Abstract
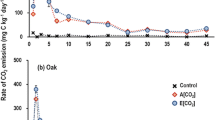
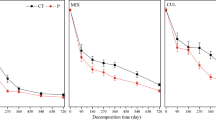
Fine root litter derived from birch (Betula pendula Roth.) and Sitka spruce (Picea sitchensis (Bong.) Carr.) plants grown under two CO2 atmospheric concentrations (350 ppm and 600 ppm) and two nutrient regimes was used for decomposition studies in laboratory microcosms. Although there were interactions between litter type, CO2/fertiliser treatments and decomposition rates, in general, an increase in the C/N ratio of the root tissue was observed for roots of both species grown under elevated CO2 in unfertilized soil. Both weight loss and respiration of decomposing birch roots were significantly reduced in materials derived from enriched CO2, whilst the decomposition of spruce roots showed no such effect.
A parallel experiment was performed using Betula pendula root litter grown under different N regimes, in order to test the relationship between C/N ratio of litter and root decomposition rate. A highly significant (p<0.001) negative correlation between C/N ratio and root litter respiration was found, with an r2=0.97. The results suggest that the increased C/N ratio of plant tissues induced by elevated CO2 can result in a reduction of decomposition rate, with a resulting increase in forest soil C stores.
Similar content being viewed by others
Abbreviations
- WTREM:
-
weight remaining
References
Adams M B, Campbell R G, Allen H L and Davey C B 1987 Root and foliar nutrient concentrations in loblolly pine: effects of season, site and fertilization. For. Sci. 33, 984–996.
Ahlström K, Persson M and Börjesson I 1988 Fertilization in a mature Scots pine (Pinus sylvestris L.) stand—effects on fine roots. Plant and Soil 106, 179–190.
Allen S E 1989 Chemical Analysis of Ecological Materials. Blackwell Scientific Publications, Oxford, UK. 368 p.
Batjes N H and Bridges E M 1992 World Inventory of Soil Emissions. Intenational Soil Reference and Information Centre, Netherlands. 204p.
Barnes J D and Pfirrmann T 1992 The influence of CO2 and O3, singly and in combination, on gas exchange, growth and nutrient status of radish (Raphanus sativus). New Phytol. 121, 403–412.
Berg B 1984 Decomposition of root litter and some factors regulating the process: long term root litter decomposition in a Scots pine forest. Soil Biol. Biochem. 16, 609–617.
Bloomfield J, Vogt K A and Vogt D J 1993 Decay rate and substrate quality of fine roots and foliage of two tropical tree species in the Luquillo Experimental Forest, Puerto Rico. Plant and Soil 150, 233–245.
Camiré C, Côté B and Brulotte S 1991 Decomposition of roots of black alder and hybrid poplar in short-rotation plantings: nitrogen and lignin control. Plant and Soil 138, 123–132.
Chu C C, Coleman J S and Mooney H A 1992 Controls of biomass partitioning between roots and shoots: Atmospheric CO2 enrichment and the acquisition and allocation of carbon and nitrogen in wild radish. Oecologia 89, 580–587.
Coûteaux M M Mousseau M, Céléier M L and Bottner P 1991 Increased atmospheric CO2 and litter quality: decomposition of sweet chestnut leaf litter with animal feed webs of different complexities. Oikos 61, 54–64.
Curtis P S, Balduman L M, Drake B G and Whigham D F 1990 Elevated atmospheric CO2 effects on belowground processes in C3 and C4 estuarine marsh communities. Ecology 71, 2001–2006.
Dai A G and Fung I Y 1993 Can climate variability contribute to the missing CO2 sink? Global Biogeochem. Cycles 7, 599–609.
Diaz S, Grime J P, Harris J and McPherson E 1993 Evidence of a feedback mechanism limiting plant response to elevated carbon dioxide. Nature 364, 616–617.
Dursun S, Ineson P, Frankland J C and Boddy L 1993 Sulphite and pH effects on CO2 evolution from decomposing angiospermous and coniferous tree leaf litters. Soil Biol. Biochem. 25, 1513–1525.
Fahey T J, Hughes J W, Pu M and Arthur M A 1988 Root decomposition and nutrient flux following whole-tree harvesting of northern hardwood forests. For. Sci. 34, 744–768.
Fog K 1988 The effect of added nitrogen on the rate of decomposition of organic matter. Biol. Rev. 63, 433–462.
Hewitt E J and Smith T A 1975 Plant Mineral Nutrition. English University Press, London. 298p.
Houghton R A 1993 Is carbon accumulating in the northern temperate zone? Global Biogeochem. Cycles 7, 611–617.
Ingestad T 1979 Nitrogen stress in birch seedlings II. N, K, P, Ca and Mg nutrition. Physiol. Plant. 45, 149–157.
Jones H E, Quarmby C and Harrison A F 1991 A root bioassay to test for nitrogen deficiency in forest trees. For. Ecol. Manage. 42, 267–282.
Joslin J D and Henderson G S 1987 Organic matter and nutrients associated with fine root turnover in a white oak stand. For. Sci. 33, 330–346.
Körner C and ArnoneIII J A 1992 Responses to elevated carbon dioxide in artifcial tropical ecosystems. Science 257, 1672–1677.
Kohen E I, Rouhier H and Mousseau M 1992 Changes in dry weight and nitrogen partitioning induced by elevated CO2 depend on soil nutrient availability in sweet chestnut (Castanea sativa Mill.). Ann. Sci. For. 49, 83–90.
Lekkerkerk L J A, Van DeGejin S C and VanVeen J A 1990 Effect of elevated atmospheric CO2-levels on the carbon economy of a soil planted with wheat. In Soils and the Greenhouse Effect. Ed. A FBouwmann. pp 423–429. John Wiley and Sons, Chichester.
Lucas P W, Cottam D A and Mansfield T A 1987 A large-scale fumigation system for investigatting interactions between air pollution and cold stress on plants. Environ. Pollut. 43, 15–28.
Maly B, Mariotti A and Morel J L 1992 Use of 13C variations at natural abundance for studying the biodegradation of root mucilage, roots and glucose in soil. Soil Biol. Biochem. 24, 1065–1072.
McClaugherty C A, Aber J D and Melillo J M 1984 Decomposition dynamics of fine roots in forested ecosystems. Oikos 42, 378–386.
Norby R J, Pastor J and Melillo J M 1986 Carbon-nitrogen interactions in CO2-enriched white oak: physiological and long term perspectives. Tree Physiol 2, 233–241.
O'Neill E G and Norby R J 1991 First year decomposition of yellow poplar leaves produced under CO2 enrichment. Bull. Ecol. Soc. Am. 72, 208.
Persson H 1979 Fine-root production, mortality and decomposition in forest ecosystems. Vegetatio 41, 101–109.
Rogers H H, Prior S A and O'Neill E G 1992 Cotton root and rhizosphere responses to free-air CO2 enrichment. Crit. Rev. Plant Sci. 11, 251–263.
Rogers H H, Runion G B and Krupa S V 1994 Plant responses to atmospheric CO2 enrichment with emphasis on roots and the rhizosphere. Environ. Pollut. 83, 155–189.
Rowland A P 1983 An automated method for the determination of ammonium N in ecological materials. Commun. Soil Sci. Plant Anal. 14, 49–63.
Santantonio D and Grace J C 1987 Estimating fine root production and tumover from biomass and decomposition data: a compartment-flow model. Can. J. For. Res. 17, 900–908.
Stulen I and Den Hertog J 1993 Root growth and functioning under atmospheric CO2 enrichment. Vegetatio 104/105, 99–115.
Taylor J 1993 The mutable carbon sink. Nature 366, 515–516.
Townend J 1993 Effects of elevated atmospheric carbon dioxide and drought on the growth and physiology of clonal spruce plants (Picea sitchensis (Bong.) Carr.). Tree Physiol. 13, 389–400.
VanVuuren M M I, Berendse F and DeVisser W 1993 Species and site differences in the decomposition of litter and roots from wet heathlands. Can. J. Bot. 71, 167–173.
Yavitt J B and Fahey T J 1982 Loss of mass and nutrient changes of decaying woody roots in Lodgepole pine forests, southeastern Wyoming. Can. J. For. Res. 12, 745–752.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cotrufo, M.F., Ineson, P. Effects of enhanced atmospheric CO2 and nutrient supply on the quality and subsequent decomposition of fine roots of Betula pendula Roth. and Picea sitchensis (Bong.) Carr.. Plant Soil 170, 267–277 (1995). https://doi.org/10.1007/BF00010479
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00010479