Abstract
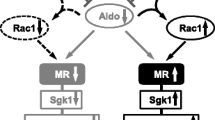
Mineralocorticoid receptors (MR) bind both mineralocorticoids and glucocorticoids with high affinity (deoxycorticosterone = corticosterone ≥ aldosterone = cortisol), and are found in both Na+ transporting epithelia (e.g. kidney, colon) and nonepithelial tissues (e.g. heart, brain). MR evolved before aldosterone synthase, consistent with their acting in nonepithelial tissues as high affinity glucocorticoid receptors, essentially always occupied by normal levels of endogenous glucocorticoids. In epithelial tissues the enzyme 11β hydroxysteroid dehydrogenase Type 2 (11βHSD2) allows aldosterone to selectively activate MR, by converting cortisol to cortisone and NAD to NADH. 11βHSD2 debulks intracellular cortisol by 90%, to levels ∼10-fold those of aldosterone, so that when the enzyme is operating most epithelial MR are occupied but not activated by cortisol. When intracellular redox state is changed—by inhibition of 11β HSD2, generation of reactive oxygen species, or intracellular introduction of oxidised glutathione (GSSG)—cortisol changes from an MR antagonist to an MR agonist. This bivalent activity of cortisol appears to underlie the therapeutic efficacy of MR blockade in heart failure (RALES, EPHESUS) and in essential hypertension, providing a rationale for MR blockade in cardiovascular disease not characterized by elevated aldosterone levels. Its wider (patho)physiologic implications, particularly for neurobiology, remain to be explored.
Similar content being viewed by others
References
Simpson S, Tait J, Wettstein A, Neher R, von Euw J, Reichstein T. Isolierung eines neuen kristallisierten Hormons aus Nebennerien mit besonders hoher Wirksamkeit auf den Mineralsoffwechsel. Experientia 1953;9:333–335.
Rousseau G, Baxter J, Funder J, Edelman I, Tomkins G. Glucocorticoid and mineralocorticoid receptors for aldosterone. J Steroid Biochem 1972;3:219–227.
Funder JW, Feldman D, Edelman IS. Specific aldosterone binding in rat kidney and parotid. J Steroid Biochemistry 1972;3:209–218.
Funder JW, Feldman D, Edelman IS. The roles of plasma binding and receptor specificity in the mineralocorticoid action of aldosterone. Endocrinology 1973;92:992–1004.
Marver D, Stewart J, Funder JW, Feldman D, Edelman IS. Renal aldosterone receptors: Studies with (3H) aldosterone and the anti-mineralocorticoid (3H) spirolactone (SC26304). Proc Natl Acad Sci USA 1974;71:1431–1435.
McEwen BS, De Kloet ER, Rostene W. Adrenal steroid receptors and actions in the nervous system. Physiol Rev 1986;66:1121–1188.
Krozowski ZS, Funder JW. Renal mineralocorticoid receptors and hippocampal corticosterone-binding species have identical intrinsic steroid specificity. Proc Natl Acad Sci USA 1983;80:6056–6060.
Patel PD, Sherman TG, Goldman DJ, Watson SJ. Molecular cloning of a mineralocorticoid (type 1) receptor complementary DNA from rat hippocampus. Mol Endocrinol 1989;3:1877–1885.
Doyle D, Krozowski Z, Morgan FJ, Funder JW. Analysis of renal and hippocampal Type I and Type II receptors by Fast Protein Liquid Chromatography. J Steroid Biochem 1988;29:415–421.
Berecek KH, Barron KW, Webb RL, Brody MJ. Vasopressin-central nervous system interactions in the development of DOCA-hypertension. Hypertension 1982;4:131–137.
Pearce P, Funder JW. High affinity aldosterone binding sites (Type 1 receptors) in rat heart. Clin Exp Pharmacol Physiol 1987;14:859–866.
Funder JW, Pearce PT, Smith R, Campbell J. Vascular type I aldosterone binding sites are physiological mineralocorticoid receptors. Endocrinology 1989;125:2224–2226.
Arriza JL, Weinberger C, Cerelli G, et al. Cloning of human mineralocorticoid receptor complementary DNA: Structural and functional kinship with the glucocorticoid receptor. Science 1987;237:268–275.
Myles K, Funder JW. Progesterone binding to mineralocorticoid receptors: in vitro and in vivo studies. Amer J Physiol 1996;270:E601–E607.
Lifton RP, Dluhy RG, Powers M, et al. A chimaeric 11 beta-hydroxylase/aldosterone synthase gene causes glucocorticoid-remediable aldosteronism and human hypertension. Nature 1992;355:262–265.
Dunlop F, Crock P, Montalto J, Funder JW, Curnow K. A compound heterozygote case of type II aldosterone synthase deficiency. J Clin Endocrinol Metab 2003;88:2518–2526.
Thornton JW. Evolution of vertebrate steroid receptors from an ancestral estrogen receptor by ligand exploitation and serial genome expansions. Proc Natl Acad Sci USA 2001;98:5671–5676.
Greenwood A, Butler P, White R, DeMarco U, Pearce D, Fernald R. Multiple corticosteroid receptors in a teleost fish: Distinct sequences. Endocrinology 2003;144:4226–4236.
Baker ME. Evolution of glucocorticoid and mineralocorticoid responses: Go fish. Endocrinology 2003;144:4223–4225.
Sturm A, Bury N, Dengreville L, et al. 11-deoxycorticosterone is a potent agonist of the rainbow trout (Oncorhynchus mykiss) mineralocorticoid receptor. Endocrinology 2004:in press.
Geller D, Farhi A, Pinkerton N, et al. Activating mineralocorticoid receptor mutation in hypertension exacerbated by pregnancy. Science 2000;289:119–123.
New MI, Levine LS, Biglieri EG, Pareira J, Ulick S. Evidence for an unidentified steroid in a child with apparent mineralocorticoid hypertension. J Clin Endocrinol Metab 1977;44:924–933.
Stewart PM, Wallace AM, Valentino R, Burt D, Shackleton CH, Edwards CR. Mineralocorticoid activity of liquorice: 11-beta-hydroxysteroid dehydrogenase deficiency comes of age. Lancet 1987;2:821–824.
Funder JW, Pearce P, Smith R, Smith AI. Mineralocorticoid action: Target-tissue specificity is enzyme, not receptor, mediated. Science 1988;242:583–585.
Edwards CR, Stewart PM, Burt D, et al. Localisation of 11 beta-hydroxysteroid dehydrogenase—tissue specific protector of the mineralocorticoid receptor. Lancet 1988;2:986–989.
Funder JW. All really great lies are half true. Science 1979;206:1139.
Funder J. Enzymes and receptors: Challenges and future directions. Steroids 1994;59:164–169.
Funder JW, Myles K. Exclusion of corticosterone from epithelial mineralocorticoid receptors is insufficient for selectivity of aldosterone action: In vivo binding studies. Endocrinology 1996;137:5264–5268.
Alzamora R, Michea L, Marusic ET. Role of 11beta-hydroxysteroid dehydrogenase on nongenomic aldosterone effects in human arteries. Hypertension 2000;35:1099–1104.
Zhang Q, Piston D, Goodman R. Regulation of corepressor function by nuclear NADH. Science 2002;295:1895–1897.
Fjeld C, Birdsong W, Goodman R. Differential binding of NAD+ and NADH allows the transcriptional corepressor carboxyl-terminal binding protein to serve as a metabolic sensor. Proc Natl Acad Sci USA 2003;100:9202–9207.
Ward MR, Kanellakis P, Ramsey D, Funder JW, Bobik A. Eplerenone suppresses constrictive remodeling and collagen accumulation after angioplasty in porcine coronary arteries. Circulation 2001;104:467–472.
Brilla CG, Weber KT. Mineralocorticoid excess, dietary sodium, and myocardial fibrosis. J Lab Clin Med 1992;120:893–901.
Young M, Fullerton M, Dilley R, Funder J. Mineralocorticoids, hypertension, and cardiac fibrosis. J Clin Invest 1994;93:2578–2583.
Rocha R, Martin-Berger C, Yang P, Scherrar R, Delyani J, McMahon E. Selective aldosterone blockade prevents angiotensin II/salt-induced vascular inflammation in the rat heart. Endocrinology 2002;143:4828–4836.
Rocha R, Rudolph A, Frierdich G, et al. Aldosterone induces a vascular inflammatory phenotype in the rat heart. Amer J Physiol 2002;283:H1802–H1810.
Blasi ER, Rocha R, Rudolph AE, Blomme EA, Polly ML, McMahon EG. Aldosterone/salt induces renal inflammation and fibrosis in hypertensive rats. Kidney International 2003;63:1791–1800.
Mihailidou AS, Mardini M, Fraser T, Knights D, Funder JW. Agonist/antagonist activity of cortisol in cardiomyocyte mineralocorticoid receptors is determined by redox state. 30th Annual Meeting of the International Aldosterone Conference, New Orleans, 2004.
Sato A, Funder JW. High glucose stimulates aldosterone-induced hypertrophy via Type I mineralocorticoid receptors in neonatal rat cardiomyocytes. Endocrinology 1996;137:4145–4153.
Selye H. The general adaptation syndrome and the disease of adaptation. J Clin Endocrinol 1946;6:117–230.
Young M, Moussa L, Dilley R, Funder JW. Early inflammatory responses in experimental cardiac hypertrophy and fibrosis: Effects of 11 beta-hydroxysteroid dehydrogenase inactivation. Endocrinology 2003;144:1121–1125.
Young MJ, Funder JW. Eplerenone, but not steroid withdrawal, reverses cardiac fibrosis in DOC/salt rats. Endocrinology 2004;145:3153–3157.
Qin W, Rudolph A, Bond B, et al. A transgenic model of aldosterone-driven cardiac hypertrophy and heart failure. Circ Res 2003;93:69–76.
Qin W, Bond BR, Donoho GP, et al. Mineralocorticoid receptor mediates heart failure in the 11beta-hydroxysteroid dehydrogenase type 2 transgenic mouse model. American Heart Association Scientific Sessions, New Orleans, 2004.
Pitt B, Reichek N, Willenbrock R, et al. Effects of eplerenone, enalapril, and eplerenone/enalapril in patients with essential hypertension and left ventricular hypertrophy: The 4E-left ventricular hypertrophy study. Circulation 2003;108:1831–1838.
Epstein M, Buckalew V, Martinez F, et al. Antiproteinuric efficacy of eplerenone, enalapril and eplerenone/enalapril combination in diabetic hypertensives with microalbuminuria. Am J Hypertens 2002;15:24A (abstract).
Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. New Eng J Med 1999;341:709–717.
Pitt B, Remme W, Zannad F, et al. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. New Engl J Med 2003;348:1309–1321.
Levy D, Rocha R, Funder JW. Distinguishing the antihypertensive and electrolyte effects of eplerenone. J Clin Endocrinol Metab 2004;89:2736–2740.
Gomez-Sanchez EP, Fort CM, Gomez-Sanchez CE. Intracerebroventricular infusion of RU28318 blocks aldosterone-salt hypertension. Am J Physiol 1990;258:E482– E484.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Funder, J.W. Mineralocorticoid Receptors: Distribution and Activation. Heart Fail Rev 10, 15–22 (2005). https://doi.org/10.1007/s10741-005-2344-2
Issue Date:
DOI: https://doi.org/10.1007/s10741-005-2344-2