Abstract
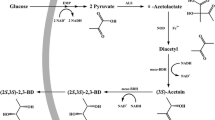
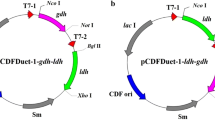
The pure stereoisomers of 1,2-propanediol (1,2-PDO) could be used as starting materials to synthesize high value-added specialty chemicals and chiral pharmaceutical products. As the stereoisomers of 1,2-PDO cannot be obtained by traditional chemical synthesis processes, biotechnological processes have gained increasing attention. However, to our knowledge, the production of S-1,2-PDO directly from glucose has not been previously reported. In this study, we demonstrate a novel artificial pathway to convert l-lactic acid to S-1,2-PDO and its integration into the genome of Escherichia coli strain BW25113∆poxB with synchronous deletion of genes responsible for branch metabolic pathways from glucose. l-lactate production was increased by replacing the native d-lactate dehydrogenase with the l-lactate dehydrogenase from Bacillus coagulans. The methylglyoxal bypass pathway was blocked to avoid synthesis of a racemic mixture of d- and l-lactate and prevent the accumulation of methylglyoxal, a toxic intermediate. To further improve the yield of S-1,2-PDO, a novel cofactor regeneration system was introduced by combining pyruvate decarboxylase and acetaldehyde-CoA dehydrogenase II to simultaneously regenerate NADH and the CoA donor of acetyl-CoA for the lactate conversion pathway. Finally, 13.7 mM S-1,2-PDO with >99 % enantiomeric purity was directly produced from glucose by disrupting the major carbon-competing pathways and strengthening the lactate transformation pathway. This study demonstrates the first attempt to synthesize S-1,2-PDO by direct fermentation of glucose.






Similar content being viewed by others
References
Altaras N, Cameron D (1999) Metabolic engineering of a 1,2-propanediol pathway in Escherichia coli. Appl Environ Microbiol 65:1180–1185
Altaras N, Cameron D (2000) Enhanced production of (R)-1,2-propanediol by metabolically engineered Escherichia coli. Biotechnol Prog 16:940–946
Badía J, Ros J, Aguilar J (1985) Fermentation mechanism of fucose and rhamnose in Salmonella typhimurium and Klebsiella pneumoniae. J Bacteriol 161:435–437
Baldomà L, Aguilar J (1987) Involvement of lactaldehyde dehydrogenase in several metabolic pathways of Escherichia coli K12. J Biol Chem 262:13991–13996
Bennett GN, San KY (2001) Microbial formation, biotechnological production and applications of 1,2-propanediol. Appl Microbiol Biotechnol 55:1–9
Berríos-Rivera SJ, San KY, Bennett GN (2003) The effect of carbon sources and lactate dehydrogenase deletion on 1,2-propanediol production in Escherichia coli. J Ind Microbiol Biotechnol 30:34–40
Booth IR, Ferguson GP, Miller S, Li C, Gunasekera B, Kinghorn S (2003) Bacterial production of methylglyoxal: a survival strategy or death by misadventure? Biochem Soc Trans 31:1406–1408
Boronat A, Aguilar J (1981) Metabolism of L-fucose and L-rhamnose in Escherichia coli: differences in induction of propanediol oxidoreductase. J Bacteriol 147:181–185
Cameron DC, Altaras NE, Hoffman ML, Shaw AJ (1998) Metabolic engineering of propanediol pathways. Biotechnol Prog 14:116–125
Cameron DC, Cooney CL (1986) A novel fermentation: the production of (R)-1,2-propanediol and acetol by Clostridium thermosaccharolyticum. Bioresour Technol 4:651–654
Clomburg J, Gonzalez R (2011) Metabolic engineering of Escherichia coli for the production of 1,2-propanediol from glycerol. Biotechnol Bioeng 108:867–879
Datsenko KA, Wanner BL (2000) One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci U S A 97:6640–6645
Hacking AJ, Lin ECC (1976) Disruption of the fucose pathway as a consequence of genetic adaptation to propanediol as a carbon source in Escherichia coli. J Bacteriol 126:1166–1172
Herrera S (2004) Industrial biotechnology—a chance at redemption. Nat Biotechnol 22:671–678
Heinl S, Wibberg D, Eikmeyer F, Szczepanowski R, Blom J, Linke B, Goesmann A, Grabherr R, Schwab H, Pühler A (2012) Insight into the completely annotated genome of Lactobacillus buchneri CD034, a strain isolated from stable grass silage. J Biotechnol 161:153–166
Jantama K, Zhang X, Moore JC, Shanmugam KT, Svoronos SA, Ingram LO (2008) Eliminating side products and increasing succinate yields in engineered strains of Escherichia coli C. Biotechnol Bioeng 101:881–893
Jain R, Yan Y (2011) Dehydratase mediated 1-propanol production in metabolically engineered Escherichia coli. Microb Cell Fact 10:97
Jain R, Sun X, Yuan Q, Yan Y (2015a) Systematically engineering Escherichia coli for enhanced production of 1,2-propanediol and 1-propanol. ACS Synth Biol 4:746–756
Jain R, Huang J, Yuan Q, Yan Y (2015b) Engineering microaerobic metabolism of E. coli for 1,2-propanediol production. J Ind Microbiol Biotechnol 42:1049–1055
Jiang X, Xue YF, Wang AY, Wang LM, Zhang GM, Zeng QT, Yu B, Ma YH (2013) Efficient production of polymer-grade L-lactate by an alkaliphilic Exiguobacterium sp. strain under nonsterile open fermentation conditions. Bioresour Technol 143:665–668
Lee SJ, Ko JH, Kang HY, Lee Y (2006) Coupled expression of MhpE aldolase and MhpF dehydrogenase in Escherichia coli. Biochem Biophys Res Commun 346:1009–1015
Li Y, Wang LM, Ju JS, Yu B, Ma YH (2013) Efficient production of polymer-grade D-lactate by Sporolactobacillus laevolacticus DSM442 with agricultural waste cottonseed as the sole nitrogen source. Bioresour Technol 142:186–191
Nishino N, Yoshida M, Shiota H, Sakaguchi E (2003) Accumulation of 1,2-propanediol and enhancement of aerobic stability in whole crop maize silage inoculated with Lactobacillus buchneri. J Appl Microbiol 94:800–807
Niu W, Guo J (2015) Stereospecific microbial conversion of lactic acid into 1,2-propanediol. ACS Synth Biol 4:378–382
Oude Elferink SJ, Krooneman J, Gottschal JC, Spoelstra SF, Faber F, Driehuis F (2001) Anaerobic conversion of lactic acid to acetic acid and 1,2-propanediol by Lactobacillus buchneri. Appl Environ Microbiol 67:125–132
Peng LL, Wang LM, Che CC, Yang G, Yu B, Ma YH (2013) Bacillus sp. strain P38: an efficient producer of L-lactate from cellulosic hydrolysate, with high tolerance for 2-furfural. Bioresour Technol 149:169–176
Raj KC, Talarico LA, Ingram LO, Maupin-Furlow JA (2002) Cloning and characterization of the Zymobacter palmae pyruvate decarboxylase gene (pdc) and comparison to bacterial homologues. Appl Environ Microbiol 68:2869–2876
Shelley S (2007) A renewable route to propylene glycol. Chem Eng Prog 103:6–9
Simon ES, Whitesides M, Cameron DC, Weitz DJ, Cooney CL (1987) A combined microbial/chemical synthesis of (+)-(R)-methyloxirane having high enantiomeric excess. J Org Chem 52:4042–4044
Sanchez-Rivera F, Cameron DC, Cooney CL (1987) Influence of environmental factors in the production of 1,2-propanediol by Clostridium thermosaccharolyticum. Biotechnol Lett 9:449–454
Slusarczyk H, Felber S, Kula MR, Pohl M (2000) Stabilization of NAD-dependent formate dehydrogenase from Candida boidinii by site-directed mutagenesis of cysteine residues. Eur J Biochem 267:1280–1289
Sun X, Shen X, Jain R, Lin Y, Wang J, Sun J, Yan Y, Yuan Q (2015) Synthesis of chemicals by metabolic engineering of microbes. Chem Soc Rev 44:3760–3785
Suzuki T, Onishi H (1968) Aerobic dissimilation of L-rhamnose and the production of L-rhamnonic acid and 1, 2-propanediol by yeasts. Agric Biol Chem 32:888–893
Totemeyer S, Booth NA, Nichols WW, Dunbar B, Booth IR (1998) From famine to feast: the role of methylglyoxal production in Escherichia coli. Mol Microbiol 27:553–562
Turner KW, Roberton AM (1979) Xylose, arabinose, and rhamnose fermentation by Bacteroides ruminicola. Appl Environ Microbiol 38:7–12
Wang LM, Cai YM, Zhu LF, Guo HL, Yu B (2014) Major role of NAD-dependent lactate dehydrogenases in high optically pure L-lactic acid production by thermophilic Bacillus coagulans. Appl Environ Microbiol 80:7134–7141
Weimer PJ (1984) Fermentation of 6-deoxyhexoses by Bacillus macerans. Appl Environ Microbiol 47:263–267
Yang TH, Kim TW, Kang HO, Lee SH, Lee EJ, Lim SC, Oh SO, Song AJ, Park SJ, Lee SY (2010) Biosynthesis of polylactic acid and its copolymers using evolved propionate CoA transferase and PHA synthase. Biotechnol Bioeng 105:150–160
Yim H, Haselbeck R, Niu W, Pujol-Baxley C, Burgard A, Boldt J, Khandurina J, Trawick JD, Osterhout RE, Stephen R, Estadilla J, Teisan S, Schreyer HB, Andrae S, Yang TH, Lee SY, Burk MJ, Van Dien S (2011) Metabolic engineering of Escherichia coli for direct production of 1,4-butanediol. Nat Chem Biol 7:445–452
Zelcbuch L, Antonovsky N, Bar-Even A, Levin-Karp A, Barenholz U, Dayagi M, Liebermeister W, Flamholz A, Noor E, Amram S, Brandis A, Bareia T, Yofe I, Jubran H, Milo R (2013) Spanning high-dimensional expression space using ribosome-binding site combinatorics. Nucleic Acids Res 41:e98
Acknowledgments
This work was supported by grants from the National Basic Research Program of China (2011CBA00800), the Key Deployment Projects of Chinese Academy of Sciences (KGZD-EW-606), the National Natural Science Foundation of China (21466007), and the Project of Guangxi Provincial Science & Technology Development, China (14125008-2-22). BY is supported by the Youth Innovation Promotion Association, Chinese Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors confirm that principles of ethical and professional conduct have been followed in this research and in the preparation of this manuscript.
Conflict of interest
The authors do not have potential conflict of interest to disclose.
Additional information
Lingfeng Zhu and Xiangchen Guan contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhu, L., Guan, X., Xie, N. et al. Fermentative production of enantiomerically pure S-1,2-propanediol from glucose by engineered E. coli strain. Appl Microbiol Biotechnol 100, 1241–1251 (2016). https://doi.org/10.1007/s00253-015-7034-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-7034-y