Abstract
Purpose
Transnasal administration is one of the most common routes for allergen challenge in mouse models of airway diseases. Although this technique is widely used, neither the amount of allergen that reaches the lung nor its airway distribution has been well established. We used positron emission tomography (PET) and computed tomography (CT) to examine the anatomical distribution of a solution containing a tracer immediately after transnasal delivery and to determine the possible influence of age and administered volume.
Procedures
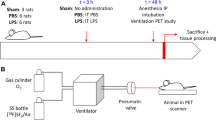
Forty-six female BALB/c mice were divided into three groups according to instillation volume and age: (A) 15 μl, 8–10 weeks old (N = 10), (B) 30 μl, 8–10 weeks old (N = 20), and (C) 30 μl, 32 weeks old (N = 16). Anesthetized animals underwent a dynamic scan in a dedicated small-animal PET scanner immediately after transnasal administration of a solution containing 18FDG. Regions of interest were used to obtain quantitative data. Animals were also imaged with a small-animal CT scanner to obtain complementary anatomical information.
Results
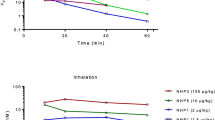
Mean ± SD (5.69 ± 4.51%) of the solution administered reached the lungs in group A, 41.84 ± 8.03% in group B, and 36.65 ± 16.15% in group C. A comparable percentage was delivered to the left and right lungs in all the groups. Analysis of variance revealed a significant difference between the groups in the proportion of the solution that reached the lungs depending on the injection volume (P < 0.001), but not depending on animal age.
Conclusions
In this first report on quantitative imaging by PET and CT in small animals, we confirmed the suitability of the transnasal route with an instilled volume of 30 μl delivering fluids into the lower airways, although only about 40% of the dose reaches the lungs.




Similar content being viewed by others
References
Amy RW, Bowes D, Burri PH, Haines J et al (1977) Postnatal growth of the mouse lung. J Anat 124(Pt 1):131–151
Arun KS, Huang TS, Blostein SD (1987) Least-squares fitting of two 3-D point sets. IEEE Trans Pattern Anal Machine Itell, PAMI 9(5):698–700
Campbell EM, Kunkel SL, Strieter RM, Lukacs NW (1998) Temporal role of chemokines in a murine model of cockroach allergen-induced airway hyperreactivity and eosinophilia. J Immunol 161(12):7047–7053
Conejero L, Higaki Y, Baeza ML, Fernandez M et al (2007) Pollen-induced airway inflammation, hyper-responsiveness and apoptosis in a murine model of allergy. Clin Exp Allergy 37(3):331–338
Costa-Pinto FA, Basso AS, Russo M (2007) Role of mast cell degranulation in the neural correlates of the immediate allergic reaction in a murine model of asthma. Brain Behav Immun 21(6):783–790
Havaux X, Zeine A, Dits A, Denis O (2005) A new mouse model of lung allergy induced by the spores of Alternaria alternata and Cladosporium herbarum molds. Clin Exp Immunol 139(2):179–188
Hogaboam CM, Blease K, Mehrad B, Steinhauser ML et al (2000) Chronic airway hyperreactivity, goblet cell hyperplasia, and peribronchial fibrosis during allergic airway disease induced by Aspergillus fumigatus. Am J Pathol 156(2):723–732
Kawakami M, Paul JL, Thurlbeck WM (1984) The effect of age on lung structure in male BALB/cNNia inbred mice. Am J Anat 170(1):1–21
Lopez E, del Pozo V, Miguel T, Sastre B et al (2006) Inhibition of chronic airway inflammation and remodeling by galectin-3 gene therapy in a murine model. J Immunol 176(3):943–950
Maes F, Collignon A, Vandermeulen D, Marchal G et al (1997) Multimodality image registration by maximization of mutual information. IEEE Trans Med Imaging 16(2):187–198
Sakai K, Yokoyama A, Kohno N, Hamada H et al (2001) Prolonged antigen exposure ameliorates airway inflammation but not remodeling in a mouse model of bronchial asthma. Int Arch Allergy Immunol 126(2):126–134
Shinagawa K, Kojima M (2003) Mouse model of airway remodeling: strain differences. Am J Respir Crit Care Med 168(8):959–967
Tomaki M, Zhao LL, Lundahl J, Sjostrand M et al (2000) Eosinophilopoiesis in a murine model of allergic airway eosinophilia: involvement of bone marrow IL-5 and IL-5 receptor alpha. J Immunol 165(7):4040–4050
Vanoirbeek JA, Tarkowski M, Vanhooren HM, De Vooght V et al (2006) Validation of a mouse model of chemical-induced asthma using trimellitic anhydride, a respiratory sensitizer, and dinitrochlorobenzene, a dermal sensitizer. J Allergy Clin Immunol 117(5):1090–1097
Vaquero JJ, Lage E, Redondo S, Abella M et al (2005) Co-planar PET/CT for small animal imaging. IEEE Nuclear Science Symposium Conference Record (CD-ROM), pp 1748–1751
Vaquero JJ, Redondo S, Lage E, Abella M et al (2008) Assessment of a new high-performance small-animal X-ray tomograph. IEEE T Nucl Sci 55(3):898–905
Wang Y, Seidel J, Tsui BMW, Vaquero JJ et al (2006) Performance evaluation of the GE healthcare explore vista dual-ring small-animal pet scanner. J Nucl Med 47:1891–1900
Yiamouyiannis CA, Schramm CM, Puddington L, Stengel P et al (1999) Shifts in lung lymphocyte profiles correlate with the sequential development of acute allergic and chronic tolerant stages in a murine asthma model. Am J Pathol 154(6):1911–1921
Zingone A, Seidel J, Aloj L, Caraco C et al (2002) Monitoring the correction of glycogen storage disease type 1a in a mouse model using [(18)F]FDG and a dedicated animal scanner. Life Sci 71(11):1293–1301
Zosky GR, Sly PD (2007) Animal models of asthma. Clin Exp Allergy 37(7):973–88
Acknowledgments
This work was supported by grants from Ministerio de Ciencia y Tecnología (TEC2004-07052-C02-01), Ministerio de Sanidad y Consumo (CIBER CB06/01/0079 CIBER CB07/09/0031 and FIS 2001/0598), Ministerio de Industria (CDTEAM Project) and Fundación de Investigación Médica Mutua Madrileña.
Author information
Authors and Affiliations
Corresponding author
Additional information
L Conejero was with the Hospital General Universitario Gregorio Marañón. She is now with the Immunology Unit, Department of Infectious and Tropical Diseases, London School of Hygiene & Tropical Medicine, London, UK.
Rights and permissions
About this article
Cite this article
Soto-Montenegro, M., Conejero, L., Vaquero, J. et al. Assessment of Airway Distribution of Transnasal Solutions in Mice by PET/CT Imaging. Mol Imaging Biol 11, 263–268 (2009). https://doi.org/10.1007/s11307-009-0199-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-009-0199-y