Abstract
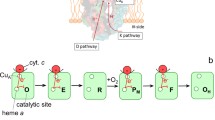
The ba 3-type cytochrome c oxidase from Thermus thermophilus is phylogenetically very distant from the aa 3–type cytochrome c oxidases. Nevertheless, both types of oxidases have the same number of redox-active metal sites and the reduction of O2 to water is catalysed at a haem a 3-CuB catalytic site. The three-dimensional structure of the ba 3 oxidase reveals three possible proton-conducting pathways showing very low homology compared to those of the mitochondrial, Rhodobacter sphaeroides and Paracoccus denitrificans aa 3 oxidases. In this study we investigated the oxidative part of the catalytic cycle of the ba 3 -cytochrome c oxidase using the flow-flash method. After flash-induced dissociation of CO from the fully reduced enzyme in the presence of oxygen we observed rapid oxidation of cytochrome b (k≅6.8 × 104 s−1) and formation of the peroxy (PR) intermediate. In the next step a proton was taken up from solution with a rate constant of ∼1.7 × 104 s−1, associated with formation of the ferryl (F) intermediate, simultaneous with transient reduction of haem b. Finally, the enzyme was oxidized with a rate constant of ∼1,100 s−1, accompanied by additional proton uptake. The total proton uptake stoichiometry in the oxidative part of the catalytic cycle was ~1.5 protons per enzyme molecule. The results support the earlier proposal that the PR and F intermediate spectra are similar (Siletsky et al. Biochim Biophys Acta 1767:138, 2007) and show that even though the architecture of the proton-conducting pathways is different in the ba 3 oxidases, the proton-uptake reactions occur over the same time scales as in the aa 3-type oxidases.
Similar content being viewed by others
References
Ädelroth P, Svensson Ek M, Mitchell DM, Gennis RB, Brzezinski P (1997) Glutamate 286 in cytochrome aa3 from Rhodobacter sphaeroides is involved in proton uptake during the reaction of the fully-reduced enzyme with dioxygen. Biochemistry 36(45):13824–13829
Ädelroth P, Ek M, Brzezinski P (1998) Factors determining electron-transfer rates in cytochrome c oxidase: investigation of the oxygen reaction in the R. sphaeroides and bovine enzymes. Biochim Biophys Acta 1367(1–3):107–117
Brändén G, Gennis RB, Brzezinski P (2006) Transmembrane proton translocation by cytochrome c oxidase. Biochim Biophys Acta 1757(8):1052–1063
Belevich I, Verkhovsky MI (2008) Molecular mechanism of proton translocation by cytochrome c oxidase. Antioxid Redox Signal 10(1):1–29
Blomberg LM, Blomberg MR, Siegbahn PE (2006) A theoretical study on nitric oxide reductase activity in a ba3-type heme-copper oxidase. Biochim Biophys Acta 1757(1):31–46
Brunori M, Giuffre A, Sarti P (2005) Cytochrome c oxidase, ligands and electrons. J Inorg Biochem 99(1):324–336
Brzezinski P, Ädelroth P (2006) Design principles of proton-pumping haem-copper oxidases. Curr Opin Struct Biol 16(4):465–472
Chen Y, Hunsicker-Wang L, Pacoma RL, Luna E, Fee JA (2005) A homologous expression system for obtaining engineered cytochrome ba 3 from Thermus thermophilus HB8. Protein Expr Purif 40:299–318
Einarsdóttir Ó (1995) Fast reactions of cytochrome-oxidase. Biochim Biophys Acta 1229(2):129–147
Faxén K, Gilderson G, Ädelroth P, Brzezinski P (2005) A mechanistic principle for proton pumping by cytochrome c oxidase. Nature 437(7056):286–289
Ferguson-Miller S, Babcock GT (1996) Heme/copper terminal oxidases. Chem Rev 96(7):2889–2907
Giuffrè A, Forte E, Antonini G, D'Itri E, Brunori M, Soulimane T, Buse G (1999) Kinetic properties of ba3 oxidase from Thermus thermophilus: effect of temperature. Biochemistry 38(3):1057–1065
Hosler JP, Ferguson-Miller S, Mills DA (2006) Energy transduction: proton transfer through the respiratory complexes. Ann Rev Biochem 75:165–187
Iwata S, Ostermeier C, Ludwig B, Michel H (1995) Structure at 2.8 Å resolution of cytochrome c oxidase from Paracoccus denitrificans. Nature 376(6542):660–669
Jasaitis A, Verkhovsky MI, Morgan JE, Verkhovskaya ML, Wikström M (1999) Assignment and charge translocation stoichiometries of the major electrogenic phases in the reaction of cytochrome c oxidase with dioxygen. Biochemistry 38(9):2697–2706
Kannt A, Soulimane T, Buse G, Becker A, Bamberg E, Michel H (1998) Electrical current generation and proton pumping catalyzed by the ba3- type cytochrome c oxidase from Thermus thermophilus. FEBS Lett 434(1–2):17–22
Keightley JA, Zimmermann BH, Mather MW, Springer P, Pastuszyn A, Lawrence DM, Fee JA (1995) Molecular genetic and protein chemical characterization of the cytochrome ba3 from Thermus thermophilus HB8. J Biol Chem 270(35):20345–20358
Namslauer A, Brzezinski P (2004) Structural elements involved in electron-coupled proton transfer in cytochrome c oxidase. FEBS Lett 567(1):103–110
Ostermeier C, Harrenga A, Ermler U, Michel H (1997) Structure at 2.7 A resolution of the Paracoccus denitrificans two-subunit cytochrome c oxidase complexed with an antibody FV fragment. Proc Natl Acad Sci U S A 94(20):10547–10553
Pereira MM, Santana M, Teixeira M (2001) A novel scenario for the evolution of haem-copper oxygen reductases. Biochim Biophys Acta-Bioenerg 1505(2–3):185–208
Siletsky SA, Belevich I, Jasaitis A, Konstantinov AA, Wikström M, Soulimane T, Verkhovsky MI (2007) Time-resolved single-turnover of ba3 oxidase from Thermus thermophilus. Biochim Biophys Acta 1767(12):1383–1392
Soulimane T, Buse G, Bourenkov GP, Bartunik HD, Huber R, Than ME (2000) Structure and mechanism of the aberrant ba(3)-cytochrome c oxidase from Thermus thermophilus. EMBO J 19(8):1766–1776
Svensson-Ek M, Abramson J, Larsson G, Törnroth S, Brzezinski P, Iwata S (2002) The X-ray crystal structures of wild-type and EQ(I-286) mutant cytochrome c oxidases from Rhodobacter sphaeroides. J Mol Biol 321:329–339
Tsukihara T, Aoyama H, Yamashita E, Tomizaki T, Yamaguchi H, Shinzawa-Itoh K, Nakashima R, Yaono R, Yoshikawa S (1996) The whole structure of the 13-subunit oxidized cytochrome c oxidase at 2.8 Å. Science 272(5265):1136–1144
Wikström M, Morgan JE (1992) The dioxygen cycle. Spectral, kinetic, and thermodynamic characteristics of ferryl and peroxy intermediates observed by reversal of the cytochrome oxidase reaction. J Biol Chem 267(15):10266–10273
Wikström M, Verkhovsky MI (2002) Proton translocation by cytochrome c oxidase in different phases of the catalytic cycle. Biochim Biophys Acta (BBA) - Bioenerg 1555(1–3):128–132
Wikström M, Verkhovsky MI (2006) Towards the mechanism of proton pumping by the haem-copper oxidases. Biochim Biophys Acta - Bioenerg 1757(8):1047–1051
Wikström M, Verkhovsky MI (2007) Mechanism and energetics of proton translocation by the respiratory heme-copper oxidases. Biochim Biophys Acta (BBA) - Bioenerg 1767(10):1200–1214
Yoshikawa S, Shinzawa-Itoh K, Tsukihara T (1998) Crystal structure of bovine heart cytochrome c oxidase at 2.8 A resolution. J Bioenerg Biomembr 30(1):7–14
Zimmermann BH, Nitsche CI, Fee JA, Rusnak F, Munck E (1988) Properties of a copper-containing cytochrome ba3: a second terminal oxidase from the extreme thermophile Thermus thermophilus. Proc Natl Acad Sci U S A 85(16):5779–5783
Author information
Authors and Affiliations
Corresponding author
Additional information
Smirnova and Zaslavsky contributed equally to the work described in this paper.
Rights and permissions
About this article
Cite this article
Smirnova, I.A., Zaslavsky, D., Fee, J.A. et al. Electron and proton transfer in the ba 3 oxidase from Thermus thermophilus . J Bioenerg Biomembr 40, 281–287 (2008). https://doi.org/10.1007/s10863-008-9157-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-008-9157-7