Abstract
Background and aims
Freeze events can strongly influence many ecosystem processes. However, the effects of freeze events on litter production, litter quality, and decomposition are rarely documented.
Methods
In this study, litter fall was measured monthly for 2 years. Two litter decomposition experiments were also performed using freeze-damaged litter and non-damaged litter in a loblolly pine forest.
Results
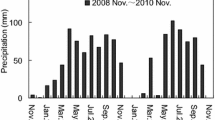
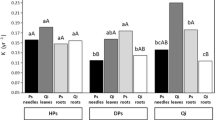
The freeze event in November 2009 caused a pronounced pulse of needle litter fall. The freeze-damaged needle litter was shown to have higher N concentration and lower C/N ratio compared with the normal falling needle litter. This finding indicates that freeze damage significantly increased needle quality because of incomplete nutrient resorption. The decomposition of freeze-damaged needle litter was faster than that of normal falling yellow needle litter and slower than that of hand-picked green needle litter. The decomposition rate constant (k) was negatively correlated with the C/N ratio in the needle litter. Our results also showed that the different climatic conditions influence patterns of litter decomposition.
Conclusions
This study suggests that freeze events significantly alter litter quantity and quality, thus affecting litter decomposition rates in a loblolly pine forest in central China.





Similar content being viewed by others
References
Aerts R (1996) Nutrient resorption from senescing leaves of perennials: are there general patterns? J Ecol 84:597–608
Aerts R, Chapin FS (2000) The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. Adv Ecol Res 30:1–67
Augspurger CK (2009) Spring 2007 warmth and frost: phenology, damage and refoliation in a temperate deciduous forest. Funct Ecol 23:1031–1039
Berg B, Ekbohm G (1991) Litter mass loss rates and decomposition patterns in some needle and leaf litter types. Long-term decomposition in a Scots pine forest VII. Can J Bot 69:1449–1456
Berg B, McClaugherty C (2008) Plant litter: decomposition, humus formation, carbon sequestration. Springer, Berlin
Berg B, Ekbohm G, Johansson M-B, McClaugherty C, Rutigliano F, Virzo De Santo A (1996) Some foliar litter types have a maximum limit for decomposition – a synthesis of data from forest systems. Can J Bot 74:659–672
Berg B, Virzo De Santo A, Rutigliano F, Ekbohm G (2003) Limit values for plant litter decomposing in two contrasting soils–influence of litter elemental composition. Acta Oecol 24:295–302
Bonanomi G, Incerti G, Antignani V, Capodilupo M, Mazzoleni S (2010) Decomposition and nutrient dynamics in mixed litter of Mediterranean species. Plant Soil 331:481–496
Bonanomi G, Incerti G, Giannino F, Mingo A, Lanzotti V, Mazzoleni S (2013) Litter quality assessed by solid state 13C NMR spectroscopy predicts decay rate better than C/N and lignin/N ratios. Soil Biol Biochem 56:40–48
Breeuwer A, Heijmans M, Robroek BJM, Limpens J, Berendse F (2008) The effect of increased temperature and nitrogen deposition on decomposition in bogs. Oikos 117:1258–1268
Caritat A, García-Berthou E, Lapeña R, Vilar L (2006) Litter production in a Quercus suber forest of Montseny (NE Spain) and its relationship to meteorological conditions. Ann For Sci 63:791–800
Cavender-Bares J (2007) Chilling and freezing stress in live oaks (Quercus section Virentes): intra- and inter-specific variation in PSII sensitivity corresponds to latitude of origin. Photosynth Res 94:437–453
Cavender-Bares J, Cortes P, Rambal S, Joffre R, Miles B, Rocheteau A (2005) Summer and winter sensitivity of leaves and xylem to minimum freezing temperatures: a comparison of co-occurring Mediterranean oaks that differ in leaf lifespan. New Phytol 168:597–611
Chen H, Rygiewicz PT, Johnson MG, Harmon ME, Tian H, Tang JW (2008) Chemistry and long-term decomposition of roots of Douglas-fir grown under elevated atmospheric carbon dioxide and warming conditions. J Environ Qual 37:1327–1336
Cheng L, Fuchigami LH (2002) Growth of young apple trees in relation to reserve nitrogen and carbohydrates. Tree Physiol 22:1297–1303
Cotrufo MF, Briones MJI, Ineson P (1998) Elevated CO2 affects field decomposition rate and palatability of tree leaf litter: importance of changes in substrate quality. Soil Biol Biochem 30:1565–1571
Dale VH, Jouce LA, Mcnulty S, Neilson RP, Ayres MP, Flannigan MD, Hanson PJ, Irland LC, Lugo AE, Peterson CJ, Simberloff D, Swanson FJ, Stocks BJ, Wotton B (2001) Climate change and forest disturbances. Bioscience 51:723–734
Ellis WL, Bowles JW, Erickson AA, Stafford N, Bell SS, Thomas M (2006) Alteration of the chemical composition of mangrove (Laguncularia racemosa) leaf litter fall by freeze damage. Estuar Coast Shelf Sci 68:363–371
Escudero A, Mediavilla S (2003) Decline in photosynthetic nitrogen use efficiency with leaf age and nitrogen resorption as determinants of leaf life span. J Ecol 91:880–889
Freschet GT, Cornelissen JHC, van Logtestijn RSP, Aerts R (2010) Substantial nutrient resorption from leaves, stems and roots in a subarctic flora: what is the link with other resource economics traits? New Phytol 186:879–889
González-Rodríguez ÁM, Jiménez MS, Morales D (2005) Seasonal and intraspecific variation of frost tolerance in leaves of three Canarian laurel forest tree species. Ann For Sci 62:423–428
Gu LH, Hanson PJ, Mac Post W, Kaiser DP, Yang B, Nemanl R, Pallardy SG, Meyers T (2008) The 2007 eastern US spring freezes: increased cold damage in a warming world? Bioscience 58:253–262
Hättenschwiler S, Coq S, Barantal S, Handa IT (2011) Leaf traits and decomposition in tropical rainforests: revisiting some commonly held views and towards a new hypothesis. New Phytol 189:950–965
Houlton BZ, Driscoll CT, Fahey TJ, Likens GE, Groffman PM, Bernhardt ES, Buso DC (2003) Nitrogen dynamics in ice storm-damaged forest ecosystems: implications for nitrogen limitation theory. Ecosystems 6:431–443
IPCC (2007) Climate change 2007: the physical sciences basis: contribution of working group I to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
Jacob M, Viedenz K, Polle A, Thomas FM (2010) Leaf litter decomposition in temperate deciduous forest stands with a decreasing fraction of beech (Fagus sylvatica). Oecologia 164:1083–1094
Killingbeck KT (1996) Nutrients in senesced leaves: keys to the search for potential resorption and resorption proficiency. Ecology 77:1716–1727
King JS, Pregitzer KS, Zak DR, Kubiske ME, Ashby JA, Holmes WE (2001) Chemistry and decomposition of litter from Populus tremuloides Michaux grown at elevated atmospheric CO2 and varying N availability. Global Change Biol 7:65–74
Lü XT, Freschet GT, Flynn DFB, Han XG (2012) Plasticity in leaf and stem nutrient resorption proficiency potentially reinforces plant–soil feedbacks and microscale heterogeneity in a semi-arid grassland. J Ecol 100:144–150
Meentemeyer V (1978) Macroclimate and lignin control of litter decomposition rates. Ecology 59:465–472
Murphy KL, Klopatek JM, Klopatek CC (1998) The effects of litter quality and climate on decomposition along an elevational gradient. Ecol Appl 8:1061–1071
Norby RJ, Long TM, Hartz-Rubin JS, O’Neill EG (2000) Nitrogen resorption in senescing tree leaves in a warmer, CO2-enriched atmosphere. Plant Soil 224:15–29
Olson JS (1963) Energy storage and the balance of producers and decomposers in ecological systems. Ecology 44:322–331
Pérez-Suárez M, Arredondo-Moreno JT, Huber-Sannwald E, Vargas-Hernández JJ (2009) Production and quality of senesced and green litterfall in a pine–oak forest in central-northwest Mexico. Forest Ecol Manag 258:1307–1315
Rigby JR, Porporato A (2008) Spring frost risk in a changing climate. Geophys Res Lett 35:L12703. doi:10.1029/2008GL033955
Sherman RE, Fahey TJ, Howarth RW (1998) Soil-plant interactions in a neotropical mangrove forest: iron, phosphorous and sulfur dynamics. Oecologia 115:553–563
van Heerwaarden LM, Toet S, Aerts R (2003) Current measures of nutrient resorption efficiency lead to a substantial underestimation of real resorption efficiency: facts and solutions. Oikos 101:664–669
Wang WQ, You SY, Wang YB, Huang L, Wang M (2011) Influence of frost on nutrient resorption during leaf senescence in a mangrove at its latitudinal limit of distribution. Plant Soil 342:105–115
Wu B (2008) Ideas of restoration and reconstruction after frozen rain and snow in South China. Sci Silvae Sin 44:2–4 (in Chinese)
Zheng YP, Yang QP, Xu M, Chi YG, Shen RC, Li PX, Dai HT (2012) Responses of Pinus massoniana and Pinus taeda to freezing in temperate forests in central China. Scand J For Res 27:520–531
Acknowledgments
This research was supported by the National Key Research and Development Program (2010CB833503), the Forestry Department of Sichuan Province (Forest Carbon Monitoring and Accounting Project, 2009-204) and National Natural Science Foundation of China (31200302). We thank two anonymous reviewers for their constructive comments and suggestions. We also thank the Jigongshan National Nature Reserve for facilitating the field work, especially Mr. Denglong Ha, Mr. Tao Zhang and Ms. Dan Liu for their field assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Tim Moore.
Rights and permissions
About this article
Cite this article
Yang, Q., Xu, M., Chi, Y. et al. Effects of freeze damage on litter production, quality and decomposition in a loblolly pine forest in central China. Plant Soil 374, 449–458 (2014). https://doi.org/10.1007/s11104-013-1882-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-013-1882-y