Abstract
Background
Administration of the human papillomavirus (HPV) vaccine decreased dramatically in Japan after extensive news of adverse vaccine events and suspension of the governmental recommendation for the vaccine. In this study, we investigated the knowledge and acceptance of vaccinated adolescents concerning cervical cancer, cancer screening and the HPV vaccine. Furthermore, we analyzed whether and by how much the news affected acceptance of the vaccination.
Methods
This study was conducted as a part of Osaka Clinical resEArch of HPV vacciNe (OCEAN) study. A questionnaire was distributed to 2,777 study registrants.
Results
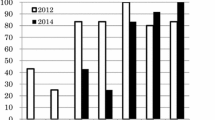
The response rate was 38 %. The recognition rate of the news of the vaccine’s adverse events was 80 %; it was 68 % for awareness of the government’s announcement of the suspension of its recommendation for the vaccine. Among those who had a chance to hear or see the negative news during their vaccination period, 46 (60 %) continued vaccination while knowing of the news, 22 (29 %) discontinued vaccination, and 9 (11 %) continued vaccination without an awareness of the news. Reports of the vaccine’s adverse events were the main reason for not continuing the vaccination series. Those who consulted doctors after hearing the adverse news were significantly more likely to continue their vaccinations than those who did not.
Conclusions
Our results should help in understanding the need for a strong promotion of vaccine usage and cancer screening after future retraction of the recommendation suspension. This may apply to other countries with an unsatisfactory rate of HPV vaccination due to fears of adverse vaccine events.
Similar content being viewed by others
References
Jemal A, Bray F, Center MM et al (2011) Global cancer statistics. CA Cancer J Clin 61:69–90
Japanese Ministry of Health, Labour and Welfare. Vital Statistics in Japan, tabulated by Center for Cancer Control and Information Services, National Cancer Center, Japan May 12, 2012. Available via DIALOG. http://ganjoho.jp/pro/statistics/en/table_download.html Accessed April 2014
Munoz N, Bosch FX, Castellsague X et al (2004) Against which human papillomavirus types shall we vaccinate and screen? The international perspective. Int J Cancer 111:278–285
Smith JS, Lindsay L, Hoots B et al (2007) Human papillomavirus type distribution in invasive cervical cancer and high-grade cervical lesions: a meta-analysis update. Int J Cancer 121:621–632
Li N, Franceschi S, Howell-Jones R et al (2011) Human papillomavirus type distribution in 30,848 invasive cervical cancers worldwide: variation by geographical region, histological type and year of publication. Int J Cancer 128:927–935
Miura S, Matsumoto K, Oki A et al (2006) Do we need a different strategy for HPV screening and vaccination in East Asia? Int J Cancer 119:2713–2715
Jacobs MV, Walboomers JM, Snijders PJ et al (2000) Distribution of 37 mucosotropic HPV types in women with cytologically normal cervical smears: the age-related patterns for high-risk and low-risk types. Int J Cancer 87:221–227
Clifford GM, Gallus S, Herrero R et al (2005) Worldwide distribution of human papillomavirus types in cytologically normal women in the International Agency for Research on Cancer HPV prevalence surveys: a pooled analysis. Lancet 366:991–998
Franceschi S, Herrero R, Clifford GM et al (2006) Variations in the age-specific curves of human papillomavirus prevalence in women worldwide. Int J Cancer 119:2677–2684
de Sanjose S, Quint WG, Alemany L et al (2010) Human papillomavirus genotype attribution in invasive cervical cancer: a retrospective cross-sectional worldwide study. Lancet Oncol 11:1048–1056
Onuki M, Matsumoto K, Satoh T et al (2009) Human papillomavirus infections among Japanese women: age-related prevalence and type-specific risk for cervical cancer. Cancer Sci 100:1312–1316
Gilmour S, Kanda M, Kusumi E et al (2013) HPV vaccination programme in Japan. Lancet 382:768
The Global Advisory Committee on Vaccine Safety (GACVS). GACVS Safety update on HPV vaccines. 13 June 2013. Available via DIALOG. http://www.who.int/vaccine_safety/committee/topics/hpv/130619HPV_VaccineGACVSstatement.pdf Accessed April 2014
The Internaional Federation of Gynecology and Obstetrics (FIGO). FIGO statement on HPV vaccination safety. 2 Aug 2013. Available via DIALOG. http://www.figo.org/files/figo-corp/Statement%20on%20Safety%20of%20HPV%20vaccination%20-%20FINAL%20-%20AUGUST%202013.pdf Accessed April 2014
Darden PM, Thompson DM, Roberts JR et al (2013) Reasons for not vaccinating adolescents: national immunization survey of teens, 2008–2010. Pediatrics 131:645–651
Wong MC, Lee A, Ngai KL et al (2013) Knowledge, attitude, practice and barriers on vaccination against human papillomavirus infection: a cross-sectional study among primary care physicians in Hong Kong. PLoS One 8:e71827
Sotiriadis A, Dagklis T, Siamanta V et al (2012) Increasing fear of adverse effects drops intention to vaccinate after the introduction of prophylactic HPV vaccine. Arch Gynecol Obstet 285:1719–1724
Bodemer N, Muller SM, Okan Y et al (2012) Do the media provide transparent health information? A cross-cultural comparison of public information about the HPV vaccine. Vaccine 30:3747–3756
Holman DM, Benard V, Roland KB et al (2014) Barriers to human papillomavirus vaccination among US adolescents: a systematic review of the literature. JAMA Pediatr 168:76–82
Kessels SJ, Marshall HS, Watson M et al (2012) Factors associated with HPV vaccine uptake in teenage girls: a systematic review. Vaccine 30:3546–3556
Hanley SJ, Yoshioka E, Ito Y et al (2012) Acceptance of and attitudes towards human papillomavirus vaccination in Japanese mothers of adolescent girls. Vaccine 30:5740–5747
Dinh TA, Rosenthal SL, Doan ED et al (2007) Attitudes of mothers in Da Nang, Vietnam toward a human papillomavirus vaccine. J Adolesc Health 40:559–563
Dempsey AF, Abraham LM, Dalton V et al (2009) Understanding the reasons why mothers do or do not have their adolescent daughters vaccinated against human papillomavirus. Ann Epidemiol 19:531–538
Japanese Ministry of Health, Labour and Welfare. Healthcare Services where HPV vaccines are inoculated. Available via DIALOG. http://www.mhlw.go.jp/file/05-Shingikai-10601000-Daijinkanboukouseikagakuka-Kouseikagakuka/0000038483.pdf Accessed April 2014
Daley MF, Crane LA, Markowitz LE et al (2010) Human papillomavirus vaccination practices: a survey of US physicians 18 months after licensure. Pediatrics 126:425–433
Chow SN, Soon R, Park JS et al (2010) Knowledge, attitudes, and communication around human papillomavirus (HPV) vaccination amongst urban Asian mothers and physicians. Vaccine 28:3809–3817
American Academy of Pediatrics. Pediatricians can lay out evidence to allay fears over HPV vaccine. Available via DIALOG. http://aapnews.aappublications.org/content/early/2014/02/12/aapnews.20140212-1 Accessed May 2014
Acknowledgments
We would like to thank Dr. G. S. Buzard, BAH, for his constructive critique and editing of our manuscript. We also thank all the doctors who registered relevant cases in the OCEAN study, and all the registrants of the OCEAN study who responded to our questionnaire. We deeply appreciate Dr. Osamu Ishiko, Dr. Hiroshi Hoshiai, Dr. Yoh Watanabe, Ms. Emi Hatano, Ms. Kanako Sakiyama, Ms. Ayaka Tanaka, Ms. Yohimi Tomine, Mr. Jun Fukuyoshi, several medical students of the Osaka University and the Executive Office of the Obstetrical Gynecological Society of Osaka for their great support for the OCEAN study.
Conflict of interest
The present study was financially supported in part by Grant-in-Aid for Cancer Research from the Ministry of Health, Labour and Welfare, Japan.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Morimoto, A., Ueda, Y., Egawa-Takata, T. et al. Effect on HPV vaccination in Japan resulting from news report of adverse events and suspension of governmental recommendation for HPV vaccination. Int J Clin Oncol 20, 549–555 (2015). https://doi.org/10.1007/s10147-014-0723-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-014-0723-1