Abstract
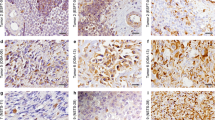
Establishment of new animal models using selected cell lines with different behaviour is very important for cancer investigations. In this study, we describe three morphologically distinct rat sarcoma clones—C4, C7 and D6—isolated from the R5-28 cell line. Cells of all clones expressed vimentin, fibronectin, laminin, collagen IV and matrix metalloproteinases 2 and 9. However, desmin, cytokeratins 8 and 18, ZO-1 and desmoplakins I and II were not detected. Significant proliferative capacity was documented by proliferating cell nuclear antigen expression and BrdU positivity. Karyotype of the C4, C7 and D6 cells greatly differed from diploid chromosome number of normal rat somatic cells. High expression of three cytokines—monocyte chemoattractant protein 1, tissue inhibitor of metalloproteinases 1 and vascular endothelial growth factor—was observed in all three clones. However, they varied in concentration of chemokines associated with neutrophil migration and activation—cytokine induced neutrophil chemoattractant 2 and lipopolysaccharide induced CXC chemokine. The C4 clone showed spontaneous tumour regression in vivo that was associated with significant changes in lymphocyte subpopulations.






Similar content being viewed by others
References
Akahane T.; Akahane M.; Shah A.; Connor C. M.; Thorgeirsson U. P. TIMP-1 inhibits microvascular endothelial cell migration by MMP-dependent and MMP-independent mechanisms. Exp Cell Res 301: 158–167; 2004.
Balamayooran G.; Batra S.; Balamayooran T.; Cai S.; Jeyaseelan S. Monocyte chemoattractant protein 1 regulates pulmonary host defense via neutrophil recruitment during Escherichia coli infection. Infect Immun 79: 2567–2577; 2011.
Barnetson R. S.; Halliday G. M. Regression in skin tumours: a common phenomenon. Australas J Dermatol 38(Suppl.1): S63–S65; 1997.
Carr M. W.; Roth S. J.; Luther E.; Rose S. S.; Springer T. A. Monocyte chemoattractant protein 1 acts as a T-lymphocyte chemoattractant. Proc Natl Acad Sci USA 91: 3652–3656; 1994.
Choong M. L.; Yong Y. P.; Tan A. C.; Luo B.; Lodish H. F. LIX: a chemokine with a role in hematopoietic stem cells maintenance. Cytokine 25: 239–245; 2004.
Connolly D. C. Animal models of ovarian cancer. Cancer Treat Res 149: 353–391; 2009.
DeClerck Y. A.; Mercurio A. M.; Stack M. S.; Chapman H. A.; Zutter M. M.; Muschel R. J.; Raz A.; Matrisian L. M.; Sloane B. F.; Noel A.; Hendrix M. J.; Coussens L.; Padarathsingh M. Proteases, extracellular matrix, and cancer: a workshop of the path B study section. Am J Pathol 164: 1131–1139; 2004.
Dodd R. D.; Mito J. K.; Kirsch D. G. Animal models of soft-tissue sarcoma. Dis Model Mech 3: 557–566; 2010. doi:10.1242/dmm.005223.
Euhus D. M.; Hudd C.; LaRegina M. C.; Johnson F. E. Tumor measurement in the nude mouse. J Surg Oncol 31: 229–234; 1986.
Ferrara N.; Gerber H. P.; LeCouter J. The biology of VEGF and its receptors. Nat Med 9: 669–676; 2003.
Freshney R. I. Culture of animal cells: a manual of basic technique. 5th ed. Wiley-Liss, Hoboken; 2005.
Halliday G. M.; Patel A.; Hunt M. J.; Tefany F. J.; Barnetson R. S. Spontaneous regression of human melanoma/nonmelanoma skin cancer: association with infiltrating CD4+ T cells. World J Surg 19: 352–358; 1995.
Horák V.; Fléchon J. E. Immunocytochemical characterisation of rabbit and mouse embryonic fibroblasts. Reprod Nutr Dev 38: 683–695; 1998.
Ikenaka Y.; Yoshiji H.; Kuriyama S.; Yoshii J.; Noguchi R.; Tsujinoue H.; Yanase K.; Namisaki T.; Imazu H.; Masaki T.; Fukui H. Tissue inhibitor of metalloproteinase-1 (TIMP-1) inhibits tumor growth and angiogenesis in the TIMP-1 transgenic mouse model. Int J Cancer 105: 340–346; 2003.
Inoue T.; Yoneda K.; Manabe M.; Demitsu T. Spontaneous regression of Merkel cell carcinoma: a comparative study of TUNEL index and tumor-infiltrating lymphocytes between spontaneous regression and non-regression group. J Dermatol Sci 24: 203–211; 2000.
Jaganjac M.; Poljak-Blazi M.; Zarkovic K.; Schaur R. J.; Zarkovic N. The involvement of granulocytes in spontaneous regression of Walker 256 carcinoma. Cancer Lett 260: 180–186; 2008.
Jain S.; Xu R.; Prieto V. G.; Lee P. Molecular classification of soft tissue sarcomas and its clinical applications. Int J Clin Exp Pathol 3: 416–428; 2010.
Kalialis L. V.; Drzewiecki K. T.; Klyver H. Spontaneous regression of metastases from melanoma: review of the literature. Melanoma Res 19: 275–282; 2009.
Kappler M.; Bache M.; Bartel F.; Kotzsch M.; Panian M.; Würl P.; Blümke K.; Schmidt H.; Meye A.; Taubert H. Knockdown of survivin expression by small interfering RNA reduces the clonogenic survival of human sarcoma cell lines independently of p53. Cancer Gene Ther 11: 186–193; 2004.
Ladanyi M.; Bridge J. A. Contribution of molecular genetic data to the classification of sarcomas. Hum Pathol 31: 532–538; 2000.
Lazar A.; Abruzzo L. V.; Pollock R. E.; Lee S.; Czerniak B. Molecular diagnosis of sarcomas: chromosomal translocations in sarcomas. Arch Pathol Lab Med 130: 1199–1207; 2006.
Leader M.; Collins M.; Patel J.; Henry K. Vimentin: an evaluation of its role as a tumour marker. Histopathology 11: 63–72; 1987.
Mackall C. L.; Meltzer P. S.; Helman L. J. Focus on sarcomas. Cancer Cell 2: 175–178; 2002.
Mantovani A. The ying-yang of tumor-associated neutrophils. Cancer Cell 16: 173–174; 2009.
Maquoi E.; Frankenne F.; Noël A.; Krell H. W.; Grams F.; Foidart J. M. Type IV collagen induces matrix metalloproteinase 2 activation in HT1080 fibrosarcoma cells. Exp Cell Res 261: 348–359; 2000.
Misra A.; Mistry N.; Grimer R.; Peart F. The management of soft tissue sarcoma. J Plast Reconstr Aesthet Surg 62: 161–174; 2009.
Morávková A.; Málek O.; Pokorná E.; Strnádel J.; Hradecký J.; Horák V. Immune characterization of the Lewis rats inoculated with K2 sarcoma cell line and newly derived R5-28 malignant cells. Folia Biol (Praha) 51: 159–165; 2005.
Ohnstad H. O.; Paulsen E. B.; Noordhuis P.; Berg M.; Lothe R. A.; Vassilev L. T.; Myklebost O. MDM2 antagonist Nutlin-3a potentiates antitumour activity of cytotoxic drugs in sarcoma cell lines. BMC Cancer 11(211): 1–11; 2011. doi:10.1186/1471-2407-11-211.
Ottaviano L.; Schaefer K. L.; Gajewski M.; Huckenbeck W.; Baldus S.; Rogel U.; Mackintosh C.; de Alava E.; Myklebost O.; Kresse S. H.; Meza-Zepeda L. A.; Serra M.; Cleton-Jansen A. M.; Hogendoorn P. C.; Buerger H.; Aigner T.; Gabbert H. E.; Poremba C. Molecular characterization of commonly used cell lines for bone tumor research: a trans-European EuroBoNet effort. Genes Chromosomes Cancer 49: 40–51; 2010.
Pattarroyo M.; Tryggvason K.; Virtanen I. Laminin isoforms in tumor invasion, angiogenesis and metastasis. Semin Cancer Biol 12: 197–207; 2002.
Penichet M. L.; Dela Cruz J. S.; Challita-Eid P. M.; Rosenblatt J. D.; Morrison S. L. A murine B cell lymphoma expressing human HER2/neu undergoes spontaneous tumor regression and elicits antitumor immunity. Cancer Immunol Immunother 49: 649–662; 2001.
Piantino C. B.; Sousa-Canavez J. M.; Srougi V.; Salvadori F.; Kato R.; Ayres P. P.; Srougi M.; Camara-Lopes L. H.; Gattás G. J.; Fridman C.; de Toledo F.; Santana I.; Leite K. R. Establishment and characterization of human bladder cancer cell lines BexBra1, BexBra2, and BexBra4. In Vitro Cell Dev Biol Anim 46: 131–139; 2010.
Pokorná E.; Zicha D.; Chaloupková A.; Matoušková E.; Veselý P. Two dynamic morphotypes of sarcoma cells, asymmetric stellate and triangle with leading lamela, are related to malignancy. Folia Biol (Praha) 49: 33–39; 2003.
Sabeh F.; Shimizu-Hirota R.; Weiss S. J. Protease-dependent versus -independent cancer cell invasion programs: three-dimensional amoeboid movement revisited. J Cell Biol 185: 11–19; 2009.
Shor A. C.; Keschman E. A.; Lee F. Y.; Muro-Cacho C.; Letson G. D.; Trent J. C.; Pledger W. J.; Jove R. Dasatinib inhibits migration and invasion in diverse human sarcoma cell lines and induces apoptosis in bone sarcoma cells dependent on SRC kinase for survival. Cancer Res 67: 2800–2808; 2007.
Smith J. B.; Herschman H. R. Glucocorticoid-attenuated response genes encode intercellular mediators, including a new C-X-C chemokine. J Biol Chem 270: 16756–16765; 1995.
Stamenkovic I. Matrix metalloproteinases in tumor invasion and metastases. Semin Cancer Biol 10: 415–433; 2000.
Strieter R. M.; Wiggins R.; Phan S. H.; Wharram B. L.; Showell H. J.; Remick D. G.; Chensue S. W.; Kunkel S. L. Monocyte chemotactic protein gene expression by cytokine-treated human fibroblasts and endothelial cells. Biochem Biophys Res Commun 162: 694–700; 1989.
Strnádel J.; Kverka M.; Horák V.; Vannucci L.; Usvald D.; Hlučilová J.; Plánská D.; Vána P.; Reisnerová H.; Jílek F. Multiplex analysis of cytokines in rat sarcoma model. Folia Biol (Praha) 53: 216–219; 2007.
Taketo M. M. Mouse models of gastrointestinal tumors. Cancer Sci 97: 355–361; 2006.
Tammela T.; Enholm B.; Alitalo K.; Paavonen K. The biology of vascular endothelial growth factors. Cardiovasc Res 65: 550–563; 2005.
Tanjore H.; Kalluri R. The role of type IV collagen and basement membranes in cancer progression and metastasis. Am J Pathol 168: 715–717; 2006.
Thomas J. A.; Badini M. The role of innate immunity in spontaneous regression of cancer. Indian J Cancer 48: 246–251; 2011.
Uematsu K.; Takahara T.; Seki N.; Seto T.; Ota S.; Nakamura M.; Eguchi K. Spontaneous regression of a primary sarcoma of the pulmonary artery. Respirat Med Extra 3: 6–8; 2007.
Vesely M. J.; Murray D. J.; Neligan P. C.; Novak C. B.; Gullane P. J.; Ghazarian D. Complete spontaneous regression in Merkel cell carcinoma. J Plast Reconstr Aesthet Surg 61: 165–171; 2008.
Watanabe K.; Koizumi F.; Kurashige Y.; Tsurufuji S.; Nakagawa H. Rat CINC, a member of the interleukin-8 family, is a neutrophil-specific chemoattractant in vivo. Exp Mol Pathol 55: 30–37; 1991.
Wibmer C.; Leithner A.; Zielonke N.; Sperl M.; Windhager R. Increasing incidence rates of soft tissue sarcomas? A population-based epidemiologic study and literature review. Ann Oncol 21: 1106–1111; 2010.
Yoo J. K.; Kwon H.; Khil L. Y.; Zhang L.; Jun H. S.; Yoon J. W. IL-18 induces monocyte chemotactic protein-1 production in macrophages through the phosphatidylinositol 3-kinase/Akt and MEK/ERK1/2 pathways. J Immunol 175: 8280–8286; 2005.
Zachriae C. O.; Anderson A. O.; Thompson H. L.; Appella E.; Mantovani A.; Oppenheim J. J.; Matsushima K. Properties of monocyte chemotactic and activating factor (MCAF) purified from a human fibrosarcoma cell line. J Exp Med 171: 2177–2182; 1990.
Acknowledgements
This work was supported by the MEYS CR (grant no.2B08063) and by the IAPG AS CR, v.v.i. (RVO: 67985904). We thank Jaroslava Sestakova and Jitka Klucinova for excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: J. Denry Sato
Rights and permissions
About this article
Cite this article
Holubova, M., Leba, M., Sedmikova, M. et al. Characterization of three newly established rat sarcoma cell clones. In Vitro Cell.Dev.Biol.-Animal 48, 610–618 (2012). https://doi.org/10.1007/s11626-012-9563-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-012-9563-3