Abstract
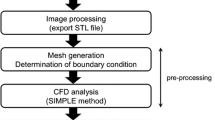
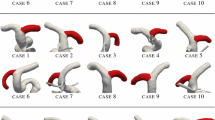
Computational hemodynamic simulations of cerebral aneurysms have traditionally relied on stereotypical boundary conditions (such as blood flow velocity and blood pressure) derived from published values as patient-specific measurements are unavailable or difficult to collect. However, controversy persists over the necessity of incorporating such patient-specific conditions into computational analyses. We perform simulations using both endovascularly-derived patient-specific and typical literature-derived inflow and outflow boundary conditions. Detailed three-dimensional anatomical models of the cerebral vasculature are developed from rotational angiography data, and blood flow velocity and pressure are measured in situ by a dual-sensor pressure and velocity endovascular guidewire at multiple peri-aneurysmal locations in 10 unruptured cerebral aneurysms. These measurements are used to define inflow and outflow boundary conditions for computational hemodynamic models of the aneurysms. The additional in situ measurements which are not prescribed in the simulation are then used to assess the accuracy of the simulated flow velocity and pressure drop. Simulated velocities using patient-specific boundary conditions show good agreement with the guidewire measurements at measurement locations inside the domain, with no bias in the agreement and a random scatter of ≈25%. Simulated velocities using the simplified, literature-derived values show a systematic bias and over-predicted velocity by ≈30% with a random scatter of ≈40%. Computational hemodynamics using endovascularly measured patient-specific boundary conditions have the potential to improve treatment predictions as they provide more accurate and precise results of the aneurysmal hemodynamics than those based on commonly accepted reference values for boundary conditions.






Similar content being viewed by others
References
Ackerstaff, R. G., M. J. Suttorp, J. C. van den Berg, T. T. Overtoom, J. A. Vos, E. T. Bal, and P. Zanen. Prediction of early cerebral outcome by transcranial Doppler monitoring in carotid bifurcation angioplasty and stenting. J. Vasc. Surg. 41(4):618–624, 2005.
Augst, A. D., D. C. Barratt, A. D. Hughes, F. P. Glor, S. A. Thom, and X. Y. Xu. Accuracy and reproducibility of CFD predicted wall shear stress using 3D ultrasound images. J. Biomech. Eng. 125:218–222, 2003.
Boussel, L., V. Rayz, C. McCulloch, A. Martin, G. Acevedo-Bolton, et al. Aneurysm growth occurs at region of low wall shear stress: patient-specific correlation of hemodynamics and growth in a longitudinal study. Stroke 39:2997–3002, 2008.
Cebral, J., M. Castro, C. Putman, and N. Alperin. Flow–area relationship in internal carotid and vertebral arteries. Physiol. Meas. 29(5):585–594, 2008.
Cebral, J. R., M. A. Castro, S. Appanaboyina, C. M. Putman, D. Millan, and A. F. Frangi. Efficient pipeline for image-based patient-specific analysis of cerebral aneurysm hemodynamics: technique and sensitivity. IEEE Trans. Med. Imaging 24(4):457–467, 2005.
Cebral, J. R., M. A. Castro, J. E. Burgess, R. S. Pergolizzi, M. J. Sheridan, and C. M. Putman. Characterization of cerebral aneurysms for assessing risk of rupture by using patient-specific computational hemodynamic models. Am. J. Neuroradiol. 26:2550–2559, 2005.
Cebral, J. R., F. Mut, M. Raschi, E. Scrivano, R. Ceratto, et al. Aneurysm rupture following treatment with flow-diverting stents: computational hemodynamics analysis of treatment. Am. J. Neuroradiol. 32:27–33, 2011.
Cebral, J. R., F. Mut, J. Weir, and C. M. Putman. Association of hemodynamic characteristics and cerebral aneurysm rupture. Am. J. Neuroradiol. 32:264–270, 2011.
Chaloupka, J. C., F. Viñuela, C. Kimme-Smith, J. Robert, and G. R. Duckwiler. Use of a Doppler guide wire for intravascular blood flow measurements: a validation study for potential neurologic endovascular applications. Am. J. Neuroradiol. 15:509–517, 1994.
Cheng, C., F. Helderman, D. Tempel, D. Segers, B. Hierck, et al. Large variations in absolute wall shear stress levels within one species and between species. Atherosclerosis 195(2):225–235, 2007.
Ford, M. D., N. Alperin, S. H. Lee, D. W. Holdsworth, and D. A. Steinman. Characterization of volumetric flow rate waveforms in the normal internal carotid and vertebral arteries. Physiol. Meas. 26(4):477–488, 2005.
Geers, A., I. Larrabide, A. G. Radaelli, H. Bogunovic, M. Kim, et al. (2011) Patient-specific computational hemodynamics of intracranial aneurysms from 3D rotational angiography and CT angiography: an in vivo reproducibility study. Am. J. Neuroradiol. 32(3):581–586, 2011.
Karmonik, C., C. Yen, O. Diaz, R. Klucznik, R. G. Grossman, and G. Benndorf. Temporal variations of wall shear stress parameters in intracranial aneurysms: importance of patient-specific inflow waveforms for CFD calculations. Acta Neurochir. 152:1391–1398, 2010.
Levitt, M. R., P. M. McGah, A. Aliseda, P. Mourad, J. D. Nerva, et al. Cerebral aneurysm treated with flow-diverting stents: computational models using intravascular blood flow measurements. Am. J. Neuroradiol., 2013. DOI: 10.3174/ajnr.A3624
Levitt, M. R., S. S. Vaidya, J. C. Mai, D. K. Hallam, L. J. Kim, and B. V. Ghodke. Balloon test occlusion with the Doppler velocity guidewire. J. Stroke Cerebrovasc Dis. 21:901–904, 2012.
Marzo, A., P. Singh, I. Larrabide, A. Radaelli, S. Coley, et al. Computational hemodynamics in cerebral aneurysms: the effects of modeled versus measured boundary conditions. Ann. Biomed. Eng. 39(2):884–896, 2011.
Melamed, E., S. Lavy, S. Bentin, G. Cooper, and Y. Rinot. Reduction in regional cerebral blood flow during normal aging in man. Stroke 11(1):31–35, 1980.
Meng, H., V. M. Tutino, J. Xiang, and A. Siddiqui. High WSS or low WSS? Complex interactions of hemodynamics with intracranial aneurysm initiation, growth, and rupture: toward a unifying hypothsis. Am. J. Neuroradiol., 2014. DOI: 10.3174/ajnr.A3558.
Miura, Y., F. Ishida, Y. Umeda, H. Tanemura, H. Suzuki, et al. Shear stress is independently associated with the rupture status of middle cerebral artery aneurysms. Stroke 44:519–521, 2013.
Mynard, J. P., and D. A. Steinman. Effect of velocity profile skewing on blood velocity and volume flow waveforms derived from maximum Doppler spectral velocity. Ultrasound Med. Biol. 39(5):870–881, 2013.
Radaelli, A. G., L. Augsburger, J. R. Cebral, M. Ohta, D. A. Rüfenacht, et al. Reproducibility of haemodynamical simulations in a subject-specific stented aneurysm model: a report on the Virtual Intracranial Stenting Challenge 2007. J. Biomech. 41:2069–2081, 2008.
Reymond, P., Y. Bohraus, F. Perren, F. Lazeyras, and N. Stergiopulos. Validation of a patient-specific one-dimensional model of the systemic arterial tree. Am. J. Physiol. Heart Circ. Physiol. 301(3):H1173–H1182, 2011.
Schneiders, J. J., S. P. Ferns, P. van Ooij, M. Siebes, A. J. Nederveen, et al. Comparison of phase-contrast MR imaging and endovascular sonography for intracranial blood flow velocity measurements. Am. J. Neuroradiol. 33:1786–1790, 2012.
Seitz, J., M. Strotzer, T. Wild, W. R. Nitz, M. Völk, M. Lenhart, and S. Feuerbach. Quantification of blood flow in the carotid arteries: comparison of doppler ultrasound and three different phase-contrast magnetic resonance imaging sequences. Invest. Radiol. 36(11):642–647, 2001.
Steinman, D. A. Computational modeling and flow diverters: a teaching moment. Am. J. Neuroradiol. 32:981–983, 2011.
Steinman, D. A., Y. Hoi, P. Fahy, L. Morris, M. T. Walsh, et al. Variability of computational fluid dynamics solutions for pressure and flow in a giant aneurysm: the ASME 2012 Summer Bioengineering Conference CFD Challenge. J. Biomech. Eng. 135:1–13, 2013.
Steinman, D. A., J. S. Milner, C. J. Norley, S. P. Lownia, and D. W. Holdsworth. Image-based computational simulation of flow dynamics in a giant intracranial aneurysm. Am. J. Neuroradiol. 24:559–566, 2003.
Sun, Q., A. Groth, and T. Aach. Comprehensive validation of computational fluid dynamics simulations of in-vivo blood flow in patient-specific cerebral aneurysms. Med. Phys. 39(2):742–754, 2012.
Sviri, G. E., B. Ghodke, G. W. Britz, C. M. Douville, D. R. Haynor, and A. H. Mesiwala. Transcranial Doppler grading criteria for basilar artery vasospasm. Neurosurgery 59:360–366, 2006.
Thomas, J. B., J. S. Milner, B. K. Rutt, and D. A. Steinman. Reproducibility of image-based computational fluid dynamics models of the human carotid bifurcation. Ann. Biomed. Eng. 31:132–141, 2003.
Turner, C. L., J. N. Higgins, and P. J. Kirkpatrick. Assessment of transcranial color-coded duplex sonography for the surveillance of intracranial aneurysms treated with Guglielmi detachable coils. Neurosurgery 53:866–871, 2003.
Venugopal, P., D. Valentino, H. Schmitt, J. P. Villablanca, F. Viñuela, et al. Sensitivity of patient-specific numerical simulation of cerebral aneurysm hemodynamics to inflow boundary conditions. J. Neurosurg. 106:1051–1060, 2007.
Xiang, J., S. K. Natarajan, M. Tremmel, D. Ma, J. Mocco, et al. Hemodynamic-morphologic discriminants for intracranial aneurysm rupture. Stroke 42:144–152, 2011.
Acknowledgments
This work was supported by NINDS/NIH grant (1R03NS078539), an NSF CAREER Award (CBET-0748133), a Washington Royalty Research Fund grant, an unrestricted grant to our academic institution from Volcano Corporation, San Diego CA and the generous support of Mark Robison and family.
Conflict of interest
This study was supported by the manufacturer of the dual-sensor Doppler guidewire (Volcano Corporation) through an unrestricted educational grant made to the Departments of Neurological Surgery and Radiology, University of Washington. The sponsor was shown the final manuscript but had no role in the study design, data collection, data analyses, or interpretation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Ender A Finol oversaw the review of this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
McGah, P.M., Levitt, M.R., Barbour, M.C. et al. Accuracy of Computational Cerebral Aneurysm Hemodynamics Using Patient-Specific Endovascular Measurements. Ann Biomed Eng 42, 503–514 (2014). https://doi.org/10.1007/s10439-013-0930-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-013-0930-3