Abstract
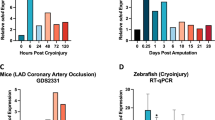
Cardiac fibroblasts become activated and differentiate to smooth muscle-like myofibroblasts in response to hypertension and myocardial infarction (MI), resulting in extracellular matrix (ECM) remodeling, scar formation and impaired cardiac function. cAMP and cGMP-dependent signaling have been implicated in cardiac fibroblast activation and ECM synthesis. Dysregulation of cyclic nucleotide phosphodiesterase (PDE) activity/expression is also associated with various diseases and several PDE inhibitors are currently available or in development for treating these pathological conditions. The objective of this study is to define and characterize the specific PDE isoform that is altered during cardiac fibroblast activation and functionally important for regulating myofibroblast activation and ECM synthesis. We have found that Ca2+/calmodulin-stimulated PDE1A isoform is specifically induced in activated cardiac myofibroblasts stimulated by Ang II and TGF-β in vitro as well as in vivo within fibrotic regions of mouse, rat, and human diseased hearts. Inhibition of PDE1A function via PDE1-selective inhibitor or PDE1A shRNA significantly reduced Ang II or TGF-β-induced myofibroblast activation, ECM synthesis, and pro-fibrotic gene expression in rat cardiac fibroblasts. Moreover, the PDE1 inhibitor attenuated isoproterenol-induced interstitial fibrosis in mice. Mechanistic studies revealed that PDE1A modulates unique pools of cAMP and cGMP, predominantly in perinuclear and nuclear regions of cardiac fibroblasts. Further, both cAMP-Epac-Rap1 and cGMP-PKG signaling was involved in PDE1A-mediated regulation of collagen synthesis. These results suggest that induction of PDE1A plays a critical role in cardiac fibroblast activation and cardiac fibrosis, and targeting PDE1A may lead to regression of the adverse cardiac remodeling associated with various cardiac diseases.








Similar content being viewed by others
References
Aizawa T, Wei H, Miano JM, Abe J, Berk BC, Yan C (2003) Role of phosphodiesterase 3 in NO/cGMP-mediated antiinflammatory effects in vascular smooth muscle cells. Circ Res 93:406–413. doi:10.1161/01.RES.0000091074.33584.F0
Andric SA, Kostic TS, Stojilkovic SS (2006) Contribution of multidrug resistance protein MRP5 in control of cyclic guanosine 5′-monophosphate intracellular signaling in anterior pituitary cells. Endocrinology 147:3435–3445. doi:10.1210/en.2006-0091
Batchelor AM, Bartus K, Reynell C, Constantinou S, Halvey EJ, Held KF, Dostmann WR, Vernon J, Garthwaite J (2010) Exquisite sensitivity to subsecond, picomolar nitric oxide transients conferred on cells by guanylyl cyclase-coupled receptors. Proc Natl Acad Sci USA 107:22060–22065. doi:10.1073/pnas.1013147107
Bax NA, van Oorschot AA, Maas S, Braun J, van Tuyn J, de Vries AA, Groot AC, Goumans MJ (2011) In vitro epithelial-to-mesenchymal transformation in human adult epicardial cells is regulated by TGFbeta-signaling and WT1. Basic Res Cardiol 106:829–847. doi:10.1007/s00395-011-0181-0
Beavo JA, Brunton LL (2002) Cyclic nucleotide research—still expanding after half a century. Nat Rev Mol Cell Biol 3:710–718. doi:10.1038/nrm911
Beavo JA, Hardman JG, Sutherland EW (1970) Hydrolysis of cyclic guanosine and adenosine 3′, 5′-monophosphates by rat and bovine tissues. J Biol Chem 245:5649–5655
Berk BC, Fujiwara K, Lehoux S (2007) ECM remodeling in hypertensive heart disease. J Clin Invest 117:568–575. doi:10.1172/JCI31044
Bode DC, Kanter JR, Brunton LL (1991) Cellular distribution of phosphodiesterase isoforms in rat cardiac tissue. Circ Res 68:1070–1079
Brown RD, Ambler SK, Mitchell MD, Long CS (2005) The cardiac fibroblast: therapeutic target in myocardial remodeling and failure. Annu Rev Pharmacol Toxicol 45:657–687. doi:10.1146/annurev.pharmtox.45.120403.095802
Butt E, Pohler D, Genieser HG, Huggins JP, Bucher B (1995) Inhibition of cyclic GMP-dependent protein kinase-mediated effects by (Rp)-8-bromo-PET-cyclic GMPS. Br J Pharmacol 116:3110–3116
Cai Y, Miller CL, Nagel DJ, Jeon KI, Lim S, Gao P, Knight PA, Yan C (2011) Cyclic nucleotide phosphodiesterase 1 regulates lysosome-dependent type I collagen protein degradation in vascular smooth muscle cells. Arterioscler Thromb Vasc Biol 31:616–623. doi:10.1161/atvbaha.110.212621
Calderone A, Thaik CM, Takahashi N, Chang DL, Colucci WS (1998) Nitric oxide, atrial natriuretic peptide, and cyclic GMP inhibit the growth-promoting effects of norepinephrine in cardiac myocytes and fibroblasts. J Clin Invest 101:812–818. doi:10.1172/JCI119883
Castoldi G, Di Gioia CR, Pieruzzi F, D’Orlando C, Van De Greef WM, Busca G, Sperti G, Stella A (2003) ANG II increases TIMP-1 expression in rat aortic smooth muscle cells in vivo. Am J Physiol Heart Circ Physiol 284:H635–H643. doi:10.1152/ajpheart.00986.2001
Castro LR, Schittl J, Fischmeister R (2010) Feedback control through cGMP-dependent protein kinase contributes to differential regulation and compartmentation of cGMP in rat cardiac myocytes. Circ Res 107:1232–1240. doi:10.1161/circresaha.110.226712
Chorianopoulos E, Heger T, Lutz M, Frank D, Bea F, Katus HA, Frey N (2010) FGF-inducible 14-kDa protein (Fn14) is regulated via the RhoA/ROCK kinase pathway in cardiomyocytes and mediates nuclear factor-kappaB activation by TWEAK. Basic Res Cardiol 105:301–313. doi:10.1007/s00395-009-0046-y
Ding B, Abe J, Wei H, Huang Q, Walsh RA, Molina CA, Zhao A, Sadoshima J, Blaxall BC, Berk BC, Yan C (2005) Functional role of phosphodiesterase 3 in cardiomyocyte apoptosis: implication in heart failure. Circulation 111:2469–2476. doi:10.1161/01.CIR.0000165128.39715.87
DiPilato LM, Cheng X, Zhang J (2004) Fluorescent indicators of cAMP and Epac activation reveal differential dynamics of cAMP signaling within discrete subcellular compartments. Proc Natl Acad Sci U S A 101:16513–16518. doi:10.1073/pnas.0405973101
Dostmann WR, Taylor MS, Nickl CK, Brayden JE, Frank R, Tegge WJ (2000) Highly specific, membrane-permeant peptide blockers of cGMP-dependent protein kinase Ialpha inhibit NO-induced cerebral dilation. Proc Natl Acad Sci USA 97:14772–14777. doi:10.1073/pnas.97.26.14772
Doyle DD, Upshaw-Earley J, Bell EL, Palfrey HC (2002) Natriuretic peptide receptor-B in adult rat ventricle is predominantly confined to the nonmyocyte population. Am J Physiol Heart Circ Physiol 282:H2117–H2123. doi:10.1152/ajpheart.00988.2001
Drake MT, Violin JD, Whalen EJ, Wisler JW, Shenoy SK, Lefkowitz RJ (2008) beta-arrestin-biased agonism at the beta2-adrenergic receptor. J Biol Chem 283:5669–5676. doi:10.1074/jbc.M708118200
Fischmeister R, Castro LR, Abi-Gerges A, Rochais F, Jurevicius J, Leroy J, Vandecasteele G (2006) Compartmentation of cyclic nucleotide signaling in the heart: the role of cyclic nucleotide phosphodiesterases. Circ Res 99:816–828. doi:10.1161/01.RES.0000246118.98832.04
Giasson E, Servant MJ, Meloche S (1997) Cyclic AMP-mediated inhibition of angiotensin II-induced protein synthesis is associated with suppression of tyrosine phosphorylation signaling in vascular smooth muscle cells. J Biol Chem 272:26879–26886
Hammoud L, Lu X, Lei M, Feng Q (2011) Deficiency in TIMP-3 increases cardiac rupture and mortality post-myocardial infarction via EGFR signaling: beneficial effects of cetuximab. Basic Res Cardiol 106:459–471. doi:10.1007/s00395-010-0147-7
Hao Y, Xu N, Box AC, Schaefer L, Kannan K, Zhang Y, Florens L, Seidel C, Washburn MP, Wiegraebe W, Mak HY (2011) Nuclear cGMP-dependent kinase regulates gene expression via activity-dependent recruitment of a conserved histone deacetylase complex. PLoS Genet 7:e1002065. doi:10.1371/journal.pgen.1002065
Haworth RS, Cuello F, Avkiran M (2011) Regulation by phosphodiesterase isoforms of protein kinase A-mediated attenuation of myocardial protein kinase D activation. Basic Res Cardiol 106:51–63. doi:10.1007/s00395-010-0116-1
Jeon KI, Jono H, Miller CL, Cai Y, Lim S, Liu X, Gao P, Abe J, Li JD, Yan C (2010) Ca2+/calmodulin-stimulated PDE1 regulates the beta-catenin/TCF signaling through PP2A B56 gamma subunit in proliferating vascular smooth muscle cells. FEBS J 277:5026–5039. doi:10.1111/j.1742-4658.2010.07908.x
Kim D, Rybalkin SD, Pi X, Wang Y, Zhang C, Munzel T, Beavo JA, Berk BC, Yan C (2001) Upregulation of phosphodiesterase 1A1 expression is associated with the development of nitrate tolerance. Circulation 104:2338–2343
Kim HE, Dalal SS, Young E, Legato MJ, Weisfeldt ML, D’Armiento J (2000) Disruption of the myocardial extracellular matrix leads to cardiac dysfunction. J Clin Invest 106:857–866. doi:10.1172/jci8040
Kim NN, Villegas S, Summerour SR, Villarreal FJ (1999) Regulation of cardiac fibroblast extracellular matrix production by bradykinin and nitric oxide. J Mol Cell Cardiol 31:457–466. doi:10.1006/jmcc.1998.0887
Klaiber M, Kruse M, Volker K, Schroter J, Feil R, Freichel M, Gerling A, Feil S, Dietrich A, Londono JE, Baba HA, Abramowitz J, Birnbaumer L, Penninger JM, Pongs O, Kuhn M (2010) Novel insights into the mechanisms mediating the local antihypertrophic effects of cardiac atrial natriuretic peptide: role of cGMP-dependent protein kinase and RGS2. Basic Res Cardiol 105:583–595. doi:10.1007/s00395-010-0098-z
Koutalos Y, Nakatani K, Yau KW (1995) Cyclic GMP diffusion coefficient in rod photoreceptor outer segments. Biophys J 68:373–382. doi:10.1016/s0006-3495(95)80198-0
Leask A, Abraham DJ (2004) TGF-beta signaling and the fibrotic response. FASEB J 18:816–827. doi:10.1096/fj.03-1273rev
Lee DI, Vahebi S, Tocchetti CG, Barouch LA, Solaro RJ, Takimoto E, Kass DA (2010) PDE5A suppression of acute beta-adrenergic activation requires modulation of myocyte beta-3 signaling coupled to PKG-mediated troponin I phosphorylation. Basic Res Cardiol 105:337–347. doi:10.1007/s00395-010-0084-5
Leineweber K, Bohm M, Heusch G (2006) Cyclic adenosine monophosphate in acute myocardial infarction with heart failure: slayer or savior? Circulation 114:365–367. doi:10.1161/circulationaha.106.642132
Li P, Wang D, Lucas J, Oparil S, Xing D, Cao X, Novak L, Renfrow MB, Chen YF (2008) Atrial natriuretic peptide inhibits transforming growth factor beta-induced Smad signaling and myofibroblast transformation in mouse cardiac fibroblasts. Circ Res 102:185–192. doi:10.1161/CIRCRESAHA.107.157677
Lukowski R, Rybalkin SD, Loga F, Leiss V, Beavo JA, Hofmann F (2010) Cardiac hypertrophy is not amplified by deletion of cGMP-dependent protein kinase I in cardiomyocytes. Proc Natl Acad Sci USA 107:5646–5651. doi:10.1073/pnas.1001360107
Masuyama H, Tsuruda T, Kato J, Imamura T, Asada Y, Stasch JP, Kitamura K, Eto T (2006) Soluble guanylate cyclase stimulation on cardiovascular remodeling in angiotensin II-induced hypertensive rats. Hypertension 48:972–978. doi:10.1161/01.HYP.0000241087.12492.47
Miller CL, Oikawa M, Cai Y, Wojtovich AP, Nagel DJ, Xu X, Xu H, Florio V, Rybalkin SD, Beavo JA, Chen YF, Li JD, Blaxall BC, Abe J, Yan C (2009) Role of Ca2 +/calmodulin-stimulated cyclic nucleotide phosphodiesterase 1 in mediating cardiomyocyte hypertrophy. Circ Res 105:956–964. doi:10.1161/CIRCRESAHA.109.198515
Nagel DJ, Aizawa T, Jeon KI, Liu W, Mohan A, Wei H, Miano JM, Florio VA, Gao P, Korshunov VA, Berk BC, Yan C (2006) Role of nuclear Ca2 +/calmodulin-stimulated phosphodiesterase 1A in vascular smooth muscle cell growth and survival. Circ Res 98:777–784. doi:10.1161/01.RES.0000215576.27615.fd
Nakamura H, Isaka Y, Tsujie M, Rupprecht HD, Akagi Y, Ueda N, Imai E, Hori M (2002) Introduction of DNA enzyme for Egr-1 into tubulointerstitial fibroblasts by electroporation reduced interstitial alpha-smooth muscle actin expression and fibrosis in unilateral ureteral obstruction (UUO) rats. Gene Ther 9:495–502. doi:10.1038/sj.gt.3301681
Nausch LW, Ledoux J, Bonev AD, Nelson MT, Dostmann WR (2008) Differential patterning of cGMP in vascular smooth muscle cells revealed by single GFP-linked biosensors. Proc Natl Acad Sci USA 105:365–370. doi:10.1073/pnas.0710387105
Nikolaev VO, Bunemann M, Hein L, Hannawacker A, Lohse MJ (2004) Novel single chain cAMP sensors for receptor-induced signal propagation. J Biol Chem 279:37215–37218. doi:10.1074/jbc.C400302200
Nikolaev VO, Lohse MJ (2006) Monitoring of cAMP synthesis and degradation in living cells. Physiology (Bethesda) 21:86–92. doi:10.1152/physiol.00057.2005
Oerlemans MI, Goumans MJ, van Middelaar B, Clevers H, Doevendans PA, Sluijter JP (2010) Active Wnt signaling in response to cardiac injury. Basic Res Cardiol 105:631–641. doi:10.1007/s00395-010-0100-9
Ostrom RS, Naugle JE, Hase M, Gregorian C, Swaney JS, Insel PA, Brunton LL, Meszaros JG (2003) Angiotensin II enhances adenylyl cyclase signaling via Ca2 +/calmodulin. Gq-Gs cross-talk regulates collagen production in cardiac fibroblasts. J Biol Chem 278:24461–24468. doi:10.1074/jbc.M212659200
Pandey KN (2010) Ligand-mediated endocytosis and intracellular sequestration of guanylyl cyclase/natriuretic peptide receptors: role of GDAY motif. Mol Cell Biochem 334:81–98. doi:10.1007/s11010-009-0332-x
Pilz RB, Casteel DE (2003) Regulation of gene expression by cyclic GMP. Circ Res 93:1034–1046. doi:10.1161/01.res.0000103311.52853.48
Ponsioen B, Zhao J, Riedl J, Zwartkruis F, van der Krogt G, Zaccolo M, Moolenaar WH, Bos JL, Jalink K (2004) Detecting cAMP-induced Epac activation by fluorescence resonance energy transfer: Epac as a novel cAMP indicator. EMBO Rep 5:1176–1180. doi:10.1038/sj.embor.7400290
Porter KE, Turner NA (2009) Cardiac fibroblasts: at the heart of myocardial remodeling. Pharmacol Ther 123:255–278. doi:10.1016/j.pharmthera.2009.05.002
Rich TC, Xin W, Mehats C, Hassell KA, Piggott LA, Le X, Karpen JW, Conti M (2007) Cellular mechanisms underlying prostaglandin-induced transient cAMP signals near the plasma membrane of HEK-293 cells. Am J Physiol Cell Physiol 292:C319–C331. doi:10.1152/ajpcell.00121.2006
Richter W, Xie M, Scheitrum C, Krall J, Movsesian MA, Conti M (2011) Conserved expression and functions of PDE4 in rodent and human heart. Basic Res Cardiol 106:249–262. doi:10.1007/s00395-010-0138-8
Rybalkin SD, Yan C, Bornfeldt KE, Beavo JA (2003) Cyclic GMP phosphodiesterases and regulation of smooth muscle function. Circ Res 93:280–291. doi:10.1161/01.RES.0000087541.15600.2B
Sassi Y, Lipskaia L, Vandecasteele G, Nikolaev VO, Hatem SN, Cohen Aubart F, Russel FG, Mougenot N, Vrignaud C, Lechat P, Lompre AM, Hulot JS (2008) Multidrug resistance-associated protein 4 regulates cAMP-dependent signaling pathways and controls human and rat SMC proliferation. J Clin Invest 118:2747–2757. doi:10.1172/JCI35067
Shishido T, Woo CH, Ding B, McClain C, Molina CA, Yan C, Yang J, Abe J (2008) Effects of MEK5/ERK5 association on small ubiquitin-related modification of ERK5: implications for diabetic ventricular dysfunction after myocardial infarction. Circ Res 102:1416–1425. doi:10.1161/circresaha.107.168138
Snyder PB, Florio VA, Ferguson K, Loughney K (1999) Isolation, expression and analysis of splice variants of a human Ca2 +/calmodulin-stimulated phosphodiesterase (PDE1A). Cell Signal 11:535–544 (S0898-6568(99)00027-3[pii])
Sonnenburg WK, Seger D, Beavo JA (1993) Molecular cloning of a cDNA encoding the “61-kDa” calmodulin-stimulated cyclic nucleotide phosphodiesterase. Tissue-specific expression of structurally related isoforms. J Biol Chem 268:645–652
Swaney JS, Roth DM, Olson ER, Naugle JE, Meszaros JG, Insel PA (2005) Inhibition of cardiac myofibroblast formation and collagen synthesis by activation and overexpression of adenylyl cyclase. Proc Natl Acad Sci USA 102:437–442. doi:10.1073/pnas.0408704102
Takizawa T, Gu M, Chobanian AV, Brecher P (1997) Effect of nitric oxide on DNA replication induced by angiotensin II in rat cardiac fibroblasts. Hypertension 30:1035–1040
Terrin A, Di Benedetto G, Pertegato V, Cheung YF, Baillie G, Lynch MJ, Elvassore N, Prinz A, Herberg FW, Houslay MD, Zaccolo M (2006) PGE(1) stimulation of HEK293 cells generates multiple contiguous domains with different [cAMP]: role of compartmentalized phosphodiesterases. J Cell Biol 175:441–451. doi:10.1083/jcb.200605050
Tiede K, Melchior-Becker A, Fischer JW (2010) Transcriptional and posttranscriptional regulators of biglycan in cardiac fibroblasts. Basic Res Cardiol 105:99–108. doi:10.1007/s00395-009-0049-8
Tomasek JJ, Gabbiani G, Hinz B, Chaponnier C, Brown RA (2002) Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat Rev Mol Cell Biol 3:349–363. doi:10.1038/nrm809
Tsai EJ, Kass DA (2009) Cyclic GMP signaling in cardiovascular pathophysiology and therapeutics. Pharmacol Ther 122:216–238. doi:10.1016/j.pharmthera.2009.02.009
van den Borne SW, Diez J, Blankesteijn WM, Verjans J, Hofstra L, Narula J (2010) Myocardial remodeling after infarction: the role of myofibroblasts. Nat Rev Cardiol 7:30–37. doi:10.1038/nrcardio.2009.199
Villarreal F, Epperson SA, Ramirez-Sanchez I, Yamazaki KG, Brunton LL (2009) Regulation of cardiac fibroblast collagen synthesis by adenosine: roles for Epac and PI3 K. Am J Physiol Cell Physiol 296:C1178–C1184. doi:10.1152/ajpcell.00291.2008
von Hayn K, Werthmann RC, Nikolaev VO, Hommers LG, Lohse MJ, Bunemann M (2010) Gq-mediated Ca2+ signals inhibit adenylyl cyclases 5/6 in vascular smooth muscle cells. Am J Physiol Cell Physiol 298:C324–C332. doi:10.1152/ajpcell.00197.2009
Willems IE, Havenith MG, De Mey JG, Daemen MJ (1994) The alpha-smooth muscle actin-positive cells in healing human myocardial scars. Am J Pathol 145:868–875
Wu M, Melichian DS, de la Garza M, Gruner K, Bhattacharyya S, Barr L, Nair A, Shahrara S, Sporn PH, Mustoe TA, Tourtellotte WG, Varga J (2009) Essential roles for early growth response transcription factor Egr-1 in tissue fibrosis and wound healing. Am J Pathol 175:1041–1055. doi:10.2353/ajpath.2009.090241
Yan C, Kim D, Aizawa T, Berk BC (2003) Functional interplay between angiotensin II and nitric oxide: cyclic GMP as a key mediator. Arterioscler Thromb Vasc Biol 23:26–36
Yokoyama U, Patel HH, Lai NC, Aroonsakool N, Roth DM, Insel PA (2008) The cyclic AMP effector Epac integrates pro- and anti-fibrotic signals. Proc Natl Acad Sci USA 105:6386–6391. doi:10.1073/pnas.0801490105
Zaccolo M (2009) cAMP signal transduction in the heart: understanding spatial control for the development of novel therapeutic strategies. Br J Pharmacol 158:50–60. doi:10.1111/j.1476-5381.2009.00185.x
Zaccolo M, Pozzan T (2002) Discrete microdomains with high concentration of cAMP in stimulated rat neonatal cardiac myocytes. Science 295:1711–1715. doi:10.1126/science.1069982
Zhou HY, Chen WD, Zhu DL, Wu LY, Zhang J, Han WQ, Li JD, Yan C, Gao PJ (2010) The PDE1A-PKCalpha signaling pathway is involved in the upregulation of alpha-smooth muscle actin by TGF-beta1 in adventitial fibroblasts. J Vasc Res 47:9–15. doi:10.1159/000231716
Acknowledgments
We thank Dr. Soyeon Lim, Dr. Nhat-Tu Le, and Dr. Yuichiro Takei (University of Rochester, USA) for providing cardiac fibroblasts. We thank Dr. Kees Jalink (The Netherlands Cancer Institute, The Netherlands) for providing the permission to use the Epac1-H30-cyto construct. We thank Dr. Rajesh Kukreja (Virginia Commonwealth University, USA) for providing Ad-PKG I shRNA. We thank Dr. Jian-Dong Li (University of Rochester, USA) for providing the Smad-binding element-luciferase reporter construct and plasmids encoding Smad2 and Smad3. This work was supported by an American Heart Association Established Investigator Award 0740021N (to C.Y.), NIH grants HL088400 and HL077789 (to C.Y.), American Heart Association Predoctoral Fellowship 0815730D (to C.L.M.), This work was also supported by NIH HL68891 (to W.R.D.) and the Totman Trust for Biomedical Research (to W.R.D.) and by the British Heart Foundation PG/07/091/23698 (to M.Z.).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Miller, C.L., Cai, Y., Oikawa, M. et al. Cyclic nucleotide phosphodiesterase 1A: a key regulator of cardiac fibroblast activation and extracellular matrix remodeling in the heart. Basic Res Cardiol 106, 1023–1039 (2011). https://doi.org/10.1007/s00395-011-0228-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00395-011-0228-2