Abstract
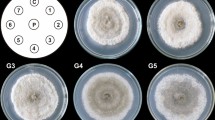
The filamentous fungusCurvularia lunata has enzymatic activities of biotechnological significance. Its 11β-hydroxylase actvity produces hydrocortisone, and its 17-ketosteroid reductase (17β-HSD) activity is important in the production of 17β-hydroxysteroids. We have examined the effects of different media on the 17β-HSD activity, and on the morphology and ultrastructure ofC. lunata in the search for a correlation between high enzymatic activity, morphology and ultrastructure. The highest 17β-HSD activity per dry weight ofC. lunata was seen in Czapek medium, while more product per volume was formed in malt extract broth. In all growth media tested, the highest 17β-HSD activity coincided with the exponential phase of growth. In Czapek broth medium, fungal hyphae formed pellets, while in malt extract and yeast nitrogen base broth media, the fungus formed branched hyphae dispersed throughout the media. Scanning electron microscopy revealed differences in the thickness of hyphal cells; those of the fungus grown in Czapek and yeast nitrogen base media were much thicker than those in malt extract medium. Transmission electron microscopy showed melanized cell walls in the fungus grown in malt extract and Czapek media, but not in yeast nitrogen base medium. Our results show that theC. lunata with the highest 17β-HSD activity grow as dispersed, melanized mycelia with thin hyphae.
Similar content being viewed by others
References
Ashford A.E., Vesk P.A., Orlovich D.A., Markovina A.L., Allaway W.G. (1999). Dispersed polyphosphate in fungal vacuoles inEucalyptus pilularis/Pisolithus tinctorius Ectomycorrhizas. Fungal Genet. Biol., 28: 21–33.
Bode H.B., Bethe B., Hofs R., Zeeck A. (2002). Big effects from small changes: possible ways to explore nature’s chemical divesity. Chembiochem., 3: 619–627.
Brakhage A.A., Langfelder K., Wanner G., Schmidt A., Jahn B. (1999). Pigment biosynthesis and virulence. Contribut. Microbiol., 2: 205–215.
Butler M.J., Day A.W., Henson J.M., Money N.P. (2001). Pathogenic properties of fungal melanins. Mycologia, 93: 1–8.
Butler M.J., Lachance M.A. (1987). Inhibition of melanin synthesis in the black yeastPhaeococcomyces sp. by growth on low pH ascorbate medium: production of spheroplasts from “albinized” cells. Can. J. Microbiol., 33: 184–187.
Carter E., Boudreaux C. (2004). Fatal Cerebral Phaeohyphomycosis due toCurvularia lunata in an immunocompetent patient. J. Clin. Microbiol., 42: 5419–5423.
Dittmann E., Erhard M., Kaebernick M., Scheler C., Neilan B.A., von Dohren H., Borner T. (2001). Altered expression of two light-dependent genes in a microcystin-lacking mutant ofMicrocystis aeruginosa PCC 7806. Microbiology, 147: 3113–3119.
Donova M.V., Egorova O.V., Nikolayeva V.M. (2005). Steroid 17β-reduction by microorganisms — a review. Process Biochem., 40: 2253–2262.
Eagen R., Brisson A., Breuil C. (1997). The sap-staining fungusOphiostoma piceae synthesized different types of melanin in different growth media. Can. J. Microbiol., 43: 592–595.
Fernandez M., Noyola D.E., Rossmann S.N., Edwards M.S. (1999). Cutaneous phaeohyphomycosis caused byCurvularia lunata and a review ofCurvularia infections in pediatrics. Pediatr. Infect. Dis. J., 18: 727–731.
Grimm L.H., Kelly S., Krull E., Hempel D.C. (2005). Morphology and productivity of filamentous fungi. Appl. Microbiol. Biotechnol., 69: 375–384.
Hoerhold C., Undisy K., Groh H., Sahm R., Schade R., Komel R. (1986). Bioconversion of steroids byCochliobolus lunatus. J. Basic Microbiol., 26: 335–339.
Jacobson E.S. (2000). Rathogenic roles for fungal melanins. Clin. Microbiol. Rev., 13: 708–717.
Kennedy M.J., Sandin R.L. (1988). Influence of growth conditions onCandida albicans adhesion hydrophobicity and cell wall ultrastructure. J. Med. Vet. Mycol., 26: 79–92.
Kristan K., Lanišnik Rižner T., Stojan J., Gerber J.K., Kremmer E., Adamski J. (2003). Significance of individual amino acid residues for coenzyme and substrate specificity of 17β-hydroxysteroid dehydrogenase from the fungusCochliobolus lunatus. Chem. Biol. Interact., 143–144: 493–501.
Kristan K., Stojan J., Adamski J., Lanišnik Rižner T. (2007). Rational design of novel mutants of fungal 17β-hydroxysteroid dehydrogenase. J. Biotechnol., 129: 123–130.
Lanišnik Rižner T., Žakelj-Mavrič M., Plemenitaš A., Zorko M. (1996). Purification and characterization of 17β-hydroxysteroid dehydrogenase from the filamentous fungusCochliobolus lunatus. J. Steroid. Biochem. Mol. Biol., 59: 205–214.
Lanišnik Rižner T., Möller G., Thole H.H., Žakelj-Mavrič M., Adamski, J. (1999). A novel 17β-hydroxysteroid dehydrogenase in the fungusCochliobolus lunatus: new insights into the evolution of steroid-hormone signalling. Biochem. J., 337: 425–431.
Lanišnik Rižner T., Adamski J., Stojan J. (2000). 17β-hydroxysteroid dehydrogenase fromCochliobolus lunatus: model structure and substrate specificity. Archiv. Biochem. Biophys., 384: 255–262.
Lanišnik Rižner T., Stojan J., Adamski J. (2001a). Searching for the physiological function of 17β-hydroxysteroid dehydrogenase from the fungusCochliobolus lunatus: studies of substrate specificity and expression analysis. Mol. Cell. Endocrinol., 171: 193–198.
Lanišnik Rižner T, Stojan J., Adamski J. (2001b). 17β-hydroxysteroid dehydrogenase from the fungusCochliobolus lunatus: structural and functional aspects. Chem. Biol. Interact., 130–132: 793–803.
Lanišnik Rižner T., Wheeler M.H. (2003). Melanin biosynthesis in the fungusCurvularia lunata (teleomorph:Cochliobolus lunatus). Can. J. Microbiol., 49: 110–119.
Martin J.F., Demain A.L. (1980). Control of antibiotic biosynthesis. Microbiol. Rev., 44: 230–251.
Ohsumi Y., Kitamoto K., Anraku Y. (1988). Changes induced in the permeability barrier of the yeast plasma membrane by cupric ion. J. Bacteriol., 170: 2676–2682.
Paraszkiewitcz K., Kanwal A., Dlugonski J. (2002). Emulsifier production by steroid transforming filamentous fungusCurvularia lunata. Growth and product characterization. J. Biotechnol., 92: 287–294.
Plemenitaš A., Žakelj-Mavrič M., Komel R. (1988). Hydroxysteroid dehydrogenase ofCochliobolus lunatus. J. Steroid Biochem., 29: 371–372.
Polak A. (1989). Melanin as a virulence factor in pathogenic fungi. Mycoses, 33: 215–224.
Prom L., Waniska R., Kollo A., Rooney W. (2003). Response of eight sorghum cultivars inoculated withFusarium thapsinum, Curvularia lunata and mixture of the two fungi. Crop Protection, 22: 623–628.
Revankar S.G., Patterson J.E., Sutton D.A., Pullen R., Rinaldi M.G. (2002). Disseminated phaeohyphomycosis: Review of an emerging mycosis. Clin. Infect. Dis., 34: 467–476.
Rinaldi M.G., Phillips P., Schwartz J.G., Winn R.E., Holt G.R., Shagets F.W., Elrod J., Nishioka G., Aufdemorte T.b. (1987). HumanCurvularia infections. Report of five cases and review of the literature. Diagn. Microbiol. Infect. Dis., 6: 27–39.
Rozman D., Komel R. (1994). Isolation of genomic DNA from filamentous fungi with high glucan level. BioTechniques, 16: 382–384.
Rozman D., Jezernik K., Komel R. (1994). Ultrastructure and genotypic characterization of the filamentous fungusCochliobolus lunatus in comparioson to the anamorphic strainCurvularia lunata. FEMS Microbiol. Lett., 117: 35–40.
Rozman D., Hennebert G.L., Kunej T., Deckock C., Komel R. (1996). Steroid biotransforming strains designatedCochliobolus lunatus m118 andCuvularia lunata AT46 are bothCurvularia lunata var.lunata. Mycotaxon, LIX: 489–498.
Saito K., Kuga-Uetake Y., Saito M., Peterson R.L. (2006). Vacuolar localization of phosphorus in hyphae ofPhialocephala fortinii, a dark septate fungal root endophyte. Can. J. Microbiol., 52: 643–650.
Sonomoto K., Hoq M.M., Tanaka, A., Fukui S. (1983). 11β-hydroxylation of cortexolone (Reichstein compound S) to hydrocortisone byCurvularia lunata entrapped in photocross-linked resin gels. Appl. Environ. Microbiol., 45: 436–443.
Vitas M., Smith K., Rozman D., Komel R. (1994). Progesterone metabolism by the filamentous fungusCochliobolus lunatus. J. Steroid. Biochem. Mol. Biol., 49: 87–92.
Vitas M., Smith K.E., Plavec J., Kesselmeier J., Pajič T., Žigon, R.D., Kelly S.L., Komel R. (1999). Induction of steroidal hydroxylase activity by plant defense compound in the filamentous fungusCochliobolus lunatus. Chemosphere, 38: 853–863.
Zahraee M.H., Wilson T.E., Scheer E.R. (1982). The effect of growth media upon the ultrastructure ofBlastomyces dermatitidis. Can. J. Microbiol., 28: 211–218.
Žnidaršic P., Pavko A., Komel R. (1992). Laboratory-scale biosynthesis ofTrichoderma mycolytic enzymes for protoplast release fromCochliobolus lunatus. J. Ind. Microbiol., 9: 115–119.
Žakelj-Mavrič M., Plemenitaš A., Komel R., Belič I. (1990). 11β-hydroxylation of steroids byCochliobolus lunatus. J. Steroid. Biochem., 35: 627–629.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lanišnik Rižner, T., Romih, R. Growth media effects on morphology and 17β-HSD activity in the fungusCurvularia lunata . Ann. Microbiol. 57, 635–643 (2007). https://doi.org/10.1007/BF03175366
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03175366