Abstract
Purpose
The aim of this study was to demonstrate regeneration of intervertebral discs undergoing laser therapy with sagittal relaxation time (T2) mapping after a long-term follow-up.
Materials and methods
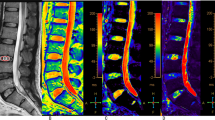
Fourteen patients (9 men, 5 women; age range 20–57 years; mean age 36.5 years) treated with percutaneous 908-nm wave-length diode laser nucleoplasty for lumbar disc prolapsus at our clinic between January 2006 and June 2009 were studied. For the application of laser nucleoplasty in the past, patients who did not have central canal stenosis and/or lateral stenosis, sequestered disc fragment, operation scars and bleeding disorders were selected. The intervertebral disc levels undergoing laser therapy were L3–L4 (n = 2) or L4–L5 (n = 12). Patients were called for follow-up visits after a maximum 6-years (n = 2) or a minimum 3 years (n = 3) with a mean of 4.4 years. The patients’ clinical status for leg pain was evaluated according to the visual analog scale (VAS) and subsequently, a lumbar magnetic resonance imaging was performed. Sagittal T2 mapping was performed for the intervertebral discs undergoing laser nucleoplasty. We analyzed the relationship between T2 in the regions of interest (ROIs), which is known to correlate with changes in the composition of intervertebral discs, and the degree of degeneration determined using the Pfirrmann grading system and VAS of patients.
Results
On the basis of the evaluation of the results of intervertebral discs in all patients, there was a significant increase in T2 in the anterior NP (ROI 2, +10.3 ms; p < 0.05). A significant increase was noted in T2 in the middle NP (ROI 3, +24.6 ms; p < 0.001). The most significant increase was recorded for the posterior NP (ROI 4, +28.6 ms; p < 0.001). No significant decrease was found in T2 in the anterior and posterior AF (ROI 1, −1.5 ms; p = 0.925; ROI 5, −0.1 ms; p = 0.683). According to the Pfirrmann grading system, disc degeneration grades before laser therapy were recorded as grade III (n = 6) and grade IV (n = 8) whereas disc degeneration grades after laser therapy were found to be grade I (n = 6) and II (n = 8). A significant decrease was noted in Pfirrmann grades of disc degeneration after laser therapy (p < 0.0005).
Conclusions
In this study, there was a prolongation of T2 indicating regeneration in the nucleus pulposus after laser therapy and these results were found to be consistent with VAS measurements after a long-term follow-up. This study, which demonstrates the quantitative efficacy of laser therapy, indicates that MRG can be more effectively used in the future.


Similar content being viewed by others
References
Watanabe A, Benneker LM, Boesch C, Watanabe T, Obata T, Anderson SE (2007) Classification of intervertebral disk degeneration with axial T2 mapping. AJR Am J Roentgenol 189(4):936–942
Masuda K, An HS (2006) Prevention of disc degeneration with growth factors. Eur Spine J 15:422–432
Leung VY, Chan D, Cheung KM (2006) Regeneration of intervertebral disc by mesenchymal stem cells: potentials, limitations, and future direction. Eur Spine J 15:406–413
Evans C (2006) Potential biologic therapies for the intervertebraldisc. J Bone Joint Surg Am 88:95–98
Burstein D, Gray ML (2006) Is MRI fulfilling its promise for molecular imaging of cartilage in arthritis? Osteoarthr Cartil 14:1087–1090
Welsch GH, Trattnig S, Domayer S, Marlovits S, White LM, Mamisch TC (2009) Multimodal approach in the use of clinical scoring, morphological MRI and biochemical T2-mapping and diffusion-weighted imaging in their ability to assess differences between cartilage repair tissue after microfracture therapy and matrix-associated autologous chondrocyte transplantation: a pilot study. Osteoarthr Cartil 17:1219–1227
Tertti M, Paajanen H, Laato M, Aho H, Komu M, Kormano M (1991) Disc degeneration in magnetic resonance imaging: a comparative biochemical, histologic, and radiologic study in cadaver spines. Spine 16:629–634
Antoniou J, Pike GB, Steffen T, Baramki H, Poole AR, Aebi M, Alini M (1998) Quantitative magnetic resonance imaging in the assessment of degenerative disc disease. Magn Reson Med 40:900–907
Chatani K, Kusaka Y, Mifune T, Nishikawa H (1993) Topographic differences of 1H-NMR relaxation times (T1, T2) in the normal intervertebral discand its relationship to water content. Spine 18:2271–2275
Eyre D, Benya P, Buckwalter J, Gatersion B, Heinegard D, Oegema T et al (1989) Intervertebral disks: part B. Basic science perspectives. In: Frymoyer JW, Gordon SL (eds) New perspectives on low back pain. Park Ridge, Chicago, pp 147–207
Thompson JP, Pearce RH, Schechter MT, Adams ME, Tsang IK, Bishop PB (1990) Preliminary evaluation of a scheme for grading the gross morphology of the human intervertebral disc. Spine 15:411–415
Haefeli M, Kalberer F, Saegesser D, Nerlich AG, Boos N, Paesold G (2006) The course of macroscopic degeneration in the human lumbar intervertebral disc. Spine 31:1522–1531
Perry J, Haughton V, Anderson PA, Wu Y, Fine J, Mistretta C (2006) The value of T2 relaxation times to characterize lumbar intervertebral disks: preliminary results. AJNR Am J Neuroradiol 27(2):337–342
Blumenkrantz G, Zuo J, Li X, Kornak J, Link TM, Majumdar S (2010) In vivo 3.0-tesla magnetic resonance T1 rho and T2 relaxation mapping in subjects with intervertebral disc degeneration and clinical symptoms. Magn Reson Med 63:1193–1200
Trattnig S, Stelzeneder D, Goed S, Reissegger M, Mamisch TC, Paternostro-Sluga T, Weber M, Szomolanyi P, Welsch GH (2010) Lumbar intervertebral disc abnormalities: comparison of quantitative T2 mapping with conventional MR at 3.0 T. Eur Radiol 20:2715–2722
Pfirrmann CW, Metzdorf A, Zanetti M, Hodler J, Boos N (2001) Magnetic resonance classification of lumbar intervertebral disc degeneration. Spine 26:1873–1878
Schenk B, Brouwer PA, van Buchem MA (2006) Experimental basis of percutaneous laser disc decompression (PLDD): a review of literature. Lasers Med Sci 21(4):245–249
Choy DS, Michelsen J, Getrajdman G, Diwan S (1992) Percutaneous laser disc decompression: an update—spring 1992. J Clin Laser Med Surg 10:177–184
Choy DS, Altman P (1995) Fall of intradiscal pressure with laser ablation. J Clin Laser Med Surg 13(3):149–151
Choi JY, Tanenbaum BS, Milner TE, Dao XV, Nelson JS, Sobol EN, Wong BJ (2001) Thermal, mechanical, optical, and morphologic changes in bovine nucleus pulposus induced by Nd:YAG (lambda = 1.32 microm) laser irradiation. Lasers Surg Med 28(3):248–254
Shen CC, Yang YC, Liu BS (2011) Large-area irradiated low-level laser effect in a biodegradable nerve guide conduit on neural regeneration of peripheral nerve injury in rats. Injury 42(8):803–813
Câmara CN, Brito MV, Silveira EL, Silva DS, Simões VR, Pontes RW (2011) Histological analysis of low-intensity laser therapy effects in peripheral nerve regeneration in Wistar rats. Acta Cir Bras 26(1):12–18
Gigo-Benato D, Russo TL, Tanaka EH, Assis L, Salvini TF, Parizotto NA (2010) Effects of 660 and 780 nm low-level laser therapy on neuromuscular recovery after crush injury in rat sciatic nerve. Lasers Surg Med 42(9):673–682
Barbosa RI, Marcolino AM, de Jesus Guirro RR, Mazzer N, Barbieri CH, de Cássia Registro Fonseca M (2010) Comparative effects of wavelengths of low-power laser in regeneration of sciatic nerve in rats following crushing lesion. Lasers Med Sci 25(3):423–430
Sobol EN, Vorobjeva NN, Sviridov AP, Omelchenko AI, Baskov AV, Shekhter AB, Baskov VA, Feldchtein FI, Kamensky VA, Kuranov RV (2000) Laser-induced activation of regeneration processes in spine disc cartilage. In: Proceedings of SPIE 3907, lasers in surgery: advanced characterization, therapeutics, and systems X, p 504. doi:10.1117/12.386293
Adah F, Benghuzzi H, Tucci M, Ragab A, Greenwald N (2008) Effect of low power laser treatment on a traumatized disc in a rat model. Biomed Sci Instrum 44:34–40
Thal DR, Werkmann K, Leheta F, Schober R, Ulrich P (1996) Effects of Nd:YAG laser radiation in cultured porcine vertebral disc tissue. Proc SPIE Int Soc Opt Eng 2623:312–320. doi:10.1117/12.230343
Stelzeneder D, Kovács BK, Goed S, Welsch GH, Hirschfeld C, Paternostro-Sluga T, Friedrich KM, Mamisch TC, Trattnig S (2012) Effect of short-term unloading on T2 relaxation time in the lumbar intervertebral disc–in vivo magnetic resonance imaging study at 3.0 tesla. Spine J 12(3):257–264
Auerbach JD, Johannessen W, Borthakur A, Wheaton AJ, Dolinskas CA, Balderston RA, Reddy R, Elliott DM (2006) In vivo quantification of human lumbar disc degeneration using T (1rho)-weighted magnetic resonance imaging. Eur Spine J 15(3):338–344
Boos N, Wallin A, Gbedegbegnon T, Aebi M, Boesch C (1993) Quantitative MR imaging of lumbar intervertebral disks and vertebral bodies: influence of diurnal water content variations. Radiology 188:351–354
Acknowledgments
We thank Dr. Derun Taner Ertugrul for performing statistical analysis of data.
Conflict of interest
There are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arslan, E., Demirci, I., Kılıncaslan, M.O. et al. Identification of intervertebral disc regeneration with magnetic resonance imaging after a long-term follow-up in patients treated with percutaneous diode laser nucleoplasty: a retrospective clinical and radiological analysis of 14 patients. Eur Spine J 23, 1044–1051 (2014). https://doi.org/10.1007/s00586-014-3194-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-014-3194-1