Abstract
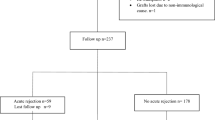
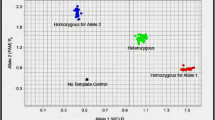
Glutathione-S-transferase (GST) has a major protective role against free radicals and plays a vital role in phase II of biotransformation of many substances. In liver transplantation, reperfusion injury, calcineurin drug consumption and infection produce free radicals that cause tissue injury and organ damage. Genetic variations of GST may influence individual susceptibility to some diseases associated with the deleterious effects of oxidative metabolism. Although it is well known that the rejection is an immunological process, however, in this study, we have investigated the gene frequency and relationship between human GST gene polymorphism and rejection in liver transplant recipients. We have assessed 51 liver transplant recipients from Shiraz, South of Iran. The GSTT1 and GSTM1 genotypes were identified by polymerase chain reaction (PCR). The gene frequency of GSTM1 and GSTT1 polymorphism were evaluated. We observed that GSTM1 null genotype was present in 68.62% of the liver transplant recipients while GSTT1 null genotype was present in 37.25% of the liver transplant subjects. There was a trend between increasing age and acute rejection episode. No statistically significant correlation was present between GSTM1 null and GSTT1 null genotypes with an acute rejection episode in transplant recipients. No relationship was observed between GST genotypes and acute rejection. It is likely that development and progression of rejection are determined by genes which is involved in immunological pathways rather than genes that is participated in free radicals destruction. However, these findings need to be confirmed in a larger series of patients.
Similar content being viewed by others
References
Onaran I, Güven G, Ozaydin A, Ulutin T (2001) The influence of GSTM1 null genotype on susceptibility to in vitro oxidative stress. Toxicology 157(3):195–205. doi:10.1016/S0300-483X(00)00358-9
Vázquez-Medina JP, Zenteno-Savín T, Elsner R (2006) Antioxidant enzymes in ringed seal tissues: potential protection against dive-associated ischemia/reperfusion. Comp Biochem Physiol C Toxicol Pharmacol 142(3–4):198–204. doi:10.1016/j.cbpc.2005.09.004
Perrea DN, Moulakakis KG, Poulakou MV, Vlachos IS, Papachristodoulou A, Kostakis AI (2006) Correlation between oxidative stress and immunosuppressive therapy in renal transplant recipients with an uneventful postoperative course and stable renal function. Int Urol Nephrol 38(2):343–348. doi:10.1007/s11255-006-0054-x
de Cal M, Silva S, Cruz D, Basso F, Corradi V, Lentini P, Nalesso F, Dissegna D, Goepel V, Chiaramonte S, Ronco C (2008) Oxidative stress and ‘monocyte reprogramming’ after kidney transplant: a longitudinal study. Blood Purif 26(1):105–110. doi:10.1159/000110575
Wierzbicka A, Pawłowska J, Socha P, Jankowska I, Skorupa E, Teisseyre M, Ismail H, Czubkowski P, Socha J (2007) Lipid, carbohydrate metabolism, and antioxidant status in children after liver transplantation. Transplant Proc 39(5):1523–1525. doi:10.1016/j.transproceed.2007.01.084
Strange RC, Spiteri MA, Ramachandran S, Fryer AA (2001) Glutathione-S-transferase family of enzymes. Mutat Res 482:21–26. doi:10.1016/S0027-5107(01)00206-8
Rocha V, Porcher R, Fernandes JF, Filion A, Bittencourt H, Silva W Jr, Vilela G, Zanette DL, Ferry C, Larghero J, Devergie A, Ribaud P, Skvortsova Y, Tamouza R, Gluckman E, Socie G, Zago MA (2008) Association of drug metabolism gene polymorphisms with toxicities, graft-versus-host disease and survival after HLA-identical sibling hematopoietic stem cell transplantation for patients with leukemia. Leukemia. doi:10.1038/leu.2008.323
Banff Working Group, Demetris AJ, Adeyi O, Bellamy CO, Clouston A, Charlotte F, Czaja A, Daskal I, El-Monayeri MS, Fontes P, Fung J, Gridelli B, Guido M, Haga H, Hart J, Honsova E, Hubscher S, Itoh T, Jhala N, Jungmann P, Khettry U, Lassman C, Ligato S, Lunz JGIII, Marcos A, Minervini MI, Mölne J, Nalesnik M, Nasser I, Neil D, Ochoa E, Pappo O, Randhawa P, Reinholt FP, Ruiz P, Sebagh M, Spada M, Sonzogni A, Tsamandas AC, Wernerson A, Wu T, Yilmaz F (2006) Liver biopsy interpretation for causes of late liver allograft dysfunction. Hepatology 44(2):489–501. doi:10.1002/hep.21280
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16(3):1215. doi:10.1093/nar/16.3.1215
Arand M, Mühlbauer R, Hengstler J, Jäger E, Fuchs J, Winkler L, Oesch F (1996) A multiplex polymerase chain reaction protocol for the simultaneous analysis of the glutathione S-transferase GSTM1 and GSTT1 polymorphisms. Anal Biochem 236:184–186. doi:10.1006/abio.1996.0153
Muro M, Rojas G, Botella C, Miras M, Campillo JA, Minguela A, Sánchez-Bueno F, Bermejo J, Ramírez P, Alvarez-López MR (2008) CT60 A/G marker of the 3′-UTR of the CTLA4 gene and liver transplant. Transpl Immunol 18(3):246–249. doi:10.1016/j.trim.2007.07.003
Tapirdamaz O, Pravica V, Metselaar HJ, Hansen B, Moons L, van Meurs JB, Hutchinson IV, Shaw J, Agarwal K, Adams DH, Day CP, Kwekkeboom J (2006) Polymorphisms in the T cell regulatory gene cytotoxic T lymphocyte antigen 4 influence the rate of acute rejection after liver transplantation. Gut 55(6):863–868. doi:10.1136/gut.2005.080937
Warlé MC, Metselaar HJ, Hop WC, Tilanus HW (2005) Cytokine gene polymorphisms and acute liver graft rejection: a meta-analysis. Liver Transpl 11(1):19–26. doi:10.1002/lt.20316
Boyer TD (1989) The glutathione S-transferases: an update. Hepatology 9:486–496. doi:10.1002/hep.1840090324
Tocher DR, Leaver MJ, Hodgson PA (1998) Recent advances in the biochemistry and molecular biology of fatty acyl desaturases. Prog Lipid Res 37:73–117. doi:10.1016/S0163-7827(98)00005-8
Landi S (2000) Mammalian class theta GST and differential susceptibility to carcinogens: a review. Mutat Res 463:247–283. doi:10.1016/S1383-5742(00)00050-8
Agrawal S, Tripathi G, Khan F, Sharma R, Baburaj VP (2007) Relationship between GSTs gene polymorphism and susceptibility to end stage renal disease among north Indians. Ren Fail 29(8):947–953. doi:10.1080/08860220701641314
Pagliuso RG, Abbud-Filho M, Alvarenga MP, Ferreira-Baptista MA, Biselli JM, Biselli PM, Goloni-Bertollo EM, Pavarino-Bertelli EC (2008) Role of glutathione S-transferase polymorphisms and chronic allograft dysfunction. Transplant Proc 40(3):743–745. doi:10.1016/j.transproceed.2008.03.008
Aguilera I, Sousa JM, Gavilan F, Bernardos A, Wichmann I, Nuñez-Roldan A (2005) Glutathione S-transferase T1 genetic mismatch is a risk factor for de novo immune hepatitis in liver transplantation. Transplant Proc 37(9):3968–3969. doi:10.1016/j.transproceed.2005.10.042
Rodriguez-Mahou M, Salcedo M, Fernandez-Cruz E, Tiscar JL, Bañares R, Clemente G, Vicario JL, Alvarez E, Rodriguez-Sainz C (2007) Antibodies against glutathione S-transferase T1 (GSTT1) in patients with GSTT1 null genotype as prognostic marker: long-term follow-up after liver transplantation. Transplantation 83(8):1126–1129. doi:10.1097/01.tp.0000259963.47350.da
Aguilera I, Sousa JM, Gavilán F, Bernardos A, Wichmann I, Nuñez-Roldán A (2004) Glutathione S-transferase T1 mismatch constitutes a risk factor for de novo immune hepatitis after liver transplantation. Liver Transpl 10(9):1166–1172. doi:10.1002/lt.20209
Aguilera I, Alvarez-Marquez A, Gentil MA, Fernandez-Alonso J, Fijo J, Saez C, Wichmann I, Nuñez-Roldan A (2008) Anti-glutathione S-transferase T1 antibody-mediated rejection in C4d-positive renal allograft recipients. Nephrol Dial Transplant 23(7):2393–2398
Tripathi S, Ghoshal U, Ghoshal UC, Mittal B, Krishnani N, Chourasia D, Agarwal AK, Singh K (2008) Gastric carcinogenesis: possible role of polymorphisms of GSTM1, GSTT1, and GSTP1 genes. Scand J Gastroenterol 43(4):431–439. doi:10.1080/00365520701742930
Oniki K, Hori M, Takata K, Yokoyama T, Mihara S, Marubayashi T, Nakagawa K (2008) Association between glutathione S-transferase A1, M1 and T1 polymorphisms and hypertension. Pharmacogenet Genomics 18(3):275–277. doi:10.1097/FPC.0b013e3282f56176
Tkacova R, Salagovic J, Ceripkova M, Tkac I, Stubna J, Kalina I (2004) Glutathione S-transferase M1 gene polymorphism is related to COPD in patients with non-small-cell lung cancer. Wien Klin Wochenschr 116:131–134
Sekine Y, Hommura S, Harada S (1995) Frequency of glutathione-Stransferase 1 gene deletion and its possible correlation with cataract formation. Exp Eye Res 60:159–163. doi:10.1016/S0014-4835(95)80006-9
Oz O, Aras Ates N, Tamer L, Yildirim O, Adiguzel U (2006) Glutathione S-transferase M1, T1, and P1 gene polymorphism in exudative age-related macular degeneration: a preliminary report. Eur J Ophthalmol 16:105–110
Abu-Amero KK, Morales J, Bosley TM (2006) Mitochondrial abnormalities in patients with primary open-angle glaucoma. Invest Ophthalmol Vis Sci 47:2533–2541. doi:10.1167/iovs.05-1639
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Azarpira, N., Nikeghbalian, S., Geramizadeh, B. et al. Influence of glutathione S-transferase M1 and T1 polymorphisms with acute rejection in Iranian liver transplant recipients. Mol Biol Rep 37, 21–25 (2010). https://doi.org/10.1007/s11033-009-9487-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-009-9487-5