Abstract
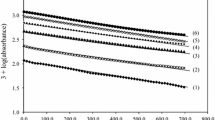
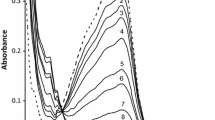
Kinetics of hexachloroplatinate(IV) (HCP) oxidation of l-histidine (His) in H2SO4 medium has been investigated in the absence and presence of silver(I) catalyst at constant ionic strength 2.0 mol dm−3 and temperature 25 °C. The progress of both uncatalyzed and silver(I)-catalyzed oxidation reactions has been monitored spectrophotometrically. Both uncatalyzed and catalyzed paths show first-order dependence on [HCP] and fractional-first-order dependence each on [His] and [acid]. The catalyzed path is first order in [AgI]. Increasing ionic strength and dielectric constant decreases the oxidation rates. The catalyzed reaction has been shown to proceed via formation of a silver(I)-histidine intermediate complex, which reacts with the oxidant by an inner-sphere mechanism leading to decomposition of the complex in the rate-determining step. The final oxidation products of histidine were identified as the corresponding aldehyde (2-imidazole acetaldehyde), ammonium ion and carbon dioxide. The mechanisms of these reactions have been proposed and the appropriate rate laws are deduced.





Similar content being viewed by others
References
Farrel N, Ugo R, James BR (eds) (1989) Catalysis by metal complexes, vol 11. Kluwer, Dordrecht
Weiss RB, Christian MC (1993) New cisplatin analogues in development. A review. Drugs 46:360–377
Keage MC, Kelland MJ, Neidles LR, Warning MJ (eds) (1993) Molecular aspects of anticover drug DNA interactions, vol 1. CRC Press, New York
Mehrotra US, Agarwal MC, Mushran SP (1970) J Inorg Nucl Chem 32:2325
Sen Gupta KK, Sen PK, Sen Gupta S (1977) Inorg Chem 16:1396
Sen Gupta KK, Sen PK (1977) J Inorg Nucl Chem 39:1651
Sen Gupta KK, Das S, Sen Gupta S (1988) Trans Met Chem 13:155
Pal B, Sen Gupta KK (2000) Bull Chem Soc Japan 73:553
Sen Gupta KK, Begum BA, Ghosh SP (1998) Trans Met Chem 23:295
Sen Gupta KK, Begum BA, Pal B (1998) Carbohydr Res 309:303
Pal B, Sen Gupta KK, Sen PK (2005) Trans Met Chem 30:593
Choi S, Filotto C, Bisanzo M, Delaney S, Lagasee D, Whitworth JL, Jusko A, Li C, Wood NA, Willingham J, Schwenker A, Spaulding K (1988) Inorg Chem 37:2500
Fawzy A. (2015) Int J Chem Kinet 47:1–12
Fawzy A (2014) Trans Met Chem 39:567
Gangopadhyay S, Ali M, Banerjee P (1992) J Chem Soc Perkin Trans 2:781
Shukla R, Upadhyay SK (2008) Ind J Chem 47A:551
Ewais HA, Habib MA, Elroby SAK (2010) Trans Met Chem 35:73
Sony V, Sindal RS, Mehrotra RN (2005) Polyhedron 24:1167
Jose TP, Nandibewoor ST, Tuwar SM (2005) E J Chem 2:75
Gupta KC, Gupta K (1985) Int J Chem Kinet 17:769
Iloukhani H, Moazenzadeh M (2001) Phys Chem Liq 39:429
Chen GN, Wu XP, Duan JP, Chen HQ (1999) Talanta 49:319
Henry B, Boubel JC, Delpuech JJ (1986) Inorg Chem 25:623
Sapper H, Paul HH, Beinhauer K, Lohmann W (1985) Inorg Chim Acta 106:25
Czoik R, Heintz A, John E, Marczak W (2008) Acta Phys Polon A 114:A-49
Georgieva M, Andonovski B (2003) Anal Bioanal Chem 375:836
Vogel AI (1973) In Text book of practical organic chemistry, 3rd edn. ELBS Longman, London, p 332 and 679
Lohdip YN, Iyum JF (1998) Bull Chem Soc Ethiopia 12:113
Ghosh MC, Reed JW, Bose RN, Gould ES (1994) Inorg Chem 33:73
Pal B, Sen Gupta KK (2012) Trans Met Chem 37:671
Shi T, Berglund J, Elding LI (1996) Inorg Chem 35:3498
Martell AE, Smith RM (1974) Critical stability constants, vol I. Plenum Press, New York, p 321
Kramer J, Koch KR (2006) Inorg Chem 45:7843
Lemma K, Sargeson AM, Elding LI (2000) J Chem Soc Dalton Trans 7:1167
Latimer WM (1961) Oxidation potentials, 2nd edn. Pentice Hall, USA, p 206
Hassan RM, Kojima T, Fukutomi H (1982) VI International symposium on solute–solute–solvent interactions. Japan, p 113
Frost AA, Person RG (1970) Kinetics and mechanism. Wiley Eastern, New Delhi, p 147
Rochester CH (1971) Progress in reaction kinetics. Pergamon Press, Oxford, p 145
Laidler K (1965) Chemical kinetics. McGraw-Hill, New York
Amis ES (1966) Solvent effects on reaction rates and mechanism. Academic Press, New York, p 28
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fawzy, A., Asghar, B.H. Kinetics and mechanism of uncatalyzed and silver(I)-catalyzed oxidation of l-histidine by hexachloroplatinate(IV) in acid medium. Transition Met Chem 40, 287–295 (2015). https://doi.org/10.1007/s11243-015-9916-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-015-9916-3