Abstract
Background
The ratio of Helicobacter pylori/NSAID-negative gastric ulcers is increasing. Idiopathic gastric ulcers have unique clinical and endoscopic features, and are associated with more bleeding complications and a higher mortality. Alterations in gastric mucin expression and sialylation pattern may be important in ulcer pathogenesis.
Aims
The purpose of this study was to determine the expression pattern of membrane-bound mucins and side chain sugars in H. pylori associated-, NSAID-, and idiopathic-gastric ulcers.
Methods
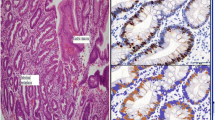
We randomly selected 92 patients with H. pylori (group 1, n = 30), NSAID (group 2, n = 18), combined H. pylori and NSAID associated gastric ulcers (group 3, n = 24), and patients with idiopathic gastric ulcers (group 4, n = 20). Immunohistochemistry for T-cell CD4/CD8, MUC1, MUC4, MUC17, and ECA and SNA lectins staining was performed on sections from the ulcer margins. Inflammation score was assessed according to the Sydney system.
Results
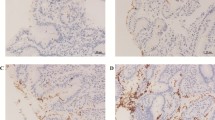
Bleeding and mortality rates were significantly higher in group 4. CD4 positive T cell count was higher in H. pylori positive patients (P = 0.009). Staining intensity of MUC17 was higher in group 1 than in group 4, foveola and glands alike, with 11.50 ± 3.47 versus 6.80 ± 4.02, and 9.61 ± 4.26 versus 7.59 ± 3.26, respectively (P < 0.0001). This was a mirror image with MUC1. SNA lectin staining was increased in group 4, in parallel to MUC1 expression, indicating more abundant α2-6 sialylation in that group.
Conclusions
Cytoplasmic MUC17 staining was significantly decreased in the cases with idiopathic ulcer. The opposite was demonstrated for MUC1. This observation might be important, since different mucins with altered sialylation patterns likely differ in their protection efficiency against acid and pepsin.

Similar content being viewed by others
References
Hyvarinen H, Salmenkyla S, Sipponen P. Helicobacter pylori-negative duodenal and pyloric ulcer: role of NSAIDs. Digestion. 1996;57:305–309.
Gisbert J, Blanco M, Mateos JM, et al. H. pylori -negative duodenal ulcer prevalence and causes in 774 patients. Dig Dis Sci. 1999;44:2295–2302.
Tsuji H, Kohli Y, Fukumitsu S, et al. Helicobacter pylori-negative gastric and duodenal ulcers. J Gastroenterol. 1999;34:455–460.
Elitsur Y, Lawrence Z. Non-Helicobacter pylori related duodenal ulcer disease in children. Helicobacter. 2001;6:239–243.
McColl KEL, E-Nujumi AM, Chittajallu RS, et al. A study of the pathogenesis of Helicobacter pylori negative chronic duodenal ulceration. Gut. 1993;34:762–768.
Jyotheeswaran S, Shah AN, Jin HO, et al. Prevalence of Helicobacter pylori in peptic ulcer patients in greater Rochester, NY: is empirical triple therapy justified? Am J Gastroenterol. 1998;93:574–578.
Niv YH. pylori/NSAID—negative peptic ulcer—the mucin theory. Med Hypotheses. 2010;75:433–435.
Ho SB, Takamura K, Anway R, et al. The adherent gastric mucous layer is composed of alternating layers of MUC5AC and MUC6 mucin proteins. Dig Dis Sci. 2004;49:1598–1606.
Tanaka S, Mizuno M, Maga T, et al. H. pylori decreases gastric mucin synthesis via inhibition of galactosyltransferase. Hepatogastroenterol. 2003;50:1739–1742.
Kawakubo M, Ito Y, Okimura Y, et al. Natural antibiotic function of a human gastric mucin against Helicobacter pylori infection. Science. 2004;305:1003–1006.
Niv Y, Boltin D, Vilkin A, et al. MUC5AC and MUC6 expression remains unchanged in H. pylori/NSAID—negative gastric ulcer (idiopathic ulcer). Gastroenterology. 2011;140:S732.
Luu Y, Junker W, Rachagani S, et al. Human intestinal MUC17 mucin augments intestinal cell restitution and enhances healing of experimental colitis. Int J Biochem Cell Biol. 2010;42:996–1006.
Shibuya N, Goldstein IJ, Broekaert WF, et al. The elderberry (Sambucus nigra L.) bark lectin recognizes the Neu5Ac(alpha 2-6)Gal/GalNAc sequence. J Biol Chem. 1987;262:1596–1601.
De Boeck H, Loontiens FG, Lis H, et al. Binding of simple carbohydrates and some N-acetyllactosamine-containing oligosaccharides to Erythrina cristagalli agglutinin as followed with a fluorescent indicator ligand. Arch Biochem Biophys. 1984;234:297–304.
Tsukashita S, Kushima R, Bamba M, et al. MUC gene expression and histogenesis of adenocarcinoma of the stomach. Int J Cancer. 2001;94:166–170.
Dixon MF, Genta RM, Yardley JH, et al. Classification and grading of gastritis. The updated Sydney System. International workshop on the histopathology of gastritis, Houston 1994. Am J Surg Pathol. 1996;20:1161–1181.
Price AB. The Sydney system: histological division. J Gastroenterol Hepatol. 1991;6:209–222.
Chan HL, Wu JC, Chan FK, et al. Is non-Helicobacter pylori, non-NSAID peptic ulcer a common cause of upper GI bleeding? A prospective study of 977 patients. Gastrointest Endosc. 2001;53:438–442.
Xia HH, Wong BC, Wong KW, et al. Clinical and endoscopic characteristics of non-Helicobacter pylori, non-NSAID duodenal ulcers: a long-term prospective study. Aliment Pharmacol Ther. 2001;15:1875–1882.
Wong GL, Wong VW, Chan Y, et al. High incidence of mortality and recurrent bleeding in patients with Helicobacter pylori-negative idiopathic bleeding ulcers. Gastroenterology. 2009;137:525–531.
Gisbert JP, Calvet X. Review article: Helicobacter pylori-negative duodenal ulcer disease. Aliment Pharmacol Ther. 2009;30:791–815.
McColl KE. How I manage H. pylori-negative, NSAID/aspirin-negative peptic ulcers. Am J Gastroenterol. 2009;104:190–193.
Niv Y. MUC1 and colorectal cancer pathophysiology considerations. World J Gastro. 2008;14:2139–2141.
Celli JP, Turner BS, Afdhal NH, et al. Helicobacter pylori moves through mucus by reducing mucin viscoelasticity. Proc Natl Acad Sci USA. 2009;106:14321–14326.
Kobayashi M, Lee H, Nakayama J, et al. Roles of gastric mucin-type O-glycans in the pathogenesis of Helicobacter pylori infection. Glycobiology. 2009;19:453–461.
Lindén S, Semino-Mora C, Liu H, et al. Role of mucin Lewis status in resistance to Helicobacter pylori infection in pediatric patients. Helicobacter. 2010;15:251–258.
Stromberg E, Lundgren A, Edebo A, et al. Increased frequency of activated T-cells in the Helicobacter pylori-infected antrum and duodenum. FEMS Immunol Med Microbiol. 2003;36:159–168.
Acknowledgments
The work was supported by National Institute of Neurological Disorders and Stroke (grant P30 NS047101; Neurosciences Microscopy Shared Facility, UCSD) and grant *** CSD018 from the G. Harold and Leila Y. Mathers Charitable Foundation (P. Gagneux). Sukhwinder Kaur, Srustidhar Das, and Poonam Sharma performed the MUC4 staining.
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article has been retracted due to plagiarism of content published in "Gastric mucin expression in Helicobacter pylori-related, nonsteroidal anti-inflammatory drug-related and idiopathic ulcers" by Doron Boltin, Marisa Halpern, Zohar Levi, Alex Vilkin, Sara Morgenstern, Samuel B Ho, Yaron Niv, published online in the World Journal of Gastroenterology on September 7, 2012, DOI: http://dx.doi.org/10.3748/wjg.v18.i33.4597.
About this article
Cite this article
Niv, Y., Boltin, D., Halpern, M. et al. RETRACTED ARTICLE: Membrane-Bound Mucins and Mucin Terminal Glycans Expression in Idiopathic or Helicobacter pylori, NSAID Associated Peptic Ulcers. Dig Dis Sci 57, 2535–2544 (2012). https://doi.org/10.1007/s10620-012-2205-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-012-2205-5