Abstract
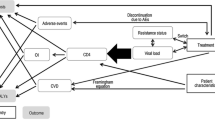
This work explores the advantages of a model based drug development (MBDD) approach for the design and analysis of antiretroviral phase II trials. Two different study settings were investigated: (1) a 5-arm placebo-controlled parallel group dose-finding/proof of concept (POC) study and (2) a comparison of investigational drug and competitor. Studies were simulated using a HIV-1 dynamics model in NONMEM. The Monte-Carlo Mapped Power method determined the sample size required for detecting a dose–response relationship and a significant difference in effect compared to the competitor using a MBDD approach. Stochastic simulation and re-estimation were used for evaluation of model parameter precision and bias given different sample sizes. Results were compared to those from an unpaired, two-sided t test and ANOVA (p ≤ 0.05). In all scenarios, the MBDD approach resulted in smaller study sizes and more precisely estimated treatment effect than conventional statistical analysis. Using a MBDD approach, a sample size of 15 patients could be used to show POC and estimate ED50 with a good precision (relative standard error, 25.7 %). A sample size of 10 patients per arm was needed using the MBDD approach for detecting a difference in treatment effect of ≥20 % at 80 % power, a 3.4-fold reduction in sample size compared to a t test. The MBDD approach can be used to achieve more precise dose–response characterization facilitating decision making and dose selection. If necessitated, the sample size needed to reach a desired power can potentially be reduced compared to traditional statistical analyses. This may allow for comparison against competitors already in early clinical studies.





Similar content being viewed by others
References
Paul SM, Mytelka DS, Dunwiddie CT, Persinger CC, Munos BH, Lindborg SR, Schacht AL (2010) How to improve R&D productivity: the pharmaceutical industry’s grand challenge. Nat Rev Drug Discov 9:203–214. doi:10.1038/nrd3078
Stone JA, Banfield C, Pfister M, Tannenbaum S, Allerheiligen S, Wetherington JD, Krishna R, Grasela DM (2010) Model-based drug development survey finds pharmacometrics impacting decision making in the pharmaceutical industry. J Clin Pharmacol 50:20S–30S. doi:10.1177/0091270010377628
Lalonde RL, Kowalski KG, Hutmacher MM, Ewy W, Nichols DJ, Milligan PA, Corrigan BW, Lockwood PA, Marshall SA, Benincosa LJ, Tensfeldt TG, Parivar K, Amantea M, Glue P, Koide H, Miller R (2007) Model-based drug development. Clin Pharmacol Ther 82:21–32. doi:10.1038/sj.clpt.6100235
Suryawanshi S, Zhang L, Pfister M, Meibohm B (2010) The current role of model-based drug development. Expert Opin Drug Discov 5:311–321. doi:10.1517/17460441003713470
Zhang L, Sinha V, Forgue ST, Callies S, Ni L, Peck R, Allerheiligen SR (2006) Model-based drug development: the road to quantitative pharmacology. J Pharmacokinet Pharmacodyn 33:369–393. doi:10.1007/s10928-006-9010-8
Rosario MC, Jacqmin P, Dorr P, van der Ryst E, Hitchcock C (2005) A pharmacokinetic–pharmacodynamic disease model to predict in vivo antiviral activity of maraviroc. Clin Pharmacol Ther 78:508–519. doi:10.1016/j.clpt.2005.07.010
Rosario MC, Jacqmin P, Dorr P, James I, Jenkins TM, Abel S, van der Ryst E (2008) Population pharmacokinetic/pharmacodynamic analysis of CCR5 receptor occupancy by maraviroc in healthy subjects and HIV-positive patients. Br J Clin Pharmacol 65(Suppl 1):86–94. doi:10.1111/j.1365-2125.2008.03140.x
Rosario MC, Poland B, Sullivan J et al (2006) A pharmacokinetic-pharmacodynamic model to optimize the phase IIa development program of maraviroc. J Acquir Immune Defic Syndr 42:183–191. doi:10.1097/01.qai.0000220021.64115.37
Fang J, Jadhav PR (2012) From in vitro EC50 to in vivo dose-response for antiretrovirals using an HIV disease model. Part I: a framework. J Pharmacokinet Pharmacodyn 39:357–368. doi:10.1007/s10928-012-9255-3
FDA (2002) Guidance for industry guidance for industry: antiretroviral drugs using plasma HIV RNA measurements—clinical considerations for accelerated and traditional approval. U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER), Rockville
MEA (2009) Guideline on the clinical development of medicinal products for the treatment of HIV infection. In: EMEA. http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2009/09/WC500003399.pdf. Accessed 8 Aug 2012
Fätkenheuer G, Pozniak AL, Johnson MA, Plettenberg A, Staszewski S, Hoepelman AI, Saag MS, Goebel FD, Rockstroh JK, Dezube BJ, Jenkins TM, Medhurst C, Sullivan JF, Ridgway C, Abel S, James IT, Youle M, van der Ryst E (2005) Efficacy of short-term monotherapy with maraviroc, a new CCR5 antagonist, in patients infected with HIV-1. Nat Med 11:1170–1172. doi:10.1038/nm1319
Goebel F, Yakovlev A, Pozniak AL, Vinogradova E, Boogaerts G, Hoetelmans R, de Béthune MP, Peeters M, Woodfall B (2006) Short-term antiviral activity of TMC278—a novel NNRTI—in treatment-naive HIV-1-infected subjects. AIDS 20:1721–1726. doi:10.1097/01.aids.0000242818.65215.bd
Fätkenheuer G, Staszewski S, Plettenburg A, Hackman F, Layton G, McFadyen L, Davis J, Jenkins TM (2009) Activity, pharmacokinetics and safety of lersivirine (UK-453,061), a next-generation nonnucleoside reverse transcriptase inhibitor, during 7-day monotherapy in HIV-1-infected patients. AIDS 23:2115–2122
Moyle G, Boffito M, Stoehr A, Rieger A, Shen Z, Manhard K, Sheedy B, Hingorani V, Raney A, Nguyen M, Nguyen T, Ong V, Yeh LT, Quart B (2010) Phase 2a randomized controlled trial of short-term activity, safety, and pharmacokinetics of a novel nonnucleoside reverse transcriptase inhibitor, RDEA806, in HIV-1-positive, antiretroviral-naive subjects. Antimicrob Agents Chemother 54:3170–3178
Min S, Sloan L, DeJesus E, Hawkins T, McCurdy L, Song I, Stroder R, Chen S, Underwood M, Fujiwara T, Piscitelli S, Lalezari J (2011) Antiviral activity, safety, and pharmacokinetics/pharmacodynamics of dolutegravir as 10-day monotherapy in HIV-1-infected adults. AIDS 25:1737–1745
Karlsson KE (2010) Benefits of pharmacometric model-based design and analysis of clinical trials. Acta Universitatis Upsaliensis, Uppsala
Vong C, Bergstrand M, Nyberg J, Karlsson MO (2012) Rapid sample size calculations for a defined likelihood ratio test-based power in mixed-effects models. AAPS J 14:176–186
Chan PL, Jacqmin P, Lavielle M, McFadyen L, Weatherley B (2011) The use of the SAEM algorithm in MONOLIX software for estimation of population pharmacokinetic–pharmacodynamic–viral dynamics parameters of maraviroc in asymptomatic HIV subjects. J Pharmacokinet Pharmacodyn 38:41–61
Röshammar D, Simonsson US, Ekvall H, Flamholc L, Ormaasen V, Vesterbacka J, Wallmark E, Ashton M, Gisslén M (2011) Non-linear mixed effects modeling of antiretroviral drug response after administration of lopinavir, atazanavir and efavirenz containing regimens to treatment-naïve HIV-1 infected patients. J Pharmacokinet Pharmacodyn 38:727–742
Fang J, Jadhav PR (2012) From in vitro EC50 to in vivo dose-response for antiretrovirals using an HIV disease model. Part II: application to drug development. J Pharmacokinet Pharmacodyn 39:369–381
Beal S, Sheiner LB, Boeckmann A, Bauer, R J NONMEM users guides (1989-2009). ICON Development Solutions, Ellicott City MD, USA, 2009
Lindbom L, Ribbing J, Jonsson EN (2004) Perl-speaks-NONMEM (PsN)—a Perl module for NONMEM related programming. Comput Methods Programs Biomed 75:85–94. http://psn.sourceforge.net/
Lindbom L, Pihlgren P, Jonsson EN (2005) PsN-toolkit—a collection of computer intensive statistical methods for non-linear mixed effect modeling using NONMEM. Comput Methods Programs Biomed 79:241–257. http://psn.sourceforge.net/. doi:10.1016/j.cmpb.2005.04.005
Jonsson EN, Karlsson MO (1998) Xpose—an S-PLUS based population pharmacokinetic/pharmacodynamic model building aid for NONMEM. Comput Methods Programs in Biomedicine 58:51–64. http://xpose.sourceforge.net/
Keizer RJ, van Benten M, Beijnen JH, Schellens JH, Huitema AD. (2011) Piraña and PCluster: a modeling environment and cluster infrastructure for NONMEM. Comput Methods Programs Biomed 101:72–79. http://pirana-software.com/
White DB, Walawander CA, Liu DY, Grasela TH (1992) Evaluation of hypothesis testing for comparing two populations using NONMEM analysis. J Pharmacokinet Biopharm 20:295–313
Lee PI (2001) Design and power of a population pharmacokinetic study. Pharm Res 18:75–82
Disclosure
Daniel Röshammar is a current employee of AstraZeneca. However, this work is not funded by AstraZeneca or any other pharmaceutical company.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rekić, D., Röshammar, D. & Simonsson, U.S.H. Model based design and analysis of phase II HIV-1 trials. J Pharmacokinet Pharmacodyn 40, 487–496 (2013). https://doi.org/10.1007/s10928-013-9324-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-013-9324-2