Eldred WD, Nolte J (1981) Multiple classes of photoreceptors and neurons in the frontal organ of Rana pipiens. J Comp Neurol 203:269–296
Erlandsen SL, Parsons JA, Burke JP, Redick JA, Van Orden DE, Van Orden LS (1975) A modification of the unlabeled antibody enzyme method using heterologous antisera for the light microscopic and ultrastructural localization of insulin, glucagon and growth hormone. J Histochem Cytochem 23:666–677
Falcon J (1979a) L'organe pinéal du Brochet (Esox lucius L.). I. Étude anatomique et cytologique. Ann Biol Anim Bioch Biophys 19:445–465
Falcon J (1979b) Unusual distribution of neurons in the pike pineal organ. Progr Brain Res 52:89–91
Falcon J, Meissl H (1981) The photosensory function of the pineal organ of the pike (Esox lucius, L.). Correlation between structure and function. J Comp Physiol 144:127–137
Franz V (1912) Beiträge zur Kenntnis des Ependyms im Fischgehirn. Biol Zbl 32:375–383
Frisch K von (1911) Beiträge zur Physiologie der Pigmentzellen in der Fischhaut. Pflügers Arch ges Physiol 138:319–387
Hafeez MA, Zerihun (1974) Studies on central projections of the pineal nerve tract in rainbow trout, Salmo gairdneri Richardson, using cobalt chloride iontophoresis. Cell Tissue Res 154:485–510
Hanyu I, Niwa H, Tamura T (1978) Salient features of photosensory function of the teleostean pineal organ. Comp Biochem Physiol 61A:49–54
Hartwig HG (1975) Neurobiologische Studien an photoneuroendokrinen Systemen. These, Justus Liebig Universität, Giessen
Hartwig HG, Baumann C (1974) Evidence for photosensitive pigments in the pineal complex of the frog. Vision Res 14:597–598
Hartwig HG, Pfautsch M (1973) Rasterelektronenmikroskopische Beobachtungen an pinealen Sinneszellen der Forelle, Salmo gairdneri (Teleostei). Z Zellforsch 138:585–589
Hartwig HG, Veen Th van (1979) Spectral characteristics of visible radiation penetrating into the brain and stimulating extraretinal photoreceptors. J Comp Physiol 130:277–282
Heller J (1969) Comparative study of a membrane protein. Characterization of bovine, rat and frog visual pigments. Biochemistry 8:675–678
Herwig HJ (1981) An ultrastructural and biochemical study on the pineal organ of Hemigrammus caudovittatus and other closely related characid fish species with special reference to the Mexican blind cave fish Astyanax mexicanus. Thesis, Utrecht
Karnovsky MJ, Roots L (1964) A “direct coloring” thiocholine method for cholinesterase. J Histochem Cytochem 12:219–221
Koelle GB (1955) The histochemical identification of acetylcholinesterase in cholinergic, adrenergic and sensory neurons. J Pharmacol Exp Ther 114:167–184
Korf HW (1974) Acetylcholinesterase-positive neurons in the pineal and parapineal organs of the rainbow trout, Salmo gairdneri (With special reference to the pineal tract). Cell Tissue Res 155:475–489
Korf HW (1976) Histological, histochemical and electron microscopical studies on the nervous apparatus of the pineal organ in the tiger salamander, Ambystoma tigrinum. Cell Tissue Res 174:475–497
Korf HW, Wagner U (1981) Nervous connections of the parietal eye in adult Lacerta s. sicula Rafinesque as demonstrated by anterograde and retrograde transport of horseradish peroxidase. Cell Tissue Res 219:567–583
Korf HW, Liesner R, Meissl H, Kirk A (1981) Pineal complex of the clawed toad, Xenopus laevis Daud.: Structure and function. Cell Tissue Res 216:113–130
Korf HW, Zimmermann NH, Oksche A (1982) Intrinsic neurons and neural connections of the pineal organ of the house sparrow, Passer domesticus, as revealed by anterograde and retrograde transport of horseradish peroxidase. Cell Tissue Res 222:243–260
La Motte I de (1963) Untersuchungen zur vergleichenden Physiologie der Lichtempfindlichkeit geblendeter Fische. Naturwissenschaften 50:363
Loew ER, Dartnall HJA (1976) Vitamin A1/A2-based visual pigment mixtures in cone of the rudd. Vision Res 16:891–896
Marucci AA, Dougherty RM (1975) Use of the unlabeled antibody histochemical technique for the detection of human antibody. J Histochem Cytochem 23:618–623
Matsuura T, Herwig HJ (1981) Histochemical and ultrastructural study of the nervous elements in the pineal organ of the eel, Anguilla anguilla. Cell Tissue Res 216:545–557
Mayor HD, Hampton JC, Rosario B (1961) A simple method for removing the resin from epoxy-embedded tissue. J Biophys Biochem Cytol 9:909–910
McNulty JA (1981) Synaptic ribbons in the pineal organ of the goldfish: circadian rhythmicity and the effect of constant light and constant darkness. Cell Tissue Res 215:491–497
Meiniel A (1981) New aspects of the phylogenetic evolution of sensory cell lines in the vertebrate pineal complex. In: Oksche A, Pévet P (eds) The pineal organ. Photobiology — Biochronometry — Endocrinology. Developments in endocrinology 14:27–48. Elsevier, Amsterdam New York
Meissl H, Dodt E (1981) Comparative physiology of pineal photoreceptor organs. In: Oksche A, Pévet P (eds) The pineal organ. Photobiology —Biochronometry — Endocrinology. Developments in endocrinology 14:61–80. Elsevier, Amsterdam New York
Morita Y (1966) Entladungsmuster pinealer Neurone der Regenbogenforelle (Salmo irideus) bei Belichtung des Zwischenhirns. Pflügers Arch ges Physiol 289:155–167
Morita Y (1975) Direct photosensory activity of the pineal. In: Knigge KM, Scott DE, Kobayashi H, Ishii S (eds) Brain-endocrine interaction II. The ventricular system. Karger, Basel, pp 376–387
Morita Y, Bergmann G (1971) Physiologische Untersuchungen und weitere Bemerkungen zur Struktur des lichtempfindlichen Pinealorgans von Pterophyllum scalare Cuv. et Val. (Cichlidae, Teleostei). Z Zellforsch 119:289–294
Morton RA (1972) The chemistry of the visual pigments. In: Dartnall HJA (ed) Photochemistry of vision. Handbook of sensory physiology VII/1. Springer, Berlin Heidelberg New York, pp 31–68
Ohba S, Wake K, Ueck M (1979) Histochemical and electron-microscopical findings in the pineal organ of Carassius gibelio (Langsd). Progr Brain Res 52:93–96
Oksche A (1970) Zur Differenzierung sensorischer und sekretorischer Strukturelemente im Zentralnervensystem. Verh Deutschen Zool Ges, 64. Tagung. Fischer, Stuttgart, pp 72–79
Oksche A (1971) Sensory and glandular elements of the pineal organ. In: Wolstenholme GEW, Knight J (eds) The pineal gland. Churchill, London, pp 127–146
Oksche A, Hartwig HG (1975) Photoneuroendocrine systems and the third ventricle. In: Knigge KM, Scott DE, Kobayashi H, Ishii S (eds) Brain-endocrine interaction II. The ventricular system. Karger, Basel, pp 40–53
Oksche A, Hartwig HG (1979) Pineal sense organs — components of photoneuroendocrine systems. Progr Brain Res 52:113–130
Oksche A, Kirschstein H (1966) Elektronenmikroskopische Feinstruktur der Sinneszellen im Pinealorgan von Phoxinus laevis L. Naturwissenschaften 53:591
Oksche A, Kirschstein H (1967) Die Ultrastruktur der Sinneszellen im Pinealorgan von Phoxinus laevis. Z Zellforsch 78:151–166
Oksche A, Kirschstein H (1971) Weitere elektronenmikroskopische Untersuchungen am Pinealorgan von Phoxinus laevis (Teleostei, Cyprinidae). Z Zellforsch 112:572–588
Oksche A, Vaupel-von Harnack M (1965) Vergleichende elektronenmikroskopische Studien am Pinealorgan. Progr Brain Res 10:237–258
Omura Y (1979) Light and electron microscopic studies on the pineal tract of rainbow trout, Salmo gairdneri. Rev Can Biol 38:105–118
Omura Y, Ali MA (1980) Responses of pineal photoreceptors in the brook and rainbow trout. Cell Tissue Res 208:111–122
Papermaster DS, Converse CA, Siu J (1975) Membrane biosynthesis in the frog retina: opsin transport in the photoreceptor cell. Biochemistry 14:1343–1352
Papermaster DS, Converse CA, Zorn M (1976) Biosynthesis and immunocytochemical characterization of a large protein in frog and cattle rod outer segment membranes. Exp Eye Res 23:105–116
Papermaster DS, Schneider BG, Zorn MA, Kraehenbuhl JP (1978) Immunocytochemical localization of opsin in outer segments and Golgi zones of frog photoreceptor cells. An electron microscope analysis of cross-linked albumin-embedded retinas. J Cell Biol 77:196–210
Rüdeberg C (1969) Structure of the parapineal organ of the adult rainbow trout, Salmo gairdneri Richardson. Z Zellforsch 93:282–304
Rüdeberg C (1971) Structure of the pineal organs of Anguilla anguilla L. and Lebistes reticulatus Peters (Teleostei). Z Zellforsch 122:227–243
Schäfer O (1964) Spektrale Empfindlichkeit und absolute Schwelle des Farbwechsels geblendeter Elritzen (Phoxinus phoxinus L.). Biol Zbl 83:47–66
Scharrer E (1928) Die Lichtempfindlichkeit blinder Elritzen I. Untersuchungen über das Zwischenhirn der Fische. Z vergl Physiol 7:1–38
Schwanzara SA (1967) The visual pigments of freshwater fishes. Vision Res 7:121–148
Sternberger LA (1979) Immunocytochemistry. John Wiley, New York Chichester Brisbane Toronto, 2nd ed
Tabata M, Tamura T, Niwa H (1975) Origin of the slow potential in the pineal organ of the rainbow trout. Vision Res 15:737–740
Takahashi H (1969) Light and electron microscopic studies on the pineal organ of the goldfish, Carassius auratus L. Bull Fac Fish, Hokkaido Univ 20:143–157
Tsin ATC, Liebman PA, Beatty DD, Drzymaka R (1981) Rod and cone visual pigment in the goldfish. Vision Res 21:943–946
Ueck M, Kobayashi H (1972) Vergleichende Untersuchungen über Acetylcholinesterase-haltige Neurone im Pinealorgan der Vögel. Z Zellforsch 129:140–160
Ueck M, Kobayashi H (1979) Neue Ergebnisse der vergleichenden Epiphysenforschung. Verh Anat Ges 73:961–963
Veen Th van (1981) A study on the basis for zeitgeber entrainment. With special reference to extraretinal photoreception in the eel. Thesis, Univ Lund
Veen Th van, Hartwig HG, Müller K (1976) Light-dependent motor activity and photo-negative behavior in the eel (Anguilla anguilla L.). Evidence for extraretinal and extrapineal photoreception. J Comp Physiol 111:209–219
Vigh B, Vigh-Teichmann I (1973) Comparative ultrastructure of the cerebrospinal fluid-contacting neurons. Int Rev Cytol 35:189–251
Vigh B, Vigh-Teichmann I (1981) Light- and electron-microscopic demonstration of immunoreactive opsin in the pinealocytes of various vertebrates. Cell Tissue Res 221:451–463
Vigh B, Teichmann I, Aros B (1969) Das Paraventrikularorgan und das Liquorkontaktneuronensystem. Anat Anz Suppl 125:683–688
Vigh B, Vigh-Teichmann I, Aros B (1975) Comparative ultrastructure of cerebrospinal fluid-contacting neurons and pinealocytes. Cell Tissue Res 158:409–424
Vigh B, Vigh-Teichmann I, Röhlich P, Aros B (1982) Immunoreactive opsin in the pineal organ of reptiles and birds. Z Mikrosk Anat Forsch 96:113–129
Vigh-Teichmann I, Vigh B (1974) The infundibular cerebrospinal-fluid contacting neurons. Adv Anat Embryol Cell Biol 50:1–91
Vigh-Teichmann I, Vigh B (1979) Comparison of epithalamic, hypothalamic and spinal neurosecretory terminals. Acta Biol Acad Sci Hung 30:1–39
Vigh-Teichmann I, Vigh B, Aros B (1976a) Cerebrospinal fluid-contacting neurons, ciliated perikarya and “peptidergic” synapses in the magnocellular preoptic nucleus of teleostean fishes. Cell Tissue Res 165:397–413
Vigh-Teichmann I, Vigh B, Aros B (1976b) Ciliated neurons and different types of synapses in anterior hypothalamic nuclei of reptiles. Cell Tissue Res 174:139–160
Vigh-Teichmann I, Vigh B, Röhlich P, Olsson R (1980a) Phylogenetic aspects of the sensory neurons of the wall of the diencephalon. In: Spatz M, Mrsulja BB, Rakic LjM, Lust WD (eds) Circulatory and developmental aspects of brain metabolism. Plenum Press, New York, pp 415–428
Vigh-Teichmann I, Röhlich P, Vigh B, Aros B (1980 b) Comparison of pineal complex, retina and cerebrospinal fluid contacting neurons by immunocytochemical antirhodopsin reaction. Z Mikrosk Anat Forsch 94:623–640
Vigh-Teichmann I, Korf HW, Nürnberger F, Oksche A, Vigh B (1982) Comparative immunocytochemical studies of pineal photoreceptors and CSF-contacting neurons. Verh Anat Ges 77 (in press)
Viven-Roels B, Pévet P, Dubois MP, Brown GM (1981) Immunocytochemical evidence for the presence of melatonin in the pineal gland, the retina and the Harderian gland. Cell Tissue Res 217:105–115
Wake K (1973) Acetylcholinesterase-containing nerve cells and their distribution in the pineal organ of the goldfish, Carassius auratus. Z Zellforsch 145:287–298
Wake K, Ueck M, Oksche A (1974) Acetylcholinesterase-containing nerve cells in the pineal complex and subcommissural area of the frogs, Rana ridibunda and Rana esculenta. Cell Tissue Res 154:423–442
Author information
Authors and Affiliations
Additional information
To the memory of Professor Karl von Frisch, pioneer and master in the field of photoneuroendocrine systems
This investigation was supported by grants from the Deutsche Forschungsgemeinschaft to A.O. (Ok 1/24; 1/25: Mechanismen biologischer Uhren) and to H.-W. K. (Ko 758/1; 758–2)
On leave from the 2nd Department of Anatomy, SOTE, Budapest, Hungary
Rights and permissions
About this article
Cite this article
Vigh-Teichmann, I., Korf, H.W., Oksche, A. et al. Opsin-immunoreactive outer segments and acetylcholinesterase-positive neurons in the pineal complex of Phoxinus phoxinus (Teleostei, Cyprinidae). Cell Tissue Res. 227, 351–369 (1982). https://doi.org/10.1007/BF00210891
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00210891