Abstract
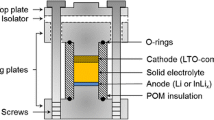
Amorphous titanium trisulfide (TiS3) active materials were prepared by ball milling of an equimolar mixture of crystalline titanium disulfide (TiS2) and sulfur. A high-resolution transmission electron microscope image revealed no periodic lattice fringes on the amorphous TiS3. The all-solid-state lithium secondary batteries using a sulfide solid electrolyte and the amorphous TiS3 electrode showed high capacity of greater than 300 mAh g−1 for 10 cycles. The amorphous TiS3 had a higher capacity than the mixture of crystalline TiS2 and S, which was used as the starting material of amorphous TiS3. The X-ray diffraction patterns and the Raman spectra of the amorphous TiS3 electrode after the first and tenth charge–discharge measurements were similar to those before the measurement. The amorphous structure of TiS3 did not change greatly during the first few cycles. The all-solid-state cells with the amorphous TiS3 electrode showed higher initial coulombic efficiency because the amorphous TiS3 active material retained its structure during the initial electrochemical test.




Similar content being viewed by others
References
Ohzuku T, Ueda A (1994) J Electrochem Soc 141:2972–2977
Endo M, Kim C, Nishimura K, Fujino T (2000) Miyashita K 38:183–197
Zheng H, Li B, Fu Y, Abe T (2006) Ogumi Z 52:1556–1562
Kamaya N, Homma K, Yamakawa Y, Hirayama M, Kanno R, Yonemura M, Kamiyama T, Kato Y, Hama S, Kawamoto K, Mitsui A (2011) Nat Mater 10:682–686
Minami K, Hayashi A, Tatsumisago M (2011) J Am Ceram Soc 94:1779–1783
Komiya R, Hayashi A, Morimoto H, Tatsumisago M, Minami T (2001) Solid State Ionics 140:83–87
Mizuno F, Hama S, Hayashi A, Tadanaga K, Minami T, Tatsumisago M (2002) Chem Lett 12:1244–1245
Mizuno F, Hayashi A, Tadanaga K, Tatsumisago M (2005) J Power Sources 146:711–714
Tatsumisago M, Hayashi A (2008) Funct Matter Lett 1:31–36
Holleck GL, Driscoll JR (1977) Electrochemica Acta 22:647–655
Whittingham MS, Panella JA (1981) Mat Res Bull 16:37–45
Iwamoto K, Aotani N, Takada K, Kondo S (1994) Solid State Ionics 70–71:658–661
Trevey JE, Stoldt CR, Lee SH (2011) J Electrochem Soc 158:A1282–A1289
Onuki Y, Inada R, Tanuma S, Yamanaka S, Kamimura H (1983) Solid State Ionics 11:195–201
Lindic MH, Martinez H, Benayad A, Pecquenard B, Vinatier P, Levasseur A, Gonbeau D (2005) Solid State Ionics 176:1529–1537
Machida N, Fuchida R, Minami T (1989) J Electrochem Soc 136:2133–2136
Ku JH, Ryu JH, Kim SH, Han OH, Oh SM (2012) Adv Funct Mater 22:3658–3664
Hayashi A, Matsuyama T, Sakuda A, Tatsumisago M (2012) Chem Lett 41:886–888
Hayashi A, Hama S, Minami T, Tatsumisago M (2003) Electrochem Commun 5:111–114
Takada K, Aotani N, Iwamoto K, Kondo K (1996) Solid State Ionics 86–88:877–882
Acknowledgments
This research was financially supported by the Japan Science and Technology Agency (JST), Core Research for Evolutional Science and Technology (CREST) project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Matsuyama, T., Sakuda, A., Hayashi, A. et al. Electrochemical properties of all-solid-state lithium batteries with amorphous titanium sulfide electrodes prepared by mechanical milling. J Solid State Electrochem 17, 2697–2701 (2013). https://doi.org/10.1007/s10008-013-2157-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-013-2157-5