Abstract
Purpose
Clinical ventilation studies are primarily performed with computerized tomography (CT) and more often with single-photon emission Computerized tomography (SPECT) using radiolabelled aerosols, both presenting certain limitations. Here, we investigate the use of the radiofluorinated gas [18F]SF6 as a positron emission tomography (PET) ventilation marker in an animal model of impaired lung ventilation.
Procedures
Sprague–Dawley rats (n = 15) were randomly assigned to spontaneous ventilation (sham group), endotracheal administration of phosphate-buffered saline (PBS group), or endotracheal administration of lipopolysaccharide (LPS group). PET-[18F]SF6 images (10-min acquisition) were acquired at t = 48 h after LPS or PBS administration under mechanical ventilation. CT images were acquired after each PET session. Volumes of interest were manually delineated in the lungs on CT images, and voxel-by-voxel analysis was carried out on PET images to obtain the corresponding histograms. After the imaging sessions, lungs were harvested to conduct histological analysis.
Results
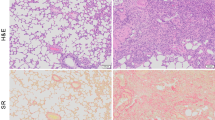
Ventilation studies in sham animals showed uniform distribution of [18F]SF6 and fast elimination of the radioactivity after discontinuation of the administration. For PBS- and LPS-treated rats, ventilation defects were observed on PET images in some animals, identified as regions with low presence of the radiolabelled gas. Hypoventilated areas co-localized with regions with higher x-ray attenuation than healthy lungs on the CT images, suggesting the presence of oedema and, in some cases, atelectasis. Histograms obtained from PET images showed quasi-Gaussian distributions for control animals, while PBS- and LPS-treated animals demonstrated the presence of hypoventilated voxels. Deviation of the histograms from Gaussian distribution correlated with histological score was obtained by ex vivo histological analysis.
Conclusions
[18F]SF6 is an appropriate marker of regional lung ventilation and may find application in the early diagnose of acute lung disease.




Similar content being viewed by others
References
Vinogradskiy Y. (2019) CT-based ventilation imaging in radiation oncology. BJR|Open.1:20180035.
Fazio F, Jones T, Tofts P, Giuntini C (1978) Studies of regional ventilation using 81(m)Kr. Am Rev Respir Dis 117:112
Jögi J, Jonson B, Ekberg M, Bajc M (2010) Ventilation-perfusion SPECT with 99mTc-DTPA versus Technegas: a head-to-head study in obstructive and nonobstructive disease. J nuclear medicine : official publication, Society of Nucl Med 51:735–741
Burch WM, Sullivan PJ, Lomas FE et al (1986) Lung ventilation studies with technetium-99m pseudogas. J Nucl Med 27:842–846
Burch WM, Sullivan PJ, McLaren CJ (1986) Technegas — a new ventilation agent for lung scanning. Nucl Med Commun 7:865–871
Coghe J, Votion D, Lekeux P (2000) Comparison between radioactive aerosol, technegas and krypton for ventilation imaging in healthy calves. Veterinary Journal 160:25–32
Bajc M, Schümichen C, Grüning T et al (2019) EANM guideline for ventilation/perfusion single-photon emission computed tomography (SPECT) for diagnosis of pulmonary embolism and beyond. Eur J Nucl Med Mol Imaging 46:2429–2451
Hofman MS, Beauregard JM, Barber TW et al (2011) 68Ga PET/CT ventilation-perfusion imaging for pulmonary embolism: a pilot study with comparison to conventional scintigraphy. J Nucl Med 52:1513–1519
Ament SJ, Maus S, Reber H et al (2013) PET lung ventilation/perfusion imaging using 68Ga aerosol (Galligas) and 68Ga-Labeled macroaggregated albumin. Recent Results Cancer Res 194:395–423
Le Roux PY, Hicks RJ, Siva S, Hofman MS (2019) PET/CT lung ventilation and perfusion scanning using Galligas and Gallium-68-MAA. Semin Nucl Med 49:71–81
Oehme L, Zöphel K, Golgor E et al (2014) Quantitative analysis of regional lung ventilation and perfusion PET with 68Ga-labelled tracers. Nucl Med Commun 35:501–510
Schreiber WG, Eberle B, Laukemper-Ostendorf S et al (2001) Dynamic 19F-MRI of pulmonary ventilation using sulfur hexafluoride SF6 gas. Magn Reson Med 45:605–613
Gómez-Vallejo V, Lekuona A, Baz Z et al (2016) Ion beam induced 18F-radiofluorination: straightforward synthesis of gaseous radiotracers for the assessment of regional lung ventilation using positron emission tomography. Chem Commun 52:11931–11934
Cossío U, Gómez-Vallejo V, Flores M et al (2018) Preclinical evaluation of aerosol administration systems using positron emission tomography. Eur J Pharm Biopharm 130:59–65
Blázquez-Prieto J, López-Alonso I, Amado-Rodríguez L et al (2015) Exposure to mechanical ventilation promotes tolerance to ventilator-induced lung injury by Ccl3 downregulation. Am J Physiol Lung Cell Mol Physiol 309:21
Olsson LE, Smailagic A, Önnervik PO, Hockings PD (2009) 1H and hyperpolarized3He MR imaging of mouse with LPS-induced inflammation. J Magn Reson Imaging 29:977–981
Thomas AC, Nouls JC, Driehuys B et al (2011) Ventilation defects observed with hyperpolarized 3He magnetic resonance imaging in a mouse model of acute lung injury. Am J Respir Cell Mol Biol 44:648–654
Guerrero T, Sanders K, Castillo E et al (2006) Dynamic ventilation imaging from four-dimensional computed tomography. Phys Med Biol 51:777–791
Reinhardt JM, Ding K, Cao K et al (2008) Registration-based estimates of local lung tissue expansion compared to xenon CT measures of specific ventilation. Med Image Anal 12:752–763
Stewart NJ, Smith LJ, Chan H-F, et al (2022) Lung MRI with hyperpolarised gases current & future clinical perspectives. Br J Radiol 0:20210207
Pike D, Kirby M, Eddy RL et al (2016) Regional heterogeneity of chronic obstructive pulmonary disease phenotypes pulmonary 3He magnetic resonance imaging and computed tomography. COPD J Chronic Obstructive Pulmonary Disease 13:601–609
Kirby M, Mathew L, Wheatley A et al (2010) Chronic obstructive pulmonary disease: longitudinal hyperpolarized 3He MR imaging. Radiology 256:280–289
Adams CJ, Capaldi DPI, Di Cesare R, McCormack DG, Parraga G (2018) On the potential role of MRI biomarkers of COPD to guide Bronchoscopic Lung Volume Reduction. Acad Radiol 25:159–168
de Lange EE, Altes TA, Patrie JT et al (2006) Evaluation of asthma with hyperpolarized Helium-3 MRI: correlation with clinical severity and spirometry. Chest 130:1055–1062
Mummy DG, Carey KJ, Evans MD et al (2020) Ventilation defects on hyperpolarized helium-3 MRI in asthma are predictive of 2-year exacerbation frequency. J Allergy and Clinical Immunology 146:831-839.e836
Thomen RP, Walkup LL, Roach DJ et al (2017) Hyperpolarized 129Xe for investigation of mild cystic fibrosis lung disease in pediatric patients. J Cyst Fibros 16:275–282
Kanhere N, Couch MJ, Kowalik K et al (2017) Correlation of lung clearance index with hyperpolarized 129Xe magnetic resonance imaging in pediatric subjects with cystic fibrosis. Am J Respir Crit Care Med 196:1073–1075
Hegi-Johnson F, de Ruysscher D, Keall P et al (2019) Imaging of regional ventilation: is CT ventilation imaging the answer? A systematic review of the validation data. Radiother Oncol 137:175–185
Kawakami K, Iwamura A, Goto E et al (1990) Kinetics and clinical application of 99mTc-technegas. Kakuigaku 27:725–733
Wolf U, Scholz A, Terekhov M, et al (2008) Fluorine-19 MRI of the lung: first human experiment In: Annual Meeting of the International Society for Magnetic Resonance Imaging Toronto, Ontario, Canada,
Semenova V, Pavlova O, Pirogov Y. (2020) Magnetic resonance imaging of lungs using fluorinated gas. In: Journal of Physics: Conference Series 1439 012043
Gutberlet M, Kaireit TF, Voskrebenzev A et al (2018) Free-breathing dynamic (19)F gas MR imaging for mapping of regional lung ventilation in patients with COPD. Radiology 286:1040–1051
Fain SB, Panth SR, Evans MD et al (2006) Early emphysematous changes in asymptomatic smokers: detection with 3He MR imaging. Radiology 239:875–883
Kirby M, Svenningsen S, Ahmed H et al (2011) Quantitative evaluation of hyperpolarized helium-3 magnetic resonance imaging of lung function variability in cystic fibrosis. Acad Radiol 18:1006–1013
He M, Kaushik SS, Robertson SH et al (2014) Extending semiautomatic ventilation defect analysis for hyperpolarized (129)Xe ventilation MRI. Acad Radiol 21:1530–1541
Funding
The work was supported by MCIN/AEI/10.13039/501100011033 (PDC2021-121497-I00 and PID2020-117656RB-I00) and Instituto de Salud Carlos III (DTS21/00008). This work was performed under the Maria de Maeztu Units of Excellence Programme — Grant MDM-2017–0720 funded by MCIN/AEI/10.13039/501100011033 and RYC-2017–22412.
Author information
Authors and Affiliations
Contributions
J. L., A. M., and L. R. designed the study, interpreted the data, and wrote the manuscript. V. G.-V., A. L., and V. S. performed the radiochemical syntheses. R. P. and U.C. performed the PET-CT experiments. L. A.-R. and G. M. A., performed the ex vivo analysis of tissue samples and contributed to the interpretation of the data and writing of the manuscript. J. R. C. contributed to the interpretation of the images and writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
Jordi Llop, Vanessa Gómez-Vallejo, and Aitor Lekuona are co-inventors of the patent that protects the production process for the radiofluorinated gas (PCT/EP2017/065900) granted in the USA (US10849994B2) and Europe (EP3474902B1). No other potential conflicts of interest relevant to this article exist.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Passannante, R., Gómez-Vallejo, V., Cossío, U. et al. Assessment of Regional Lung Ventilation with Positron Emission Tomography Using the Radiofluorinated Gas [18F]SF6: Application to an Animal Model of Impaired Ventilation. Mol Imaging Biol 25, 413–422 (2023). https://doi.org/10.1007/s11307-022-01773-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-022-01773-7