Abstract
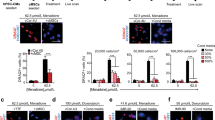
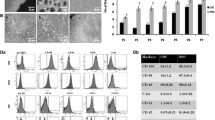
We previously showed the emergence of predominantly non-fused murine cells co-expressing cardiac and stromal determinants in co-cultures of murine mesenchymal stromal cells (MSCs) and rat embryonic cardiomyocytes. To determine whether a similar phenotype is detectable in vivo in ischemic myocardium, we infused green fluorescence protein (GFP)-marked MSCs intravenously into wild-type mice in an acute myocardial infarction (AMI) model generated by ischemia/reperfusion (I/R) or fixed coronary artery ligation. We found that infused GFP+ cells were confined strictly to ischemic areas and represented approximately 10 % of total cellularity. We showed that over 60 % of the cells co-expressed collagen type IV and troponin T or myosin heavy chain, characteristic of MSCs and cardiomyocytes, respectively, and were CD45(-). Nonetheless, up to 25 % of the GFP+ donor cells expressed one of two cardiomyocyte markers, either myosin heavy chain or troponin T, in the absence of MSC determinants. We also observed a marked reduction in OCT4 expression in MSCs pre-infusion compared with those lodged in the myocardium, suggesting reduced stem cell properties. Despite the low frequency of lodged donor MSCs, left-ventricular end-diastolic pressure was significantly better in experimental versus saline animals for both AMI (12.10 ± 1.81 vs. 20.50 ± 1.53 mmHg, p < 0.001) and I/R models (8.75 ± 2.95 vs. 17.53 ± 3.85 mmHg, p = 0.004) when measured 21 days after MSC infusion and is consistent with a paracrine effect. Our data indicate that donor MSCs undergo variable degrees of cardiomyocyte reprogramming with the majority co-expressing cardiomyocyte and stromal markers. Further studies are needed to elucidate the factors mediating the extent of cardiomyocyte reprogramming and importance of the cellular changes on tissue repair.







Similar content being viewed by others
References
Lloyd-Jones, D., Adams, R., Carnethon, M., et al. (2009). Heart disease and stroke statistics–2009 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation, 119, 480–486.
Hare, J. M., Fishman, J. E., Gerstenblith, G., et al. (2012). Comparison of allogeneic vs autologous bone marrow–derived mesenchymal stem cells delivered by transendocardial injection in patients with ischemic cardiomyopathy: the POSEIDON randomized trial. JAMA, 308, 2369–2379.
Psaltis, P. J., Zannettino, A. C., Worthley, S. G., & Gronthos, S. (2008). Concise review: mesenchymal stromal cells: potential for cardiovascular repair. Stem Cells, 26, 2201–2210.
Li, X., Yu, X., Lin, Q., et al. (2007). Bone marrow mesenchymal stem cells differentiate into functional cardiac phenotypes by cardiac microenvironment. Journal of Molecular and Cellular Cardiology, 42, 295–303.
Makino, S., Fukuda, K., Miyoshi, S., et al. (1999). Cardiomyocytes can be generated from marrow stromal cells in vitro. Journal of Clinical Investigation, 103, 697–705.
Shim, W. S., Jiang, S., Wong, P., et al. (2004). Ex vivo differentiation of human adult bone marrow stem cells into cardiomyocyte-like cells. Biochemical and Biophysical Research Communications, 324, 481–488.
Toma, C., Pittenger, M. F., Cahill, K. S., Byrne, B. J., & Kessler, P. D. (2002). Human mesenchymal stem cells differentiate to a cardiomyocyte phenotype in the adult murine heart. Circulation, 105, 93–98.
Tomita, S., Li, R. K., Weisel, R. D., et al. (1999). Autologous transplantation of bone marrow cells improves damaged heart function. Circulation, 100, II247–II256.
Keating, A. (2005). Bone marrow cells for cardiac repair. Biology of Blood and Marrow Transplantation, 11, 2–6.
Hale, S. L., Dai, W., Dow, J. S., & Kloner, R. A. (2008). Mesenchymal stem cell administration at coronary artery reperfusion in the rat by two delivery routes: a quantitative assessment. Life Sciences, 83, 511–515.
Dayan, V., Yannarelli, G., Billia, F., et al. (2011). Mesenchymal stromal cells mediate a switch to alternatively activated monocytes/macrophages after acute myocardial infarction. Basic Research in Cardiology, 106, 1299–1310.
Fazel, S., Cimini, M., Chen, L., et al. (2006). Cardioprotective c-kit+ cells are from the bone marrow and regulate the myocardial balance of angiogenic cytokines. Journal of Clinical Investigation, 116, 1865–1877.
Gallo, M. P., Ramella, R., Alloatti, G., et al. (2007). Limited plasticity of mesenchymal stem cells cocultured with adult cardiomyocytes. Journal of Cellular Biochemistry, 100, 86–99.
Lee, R. H., Pulin, A. A., Seo, M. J., et al. (2009). Intravenous hMSCs improve myocardial infarction in mice because cells embolized in lung are activated to secrete the anti-inflammatory protein TSG-6. Cell Stem Cell, 5, 54–63.
Mirotsou, M., Zhang, Z., Deb, A., et al. (2007). Secreted frizzled related protein 2 (Sfrp2) is the key Akt-mesenchymal stem cell-released paracrine factor mediating myocardial survival and repair. Proceedings of the National Academy of Sciences of the United States of America, 104, 1643–1648.
Noiseux, N., Gnecchi, M., Lopez-Ilasaca, M., et al. (2006). Mesenchymal stem cells overexpressing Akt dramatically repair infarcted myocardium and improve cardiac function despite infrequent cellular fusion or differentiation. Molecular Therapy, 14, 840–850.
Yannarelli, G., Dayan, V., Pacienza, N., et al. (2013). Human umbilical cord perivascular cells exhibit enhanced cardiomyocyte reprogramming and cardiac function after experimental acute myocardial infarction. Cell Transplantation, 22, 1651–1666. doi:10.3727/096368912X657675.
Rose, R. A., Jiang, H., Wang, X., et al. (2008). Bone marrow-derived mesenchymal stromal cells express cardiac-specific markers, retain the stromal phenotype, and do not become functional cardiomyocytes in vitro. Stem Cells, 26, 2884–2892.
Kasper, G., Dankert, N., Tuischer, J., et al. (2007). Mesenchymal stem cells regulate angiogenesis according to their mechanical environment. Stem Cells, 25, 903–910.
Bartunek, J., Croissant, J. D., Wijns, W., et al. (2007). Pretreatment of adult bone marrow mesenchymal stem cells with cardiomyogenic growth factors and repair of the chronically infarcted myocardium. American Journal of Physiology - Heart and Circulatory Physiology, 292, H1095–H1104.
Behfar, A., Yamada, S., Crespo-Diaz, R., et al. (2010). Guided cardiopoiesis enhances therapeutic benefit of bone marrow human mesenchymal stem cells in chronic myocardial infarction. Journal of the American College of Cardiology, 56, 721–734.
Okura, H., Matsuyama, A., Lee, C. M., et al. (2010). Cardiomyoblast-like cells differentiated from human adipose tissue-derived mesenchymal stem cells improve left ventricular dysfunction and survival in a rat myocardial infarction model. Tissue Engineering. Part C, Methods, 16, 417–425.
Potapova, I. A., Doronin, S. V., Kelly, D. J., et al. (2008). Enhanced recovery of mechanical function in the canine heart by seeding an extracellular matrix patch with mesenchymal stem cells committed to a cardiac lineage. American Journal of Physiology - Heart and Circulatory Physiology, 295, H2257–H2263.
Yoon, J., Min, B. G., Kim, Y. H., Shim, W. J., Ro, Y. M., & Lim, D. S. (2005). Differentiation, engraftment and functional effects of pre-treated mesenchymal stem cells in a rat myocardial infarct model. Acta Cardiologica, 60, 277–284.
Bubnic, S. J., Wang, X. H., Clark, B. R., & Keating, A. (2002). W/Wv marrow stromal cells engraft and enhance early erythropoietic progenitors in unconditioned Sl/Sld murine recipients. Bone Marrow Transplantation, 30, 867–872.
Desjardins, J. F., Pourdjabbar, A., Quan, A., et al. (2009). Lack of S100A1 in mice confers a gender-dependent hypertensive phenotype and increased mortality after myocardial infarction. American Journal of Physiology - Heart and Circulatory Physiology, 296, H1457–H1465.
Tsoporis, J. N., Marks, A., Haddad, A., Dawood, F., Liu, P. P., & Parker, T. G. (2005). S100B expression modulates left ventricular remodeling after myocardial infarction in mice. Circulation, 111, 598–606.
Liedtke, S., Enczmann, J., Waclawczyk, S., Wernet, P., & Kögler, G. (2007). Oct4 and its pseudogenes confuse stem cell research. Cell Stem Cell, 1, 364–366.
Maniatis, T., Sambrook, J., & Fritsch, E. F. (1989). Molecular Cloning: A Laboratory Manual (2nd ed., pp. 18.38–18.39). New York: Cold Spring Harbour Laboratory Press.
Dominici, M., Le Blanc, K., Mueller, I., et al. (2006). Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy, 8, 315–317.
Keating, A. (2012). Mesenchymal stromal cells: new directions. Cell Stem Cell, 10, 709–716.
Jackson, K. A., Majka, S. M., Wang, H., et al. (2001). Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells. Journal of Clinical Investigation, 107, 1395–1402.
Orlic, D., Kajstura, J., Chimenti, S., et al. (2001). Bone marrow cells regenerate infarcted myocardium. Nature, 410, 701–705.
Orlic, D., Kajstura, J., Chimenti, S., et al. (2001). Mobilized bone marrow cells repair the infarcted heart, improving function and survival. Proceedings of the National Academy of Sciences of the United States of America, 98, 10344–10349.
Yeh, E. T., Zhang, S., Wu, H. D., Körbling, M., Willerson, J. T., & Estrov, Z. (2003). Transdifferentiation of human peripheral blood CD34+-enriched cell population into cardiomyocytes, endothelial cells, and smooth muscle cells in vivo. Circulation, 108, 2070–2073.
Martin-Rendon, E., Brunskill, S. J., Hyde, C. J., Stanworth, S. J., Mathur, A., & Watt, S. M. (2008). Autologous bone marrow stem cells to treat acute myocardial infarction: a systematic review. European Heart Journal, 29, 1807–1818.
Balsam, L. B., Wagers, A. J., Christensen, J. L., Kofidis, T., Weissman, I. L., & Robbins, R. C. (2004). Haematopoietic stem cells adopt mature haematopoietic fates in ischaemic myocardium. Nature, 428, 668–673.
Murry, C. E., Soonpaa, M. H., Reinecke, H., et al. (2004). Haematopoietic stem cells do not transdifferentiate into cardiac myocytes in myocardial infarcts. Nature, 428, 664–668.
Ohtsuka, S., & Dalton, S. (2008). Molecular and biological properties of pluripotent embryonic stem cells. Gene Therapy, 15, 74–81.
Silva, J., & Smith, A. (2008). Capturing pluripotency. Cell, 132, 532–536.
Camara-Clayette, V., Le Pesteur, F., Vainchenker, W., & Sainteny, F. (2006). Quantitative Oct4 overproduction in mouse embryonic stem cells results in prolonged mesoderm commitment during hematopoietic differentiation in vitro. Stem Cells, 24, 1937–1945.
Niwa, H., Miyazaki, J., & Smith, A. G. (2000). Quantitative expression of Oct-3/4 defines differentiation, dedifferentiation or self-renewal of ES cells. Nature Genetics, 24, 372–376.
Greco, S. J., Liu, K., & Rameshwar, P. (2007). Functional similarities among genes regulated by OCT4 in human mesenchymal and embryonic stem cells. Stem Cells, 25, 3143–3154.
Ramkisoensing, A. A., Pijnappels, D. A., Askar, S. F., et al. (2011). Human embryonic and fetal mesenchymal stem cells differentiate toward three different cardiac lineages in contrast to their adult counterparts. PLoS One, 6, e24164.
Acknowledgments
We are grateful to Huijie Xiang, University Health Network for outstanding technical assistance, to Qi-Long Yi (Biostatistics Department, Princess Margaret Hospital/Ontario Cancer Institute) for assistance in statistical analysis and to Ye Yang, Department of Pathology, University Health Network for technical assistance in preparation of histological sections. A.K holds the Gloria and Seymour Epstein Chair in Cell Therapy and Transplantation at the University Health Network and the University of Toronto.
Author Contributions
Conceived and designed the experiments: GY, JNT, AK. Performed the experiments: GY, JNT, JFD, XHW, AP. Analyzed the data: GY, JNT. Contributed reagents/materials/analysis tools: SV, TGP, AK. Wrote the paper: GY, JNT, SV, AK.
Conflict of Interest
The authors declare that they have no competing interests.
Funding
This work was supported by the Orsino Translational Research Laboratory, PMH. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Gustavo Yannarelli and James N. Tsoporis contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 1389 kb)
Rights and permissions
About this article
Cite this article
Yannarelli, G., Tsoporis, J.N., Desjardins, JF. et al. Donor Mesenchymal Stromal Cells (MSCs) Undergo Variable Cardiac Reprogramming in Vivo and Predominantly Co-Express Cardiac and Stromal Determinants after Experimental Acute Myocardial Infarction. Stem Cell Rev and Rep 10, 304–315 (2014). https://doi.org/10.1007/s12015-013-9483-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-013-9483-y