Abstract
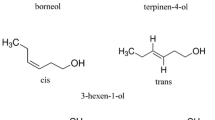
Cruciferous plants (Brassicaceae) are characterized by the accumulation of a group of secondary metabolites known as glucosinolates that, following attack by pathogens or herbivores, may be hydrolyzed to one of a number of products including isothiocyanates and nitriles. Despite the range of hydrolysis products that may be produced, the toxicity of glucosinolates to pathogens and herbivores may be explained largely by the production of isothiocyanates. Isothiocyanates are also known to provide an indirect defense by acting as host finding cues for parasitoids of insect herbivores that attack crucifers. It has been speculated that nitriles may provide a similar indirect defense. Here, we investigate the olfactory perception and orientation behavior of the aphid parasitoid Diaeretiella rapae, to a range of alkenylglucosinolate hydrolysis products, including isothiocyanates, nitriles, and epithionitriles. Electroantennogram responses indicated peripheral odor perception in D. rapae females to all 3-butenylglucosinolate hydrolysis products tested. By contrast, of the 2-propenylglucosinolate hydrolysis products tested, only the isothiocyanate elicited significant responses. Despite showing peripheral olfactory detection of a range of 3-butenylglucosinolate hydrolysis products, naïve females oriented only to the isothiocyanate. Similarly, parasitoids oriented to 3-isothiocyanatoprop-1-ene, but not to the corresponding nitrile or epithionitrile. However, by rearing D. rapae either on Brassica nigra, characterized by the accumulation of 2-propenylglucosinolate, or Brassica rapa var rapifera, characterized by the accumulation of 3-butenylglucosinolate, altered the innate response of parasitoids to 3-isothiocyanatoprop-1-ene and 4-isothiocyanatobut-1-ene. These results are discussed in relation to the defensive roles of glucosinolate hydrolysis products and the influence of the host plant on aphid parasitoid behavior.




Similar content being viewed by others
References
Baker, T. C., and Roelofs, W. L. 1976. Electroantennogram responses of the male moth, Argyrotaenia velutinana to mixtures of sex pheromone components of the female. J. Insect Physiol. 22:1357–1364.
Benton, R. 2006. On the origin of smell: odorant receptors in insects. Cell. Mol. Life Sci. 63:1579–1585.
Blande, J. D. 2004. Differential signaling from specialist and generalist Brassica feeding aphids to differentially adapted aphid parasitoids. Ph.D. thesis, University of Southampton, UK.
Blande, J. D., Pickett, J. A., and Poppy, G. M. 2004. Attack rate and success of the parasitoid Diaeretiella rapae on specialist and generalist feeding aphids. J. Chem. Ecol. 30:1781–1795.
Blande, J. D., Pickett, J. A., and Poppy, G. M. 2007. A comparison of semiochemically mediated interactions involving specialist and generalist Brassica-feeding aphids and the braconid parasitoid Diaeretiella rapae. J. Chem. Ecol. 33:767–779.
Bones, A. M., and Rossiter, J. T. 1996. The myrosinase-glucosinolate system, its organisation and biochemistry. Physiol. Plant. 97:194–208.
Bones, A. M., and Rossiter, J. T. 2006. The enzymic and chemically induced decomposition of glucosinolates. Phytochemistry 67:1053–1067.
Bradburne, R. P., and Mithen, R. 2000. Glucosinolate genetics and the attraction of the aphid parasitoid Diaeretiella rapae to Brassica. Proc. R. Soc. Lond., Ser. B Biol. Sci. 267:89–95.
Burow, M., Muller, R., Gershenzon, J., and Wittstock, U. 2006. Altered glucosinolate hydrolysis in genetically engineered Arabidopsis thaliana and its influence on the larval development of Spodoptera littoralis. J. Chem. Ecol. 32:2333–2349.
Carlson, D. G., Daxenbichler, M. E., Vanetten, C. H., and Tookey, H. L. 1981. Glucosinolates in crucifer vegetables—turnips and rutabagas. J. Agric. Food Chem. 29:1235–1239.
Caterina, M. J., Schumacher, M. A., Tominaga, M., Rosen, T. A., Levine, J. D., and Julius, D. 1997. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389:816–824.
Caterina, M. J., Rosen, T. A., Tominaga, M., Brake, A. J., and Julius, D. 1999. A capsaicin-receptor homologue with a high threshold for noxious heat. Nature 398:436–441.
Cole, R. 1976. Isothiocyanates, nitriles and thiocyanates as products of autolysis of glucosinolates in Cruciferae. Phytochemistry 15:759–762.
Dawson, G. W., Griffiths, D. C., Pickett, J. A., Wadhams, L. J., and Woodcock, C. M. 1987. Plant-derived synergists of alarm pheromone from turnip aphid, Lipaphis (Hyadaphis) erysimi (Homoptera, Aphididae). J. Chem. Ecol. 13:1663–1671.
Du, Y. J., Poppy, G. M., and Powell, W. 1996. Relative importance of semiochemicals from first and second trophic levels in host foraging behavior of Aphidius ervi. J. Chem. Ecol. 22:1591–1605.
Girling, R. D., Hassall, M., Turner, J. G., and Poppy, G. M. 2006. Behavioural responses of the aphid parasitoid Diaeretiella rapae to volatiles from Arabidopsis thaliana induced by Myzus persicae. Entomol. Exp. Appl. 120:1–9.
Halkier, B. A., and Gershenzon, J. 2006. Biology and biochemistry of glucosinolates. Annu. Rev. Plant Biol. 57:303–333.
Jander, G., Cui, J. P., Nhan, B., Pierce, N. E., and Ausubel, F. M. 2001. The TASTY locus on chromosome 1 of Arabidopsis affects feeding of the insect herbivore Trichoplusia ni. Plant Physiol. 126:890–898.
Kelly, P. J., Bones, A., and Rossiter, J. T. 1998. Sub-cellular immunolocalization of the glucosinolate sinigrin in seedlings of Brassica juncea. Planta 206:370–377.
Kliebenstein, D. J. 2004. Secondary metabolites and plant/environment interactions: a view through Arabidopsis thaliana tinged glasses. Plant Cell Environ. 27:675–684.
Kliebenstein, D. J., Kroymann, J., and Mitchell-Olds, T. 2005a. The glucosinolate-myrosinase system in an ecological and evolutionary context. Curr. Opin. Plant Biol. 8:264–271.
Kliebenstein, D. J., Rowe, H. C., and Denby, K. J. 2005b. Secondary metabolites influence Arabidopsis/Botrytis interactions: variation in host production and pathogen sensitivity. Plant J. 44:25–36.
Koroleva, O. A., Davies, A., Deeken, R., Thorpe, M. R., Tomos, A. D., and Hedrich, R. 2000. Identification of a new glucosinolate-rich cell type in Arabidopsis flower stalk. Plant Physiol. 124:599–608.
Lambrix, V., Reichelt, M., Mitchell-Olds, T., Kliebenstein, D. J., and Gershenzon, J. 2001. The Arabidopsis epithiospecifier protein promotes the hydrolysis of glucosinolates to nitriles and influences Trichoplusia ni herbivory. Plant Cell 13:2793–2807.
Luthy, J., and Benn, M. 1979. Conversion of potassium allylglucosinolate to 3,4-epithiobutanenitrile by crambe-abyssinica seed flour. Phytochemistry 18:2028–2029.
Nottingham, S. F., Hardie, J., Dawson, G. W., Hick, A. J., Pickett, J. A., Wadhams, L. J., and Woodcock, C. M. 1991. Behavioral and electrophysiological responses of aphids to host and nonhost plant volatiles. J. Chem. Ecol. 17:1231–1242.
Ohta, T., Imagawa, T., and Ito, S. 2007. Novel agonistic action of mustard oil on recombinant and endogenous porcine transient receptor potential V1 (pTRPV1) channels. Biochem. Pharmacol. 73:1646–1656.
Read, D. P., Feeny, P. P., and Root, R. B. 1970. Habitat selection by the aphid parasite Diaeretiella rapae (Hymenoptera: Braconidae) and hyperparasite Charips brassicae (Hymenoptera: Cynipidae). Can. Entomol. 102:1567–&.
Reed, H. C., Tan, S. H., Haapanen, K., Killmon, M., Reed, D. K., and Elliott, N. C. 1995. Olfactory responses of the parasitoid Diaeretiella rapae (Hymenoptera: Aphidiidae) to odor of plants, aphids, and plant–aphid complexes. J. Chem. Ecol. 21:407–418.
Storeck, A., Poppy G. M., van Emden, H. F., and Powell, W. 2000. The role of plant chemical cues in determining host preference in the generalist aphid parasitoid Aphidius colemani. Entomol. Exp. Appl. 97:41–46.
Van, Poecke, R. M. P., Posthumus, M. A., and Dicke, M. 2001. Herbivore-induced volatile production by Arabidopsis thaliana leads to attraction of the parasitoid Cotesia rubecula: chemical, behavioral, and gene-expression analysis. J. Chem. Ecol. 27:1911–1928.
Vaughn, T. T., Antolin, M. F., and Bjostad, L. B. 1996. Behavioral and physiological responses of Diaeretiella rapae to semiochemicals. Entomol. Exp. Appl. 78:187–196.
Wickremasinghe, M. G. V., and van, Emden, H. F. 1992. Reactions of adult female parasitoids, particularly Aphidius rhopalosiphi, to volatile chemical cues from the host plants of their aphid prey. Physiol. Entomol. 17:297–304.
Wink, M. 1988. Plant breeding—importance of plant secondary metabolites for protection against pathogens and herbivores. Theor. Appl. Gen. 75:225–233.
Wittstock, U., and Burow, M. 2007. Tipping the scales—Specifier proteins in glucosinolate hydrolysis. IUBMB Life 59:744–751.
Zabala, M. D., Grant, M. Bones, A. M., Bennett, R., Lim, Y. S., Kissen, R., and Rossiter, J. T. 2005. Characterisation of recombinant epithiospecifier protein and its over-expression in Arabidopsis thaliana. Phytochemistry 66:859–867.
Acknowledgments
We thank Valerie Elliot for technical assistance and the BBSRC for providing financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pope, T.W., Kissen, R., Grant, M. et al. Comparative Innate Responses of the Aphid Parasitoid Diaeretiella rapae to Alkenyl Glucosinolate Derived Isothiocyanates, Nitriles, and Epithionitriles. J Chem Ecol 34, 1302–1310 (2008). https://doi.org/10.1007/s10886-008-9531-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-008-9531-2