Abstract
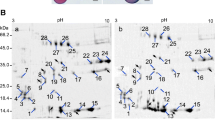
The identification of proteins involved in pollen germination and tube growth is important for fundamental studies of fertility and reproduction in flowering plants. We used 2-DE and MALDI-TOF-MS to identify differentially expressed proteins in mature (P0) and 1-h germinated (P1) maize pollen. Among about 470 proteins separated in 2D gels, the abundances of 26 protein spots changed (up- or down-regulation) between P0 and P1. The 13 up-regulated protein spots were mainly involved in tube wall modification, actin cytoskeleton organization, energy metabolism, signaling, protein folding and degradation. In particular, pectin methylesterase, inorganic pyrophosphatase, glucose-1-phosphate uridylyltransferase and rab GDP dissociation inhibitor α are highly up-regulated, suggesting their potential role in pollen tube growth. The down-regulated 13 protein spots mainly include defense-related proteins, pollen allergens and some metabolic enzymes. This study would contribute to the understanding of the changes in protein expression associated with pollen tube development.


Similar content being viewed by others
Abbreviations
- 2D:
-
Two dimensional
- 2-DE:
-
Two-dimensional electrophoresis
- ADK:
-
Adenosine kinase
- MPP:
-
Mitochondrial-processing peptidase
- MALDI-TOF:
-
Matrix-assisted laser desorption/ionization time-of-flight
- Mr:
-
Molecular weight
- MS:
-
Mass spectrometry
- P0:
-
Mature pollen
- P1:
-
Pollen germinated for 1 h
- pI:
-
Isoelectric point
- PMEs:
-
Pectin methylesterases
- rab-GDI:
-
rab GDP dissociation inhibitor
References
Barnabas B, Fridvalszky L (1984) Adhesion and germination of differently treated maize pollen grains on the stigma. Acta Bot Hung 30:329–332
Bosch M, Hepler PK (2005) Pectin methylesterases and pectin dynamics in pollen tubes. Plant Cell 17:3219–3226
Bosch M, Cheung AY, Hepler PK (2005) Pectin methylesterase, a regulator of pollen tube growth. Plant Physiol 138:1334–1346
Cai G, Cresti M (2009) Organelle motility in the pollen tube: a tale of 20 years. J Exp Bot 60:495–508
Cardenas L, Lovy-Wheeler A, Wilsen KL, Hepler PK (2005) Actin polymerization promotes the reversal of streaming in the apex of pollen tubes. Cell Motil Cytoskel 61:112–127
Chen YM, Chen T, Shen SH, Zheng MZ, Guo Y, Lin J, Baluska F, Samaj J (2006) Differential display proteomic analysis of Picea meyeri pollen germination and pollen-tube growth after inhibition of actin polymerization by latrunculin B. Plant J 47:174–195
Cosgrove DJ, Bedinger P, Durachko DM (1997) Group I allergens of grass pollen as cell wall-loosening agents. Proc Natl Acad Sci USA 94:6559–6564
Dai S, Li L, Chen T, Chong K, Xue Y, Wang T (2006) Proteomic analysis of Oryza sativa mature pollen reveal novel proteins associated with pollen germination and tube growth. Proteomics 6:2504–2529
Dai S, Chen T, Chong K, Xue Y, Liu S, Wang T (2007) Proteomics identification of differentially expressed proteins associated with pollen germination and tube growth reveals characteristics of germinated Oryza sativa pollen. Mol Cell Proteomics 6:207–230
Feijo JA, Costa SS, Prado AM, Becker JD, Certal AC (2004) Signalling by tips. Curr Opin Plant Biol l7:589–598
Fernando DD (2005) Characterization of pollen tube development in eastern white pine (Pinus strobus) through proteomic analysis of differentially expressed proteins. Proteomics 5:4917–4926
Gakh O, Cavadini P, Isaya G (2002) Mitochondrial processing peptidases. Biochem Biophys Acta 1592:63–77
Geitmann A, Snowman BN, Emons AMC, Franklin-Tong VE (2000) Alterations in the actin cytoskeleton of pollen tubes are induced by the self-incompatibility reaction in Papaver rhoeas. Plant Cell 12:1239–1252
Golovkin M, Reddy ASN (2003) A calmodulin-binding protein from Arabidopsis has an essential role in pollen germination. Proc Natl Acad Sci USA 100:10563–10588
Grobei MN, Qeli E, Brunner E, Rehrauer H, Zhang RX, Roschitzki B, Basler K, Ahrens CH, Grossniklaus U (2009) Deterministic protein inference for shotgun proteomics data provides new insights into Arabidopsis pollen development and function. Genome Res 19:1786–1800
Holmes-Davis R, Tanaka CK, Vensel WH, Hurkman WJ, McCormick S (2005) Proteome mapping of mature pollen of Arabidopsis thaliana. Proteomics 5:4864–4884
Imin N, Kerim T, Rolfe BG, Weinman JJ (2004) Effect of early cold stress on the maturation of rice anthers. Proteomics 4:1873–1882
Krichevsky A, Kozlovsky SV, Tian GW, Chen MH, Zaltsman A, Citovsky V (2007) How pollen tubes grow. Dev Biol 303:405–420
Li LC, Bedinger PA, Volk C, Jones D, Cosgrove DJ (2003) Purification and characterization of four β-expansins (Zea m 1 isoforms) from maize pollen. Plant Physiol 132:2073–2085
Lockhart DJ, Winzeler EA (2000) Genomics, gene expression and DNA arrays. Nature 405:827–836
Malho R, Camacho L, Moutinho A (2006) Signaling pathways in pollen tube growth and reorientation. Ann Bot 85:59–68
Moffatt BA, Wang L, Allen MS, Stevens YY, Qin W, Snider J, von Schwartzenberg K (2000) Adenosine kinase of Arabidopsis. Kinetic properties and gene expression. Plant Physiol 124:1775–1785
Noir S, Brautigam A, Colby T, Schmidt J, Panstruga R (2005) A reference map of the Arabidopsis thaliana mature pollen proteome. Biochem Biophys Res Commun 337:1257–1266
Pertl H, Schulze WX, Obermeyer G (2009) The pollen organelle membrane proteome reveals highly spatial–temporal dynamics during germination and tube growth of lily pollen. J Proteome Res 8:5142–5152
Petersen A, Dresselhaus T, Grobe K, Becker WM (2006) Proteome analysis of maize pollen for allergy-relevant components. Proteomics 6:6317–6325
Pomés A (2008) Allergen structures and biologic functions: the cutting edge of allergy research. Curr Allergy Asthm Rep 8:425–432
Sagi G, Katz A, Guenoune-Gelbart D, Epel BL (2005) Class 1 reversibly glycosylated polypeptides are plasmodesmal-associated proteins delivered to plasmodesmata via the Golgi apparatus. Plant Cell 17:1788–1800
Sheoran IS, Sproule KA, Olson DJH, Ross ARS, Sawhney VK (2006) Proteome profile and functional classification of proteins in Arabidopsis thaliana (Landsberg erecta) mature pollen. Sex Plant Reprod 19:185–196
Sheoran IS, Ross AR, Olson DJ, Sawhney VK (2007) Proteomic analysis of tomato (Lycopersicon esculentum) pollen. J Exp Bot 58:3525–3535
Sheoran IS, Pedersen EJ, Ross AR, Sawhney VK (2009) Dynamics of protein expression during pollen germination in canola (Brassica napus). Planta 230:779–793
Suen DF, Huang AHC (2007) Maize pollen coat xylanase facilitates pollen tube penetration into silk during sexual reproduction. J Biol Chem 282:625–636
Szumlanski AL, Nielsen E (2009) The rab GTPase rabA4D regulates pollen tube tip growth in Arabidopsis thaliana. Plant Cell 21:526–544
Vidali L, McKenna ST, Hepler PK (2001) Actin polymerization is essential for pollen tube growth. Mol Biol Cell 12:2534–2545
Wang W, Vignani R, Scali M, Cresti M (2004a) Post-translational modifications of α-tubulin in Zea mays are highly tissue specific. Planta 218:460–465
Wang W, Scali M, Vignani R, Milanesi C, Petersen A, Sari-Gorla M, Cresti M (2004b) Male-sterile mutation alters Zea m 1 (β-expansin 1) accumulation in a maize mutant. Sex Plant Reprod 17:41–47
Wang W, Vignani R, Scali M, Cresti M (2006) A universal and rapid protocol for protein extraction from recalcitrant plant tissues for proteomic analysis. Electrophoresis 27:2782–2786
Wang Y, Zhang WZ, Song LF, Zou JJ, Su Z, Wu WH (2008) Transcriptome analyses show changes in gene expression to accompany pollen germination and tube growth in Arabidopsis. Plant Physiol 148:1201–1211
Wang W, Bianchi L, Scali M, Liu L, Bini L, Cresti M (2009) Proteomic analysis of β-1,3-glucanase in grape berry tissues. Acta Physiol Plant 31:597–604
Wilkins MR, Pasquali C, Appel RD, Ou K, Golaz O, Sanchez JC, Yan JX, Gooley AA, Hughes G, Humphery-Smith I, Williams KL, Hochstrasser DF (1996) From proteins to proteomes: large scale protein identification by two-dimensional electrophoresis and amino acid analysis. Nature Biotech 14:61–65
Wittink FRA, Knuiman B, Derksen J, Čapková V, Twell D, Schrauwen JAM, Wullems GJ (2000) The pollen-specific gene Ntp303 encodes a 69-kDa glycoprotein associated with the vegetative membranes and the cell wall. Sex Plant Reprod 12:276–284
Yang Z (2002) Small GTPases: versatile signaling switches in plants. Plant Cell 14:S375–S388
Zou JJ, Song LF, Zhang WZ, Wang Y, Ruan S, Wu WH (2009) Comparative proteomic analysis of Arabidopsis mature pollen and germinated pollen. J Integr Plant Biol 51:438–455
Acknowledgments
This work was supported by the National Natural Science Foundation of China (project 30971705) by the Program for Science and Technology Innovation Talents in Universities of Henan Province (project 2008HASTIT005) and by Innovation Scientists and Technicians Troop Construction Projects of Henan Province (project 9410051003).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Z.-L. Zhang.
The authors Y. Zhu and P. Zhao contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhu, Y., Zhao, P., Wu, X. et al. Proteomic identification of differentially expressed proteins in mature and germinated maize pollen. Acta Physiol Plant 33, 1467–1474 (2011). https://doi.org/10.1007/s11738-010-0683-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-010-0683-7