Abstract
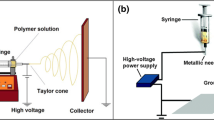
The fabrication of nonwoven mats containing chitosan and bacterial nanocellulose by electrospinning were considered using two different approaches: (1) simultaneous spinning of chitosan and bacterial nanocellulose solutions using two separate syringes towards the same target and (2) coaxial electrospinning, where chitosan and bacterial nanocellulose were simultaneously electrospun through a spinneret composed of two concentric needles to produce core–shell structures. Co-spinning agents were required in both approaches. A direct blend of chitosan and bacterial nanocellulose and subsequent electrospinning was not feasible due to the incompatibility of their respective solvents. The first approach led to the production of mats containing both chitosan and bacterial nanocellulose nanofibers. However, few bacterial nanocellulose fibers were deposited on the collector. Addition of polylactide as a co-spinning agent and an increase in solution temperature (from 22 to 60 °C) during electrospinning was required to improve both fiber formation and collection. On the other hand, coaxial electrospinning showed the best results for the production of nanofibers containing both chitosan and bacterial nanocellulose. Nanofibers with a good yield were obtained by using a chitosan/poly(ethylene oxide) (2.4/0.6 wt/v%) aqueous solution as the inner layer, and a bacterial nanocellulose solution (0.6 wt/v%) as the outer layer. Co-electrospun nanofibers had a diameter of 85 nm in average, and a narrow size distribution. The core/shell nanostructure was validated by transmission electron microscopy whilst energy-dispersive X-ray spectroscopy analysis showed that the nanofibers contained both chitosan and bacterial nanocellulose along their structure. Finally, the mats obtained by the coaxial approach exhibited strong antimicrobial activity with a decrease of 99.9 % of an Escherichia coli population.











Similar content being viewed by others
References
Ahn Y, Lee SH, Kim HJ, Yang YH, Hong JH, Kim YH, Kim H (2012) Electrospinning of lignocellulosic biomass using ionic liquid. Carbohydr Polym 88:395–398
Basmaji P, de Olyveira G, Manzine Costa L, Francozo G, da Costa OJ (2015) Nanoderm extracellular matrix for reconstructive surgery applications. Peertechz J Biomed Eng 1:021–024
Bhattarai N, Edmondson D, Veiseh O, Matsen FA, Zhang M (2005) Electrospun chitosan-based nanofibers and their cellular compatibility. Biomaterials 26:6176–6184
Cao Z, Xiaogang L, Hao Z, Zhen F, Zhi S, Ning C, Yanan X, Faquan Y (2016) A facile and green strategy for the preparation of porous chitosan-coated cellulose composite membranes for potential applications as wound dressing. Cellulose 23:1349–1361
Costa LMM, de Olyveira GM, Basmaji P, Xavier Filho L (2012) Nanopores structure in electrospun bacterial cellulose. J Biomater Nanobiotechnol 3:92
Desai K, Kit K, Li J, Zivanovic S (2008) Morphological and surface properties of electrospun chitosan nanofibers. Biomacromolecules 9:1000–1006
Dror Y et al (2007) One-step production of polymeric microtubes by co-electrospinning. Small 3:1064–1073
El-Kafrawy A (1982) Investigation of the cellulose/LiCl/dimethylacetamide and cellulose/LiCl/N-methyl-2-pyrrolidinone solutions by 13C NMR spectroscopy. J Appl Polym Sci 27:2435–2443
Frenot A, Henriksson MW, Walkenström P (2007) Electrospinning of cellulose-based nanofibers. J Appl Polym Sci 103:1473–1482
Gama M, Gatenholm P, Klemm D (2012) Bacterial nanocellulose: a sophisticated multifunctional material. CRC Press, London
Geng X, Kwon O-H, Jang J (2005) Electrospinning of chitosan dissolved in concentrated acetic acid solution. Biomaterials 26:5427–5432
Goosen MF (1997) Applications of chitin and chitosan. CRC Press, Florida
Greiner A, Wendorff JH (2007) Electrospinning: a fascinating method for the preparation of ultrathin fibers. Angew Chem Int Ed 46:5670–5703
Homayoni H, Ravandi SAH, Valizadeh M (2009) Electrospinning of chitosan nanofibers: processing optimization. Carbohydr Polym 77:656–661
Horii F (2000) Structure of cellulose: recent developments in its characterization. Wood and cellulosic chemistry, 2nd edn. Taylor & Francis, London, pp 83–108
Ignatova M, Manolova N, Markova N, Rashkov I (2009) Electrospun non-woven nanofibrous hybrid mats based on chitosan and PLA for wound-dressing applications. Macromol Biosci 9:102–111
Kim CW, Frey MW, Marquez M, Joo YL (2005) Preparation of submicron-scale, electrospun cellulose fibers via direct dissolution. J Polym Sci B Polym Phys 43:1673–1683
Kim C-W, Kim D-S, Kang S-Y, Marquez M, Joo YL (2006) Structural studies of electrospun cellulose nanofibers. Polymer 47:5097–5107
Kong M, Chen XG, Xing K, Park HJ (2010) Antimicrobial properties of chitosan and mode of action: a state of the art review. Int J Food Microbiol 144:51–63
Kulpinski P (2005) Cellulose nanofibers prepared by the N-methylmorpholine-N-oxide method. J Appl Polym Sci 98:1855–1859
Li L, Hsieh Y-L (2006) Chitosan bicomponent nanofibers and nanoporous fibers. Carbohydr Res 341:374–381
Lin W-C, Lien C-C, Yeh H-J, Yu C-M, S-h H (2013) Bacterial cellulose and bacterial cellulose–chitosan membranes for wound dressing applications. Carbohydr Polym 94:603–611
Liu H, Hsieh YL (2002) Ultrafine fibrous cellulose membranes from electrospinning of cellulose acetate. J Polym Sci B Polym Phys 40:2119–2129
Liu H, Tang C (2007) Electrospinning of cellulose acetate in solvent mixture N,N-dimethylacetamide (DMAc)/acetone. Polym J 39:65–72
Matsumoto T, Tatsumi D, Tamai N, Takaki T (2001) Solution properties of celluloses from different biological origins in LiCl·DMAc. Cellulose 8:275–282
McCormick CL, Callais PA, Hutchinson BH Jr (1985) Solution studies of cellulose in lithium chloride and N, N-dimethylacetamide. Macromolecules 18:2394–2401
Mohite BV, Patil SV (2014) A novel biomaterial: bacterial cellulose and its new era applications. Biotechnol Appl Biochem 61:101–110
Muzzarelli RA (1973) Natural chelating polymers; alginic acid, chitin and chitosan. Pergamon Press, Oxford
Ohkawa K, Cha D, Kim H, Nishida A, Yamamoto H (2004) Electrospinning of chitosan. Macromol Rapid Commun 25:1600–1605
Ohkawa K, Minato K-I, Kumagai G, Hayashi S, Yamamoto H (2006) Chitosan nanofiber. Biomacromolecules 7:3291–3294
Okushita K, Chikayama E, Kikuchi J (2012) Solubilization mechanism and characterization of the structural change of bacterial cellulose in regenerated states through ionic liquid treatment. Biomacromolecules 13:1323–1330
Ostadhossein F, Mahmoudi N, Morales-Cid G, Tamjid E, Navas-Martos FJ, Soriano-Cuadrado B, López JM, Simchi A (2015) Development of chitosan/bacterial cellulose composite films containing nanodiamonds as a potential flexible platform for wound dressing. Materials 8:6401–6418
Pakravan M, Heuzey M-C, Ajji A (2011) A fundamental study of chitosan/PEO electrospinning. Polymer 52:4813–4824
Pakravan M, Heuzey M-C, Ajji A (2012) Core–shell structured PEO–chitosan nanofibers by coaxial electrospinning. Biomacromolecules 13:412–421
Park TJ, Yeon JJ, Sung-Wook C, Hongkwan P, Hyungsup K, Eunkyoung K, Sang HL, Jung HK (2011) Native chitosan/cellulose composite fibers from an ionic liquid via electrospinning. Macromol Res 19:213–215
Phisalaphong M, Jatupaiboon N (2008) Biosynthesis and characterization of bacteria cellulose–chitosan film. Carbohydr Polym 74:482–488
Potthast A, Rosenau T, Sixta H, Kosma P (2002) Degradation of cellulosic materials by heating in DMAc/LiCl. Tetrahedron Lett 43:7757–7759
Röder T, Morgenstern B, Schelosky N, Glatter O (2001) Solutions of cellulose in N,N-dimethylacetamide/lithium chloride studied by light scattering methods. Polymer 42:6765–6773
Schiffman JD, Schauer CL (2007) Cross-linking chitosan nanofibers. Biomacromolecules 8:594–601
Schlufter K, Schmauder HP, Dorn S, Heinze T (2006) Efficient homogeneous chemical modification of bacterial cellulose in the ionic liquid 1-N-butyl-3-methylimidazolium chloride. Macromol Rapid Commun 27:1670–1676
Sekwon K (2010) Chitin, chitosan, oligosaccharides and their derivatives: biological activities and applications. CRC Press, London
Shanshan G, Jianqing W, Zhengwei J (2012) Preparation of cellulose films from solution of bacterial cellulose in NMMO. Carbohydr Polym 87:1020–1025
Siró I, Plackett D (2010) Microfibrillated cellulose and new nanocomposite materials: a review. Cellulose 17:459–494
Subramanian A, Vu D, Larsen GF, Lin H-Y (2005) Preparation and evaluation of the electrospun chitosan/PEO fibers for potential applications in cartilage tissue engineering. J Biomater Sci Polymer Ed 16:861–873
Swatloski RP, Spear SK, Holbrey JD, Rogers RD (2002) Dissolution of cellose with ionic liquids. J Am Chem Soc 124:4974–4975
Xu S, Zhang J, He A, Li J, Zhang H, Han CC (2008) Electrospinning of native cellulose from nonvolatile solvent system. Polymer 49:2911–2917
Yarin A (2011) Coaxial electrospinning and emulsion electrospinning of core–shell fibers. Polymer Adv Tech 22:310–317
Yu JH, Fridrikh SV, Rutledge GC (2004) Production of submicrometer diameter fibers by two-fluid electrospinning. Adv Mater 16:1562–1566
Acknowledgments
The authors thank the Fonds de Recherche du Québec-Nature et Technologies (FRQNT) and the Natural Sciences and Engineering Research Council of Canada (NSERC) for their financial support of this work. The authors would like also to acknowledge Prof. France Daigle from the Department of Microbiology, Infectiology and Immunology, Université de Montréal for her training and support in the antibacterial tests. Finally, the authors thank the reviewers of Cellulose for their valuable comments to improve this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ardila, N., Medina, N., Arkoun, M. et al. Chitosan–bacterial nanocellulose nanofibrous structures for potential wound dressing applications. Cellulose 23, 3089–3104 (2016). https://doi.org/10.1007/s10570-016-1022-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-016-1022-y