Abstract
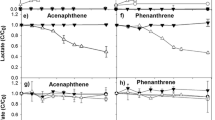
Various microbial activities determine the effectiveness of bioremediation processes. In this work, we evaluated the feasibility of gene array hybridization for monitoring the efficiency of biodegradation processes. Biodegradation of 14C-labelled naphthalene and toluene by the aromatic hydrocarbon-degrading Pseudomonas putida F1, P. putida mt-2 and P. putida G7 was followed in mixed liquid culture microcosm by a preliminary, nylon membrane-based gene array. In the beginning of the study, toluene was degraded rapidly and increased amount of toluene degradation genes was detected by the preliminary gene array developed for the study. After toluene was degraded, naphthalene mineralization started and the amount of naphthalene degradation genes increased as biodegradation proceeded. The amount of toluene degradation genes decreased towards the end of the study. The hybridization signal intensities determined by preliminary gene array were in good agreement with mineralization of naphthalene and toluene and with the amount of naphthalene dioxygenase and toluene dioxygenase genes quantified by dot blot hybridization. The clear correlation between the results obtained by the preliminary array and the biodegradation process suggests that gene array methods can be considered as a promising tool for monitoring the efficiency of biodegradation processes.





Similar content being viewed by others
References
Aalto MK, Ronne H, Keranen S (1993) Yeast syntaxins Sso1p and Sso2p belong to a family of related membrane proteins that function in vesicular transport. EMBO J 12(11):4095–4104
Ahn Y, Sanseverino J, Sayler GS (1999) Analyses of polycyclic aromatic hydrocarbon-degrading bacteria isolated from contaminated soils. Biodegradation 10(2):149–157
Baelum J, Henriksen T, Hansen HC, Jacobsen CS (2006) Degradation of 4-chloro-2-methylphenoxyacetic acid in top- and subsoil is quantitatively linked to the class III tfdA gene. Appl Environ Microbiol 72(2):1476–1486
Bavykin SG, Akowski JP, Zakhariev VM, Barsky VE, Perov AN, Mirzabekov AD (2001) Portable system for microbial sample preparation and oligonucleotide microarray analysis. Appl Environ Microbiol 67(2):922–928
Bodrossy L, Stralis-Pavese N, Murrell JC, Radajewski S, Weilharter A, Sessitsch A (2003) Development and validation of a diagnostic microbial microarray for methanotrophs. Environ Microbiol 5(7):566–582
Bouchez M, Blanchet D, Vandecasteele JP (1995) Degradation of polycyclic aromatic hydrocarbons by pure strains and by defined strain associations: inhibition phenomena and cometabolism. Appl Microbiol Biotechnol 43(1):156–164
Brodie EL, Desantis TZ, Joyner DC et al (2006) Application of a high-density oligonucleotide microarray approach to study bacterial population dynamics during uranium reduction and reoxidation. Appl Environ Microbiol 72(9):6288–6298
Burlage RS, Hooper SW, Sayler GS (1989) The TOL (pWW0) catabolic plasmid. Appl Environ Microbiol 55(6):1323–1328
Cavalca L, Di Gennaro P, Colombo M et al (2000) Distribution of catabolic pathways in some hydrocarbon-degrading bacteria from a subsurface polluted soil. Res Microbiol 151(10):877–887
Cho JC, Tiedje JM (2002) Quantitative detection of microbial genes by using DNA microarrays. Appl Environ Microbiol 68(3):1425–1430
Cook KL, Sayler GS (2003) Environmental application of array technology: promise, problems and practicalities. Curr Opin Biotechnol 14(3):311–318
Denef VJ, Park J, Rodrigues JL, Tsoi TV, Hashsham SA, Tiedje JM (2003) Validation of a more sensitive method for using spotted oligonucleotide DNA microarrays for functional genomics studies on bacterial communities. Environ Microbiol 5(10):933–943
Dennis P, Edwards EA, Liss SN, Fulthorpe R (2003) Monitoring gene expression in mixed microbial communities by using DNA microarrays. Appl Environ Microbiol 69(2):769–778
Eaton RW (1994) Organization and evolution of naphthalene catabolic pathways: sequence of the DNA encoding 2-hydroxychromene-2-carboxylate isomerase and trans-o-hydroxybenzylidenepyruvate hydratase-aldolase from the NAH7 plasmid. J Bacteriol 176(24):7757–7762
Ferrero M, Llobet-Brossa E, Lalucat J, Garcia-Valdes E, Rossello-Mora R, Bosch R (2002) Coexistence of two distinct copies of naphthalene degradation genes in Pseudomonas strains isolated from the western Mediterranean region. Appl Environ Microbiol 68(2):957–962
Franke-Whittle IH, Klammer SH, Insam H (2005) Design and application of an oligonucleotide microarray for the investigation of compost microbial communities. J Microbiol Methods 62(1):37–56
Gentry TJ, Wickham GS, Schadt CW, He Z, Zhou J (2006) Microarray applications in microbial ecology research. Microb Ecol 52(2):159–175
Ghosal D, You IS, Gunsalus IC (1987) Nucleotide sequence and expression of gene nahH of plasmid NAH7 and homology with gene xylE of TOL pWWO. Gene 55(1):19–28
Harayama S, Rekik M, Wasserfallen A, Bairoch A (1987) Evolutionary relationships between catabolic pathways for aromatics: conservation of gene order and nucleotide sequences of catechol oxidation genes of pWW0 and NAH7 plasmids. Mol Gen Genet 210(2):241–247
Jantti J, Aalto MK, Oyen M, Sundqvist L, Keranen S, Ronne H (2002) Characterization of temperature-sensitive mutations in the yeast syntaxin 1 homologues Sso1p and Sso2p, and evidence of a distinct function for Sso1p in sporulation. J Cell Sci 115(Pt 2):409–420
Koizumi Y, Kelly JJ, Nakagawa T et al (2002) Parallel characterization of anaerobic toluene- and ethylbenzene-degrading microbial consortia by PCR-denaturing gradient gel electrophoresis, RNA-DNA membrane hybridization, and DNA microarray technology. Appl Environ Microbiol 68(7):3215–3225
Lane S, Evermann J, Loge F, Call DR (2004) Amplicon secondary structure prevents target hybridization to oligonucleotide microarrays. Bios Bioelectron 20(4):728–735
Lee ML, Kuo FC, Whitmore GA, Sklar J (2000) Importance of replication in microarray gene expression studies: statistical methods and evidence from repetitive cDNA hybridizations. Proc Natl Acad Sci USA 97(18):9834–9839
Letowski J, Brousseau R, Masson L (2004) Designing better probes: effect of probe size, mismatch position and number on hybridization in DNA oligonucleotide microarrays. J Microbiol Methods 57(2):269–278
Lievens B, Brouwer M, Vanachter AC, Levesque CA, Cammue BP, Thomma BP (2005) Quantitative assessment of phytopathogenic fungi in various substrates using a DNA macroarray. Environ Microbiol 7(11):1698–1710
Loy A, Schulz C, Lucker S et al (2005) 16S rRNA gene-based oligonucleotide microarray for environmental monitoring of the betaproteobacterial order “Rhodocyclales”. Appl Environ Microbiol 71(3):1373–1386
Martin-Magniette ML, Aubert J, Cabannes E, Daudin JJ (2005) Evaluation of the gene-specific dye bias in cDNA microarray experiments. Bioinformatics (Oxford, England) 21(9):1995–2000
Meinkoth J, Wahl G (1984) Hybridization of nucleic acids immobilized on solid supports. Anal Biochem 138(2):267–284
Muyzer G, de Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59(3):695–700
Okamoto M, Sudhof TC (1997) Mints, Munc18-interacting proteins in synaptic vesicle exocytosis. J Biol Chem 272(50):31459–31464
Peplies J, Lachmund C, Glockner FO, Manz W (2006) A DNA microarray platform based on direct detection of rRNA for characterization of freshwater sediment-related prokaryotic communities. Appl Environ Microbiol 72(7):4829–4838
Piskonen R, Kapanen A, Mansikka T, Rytkonen J, Itavaara M (2002) Evaluation of bioremediation treatments in a Shoreline-Simulating Microcosm. Biorem J 6(2):143–158
Piskonen R, Nyyssonen M, Rajamaki T, Itavaara M (2005) Monitoring of accelerated naphthalene-biodegradation in a bioaugmented soil slurry. Biodegradation 16(2):127–134
Rhee SK, Liu X, Wu L, Chong SC, Wan X, Zhou J (2004) Detection of genes involved in biodegradation and biotransformation in microbial communities by using 50-mer oligonucleotide microarrays. Appl Environ Microbiol 70(7):4303–4317
Sambrook J, Russell D (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, New York, US
Sanseverino J, Werner C, Fleming J, Applegate B, King JMH, Sayler GS (1993) Molecular diagnostic of polycyclic aromatic hydrocarbon biodegradation in manufactured gas plant soils. Biodegradation 4:303–321
Shalon D, Smith SJ, Brown PO (1996) A DNA microarray system for analyzing complex DNA samples using two-color fluorescent probe hybridization. Genome Res 6(7):639–645
Shchepinov MS, Case-Green SC, Southern EM (1997) Steric factors influencing hybridisation of nucleic acids to oligonucleotide arrays. Nucleic Acids Res 25(6):1155–1161
Simon MJ, Osslund TD, Saunders R et al (1993) Sequences of genes encoding naphthalene dioxygenase in Pseudomonas putida strains G7 and NCIB 9816-4. Gene 127(1):31–37
Stephen JR, Chang YJ, Gan YD et al (1999) Microbial characterization of a JP-4 fuel-contaminated site using a combined lipid biomarker/polymerase chain reaction–denaturing gradient gel electrophoresis (PCR-DGGE)-based approach. Environ Microbiol 1(3):231–241
Steward GF, Jenkins BD, Ward BB, Zehr JP (2004) Development and testing of a DNA macroarray to assess nitrogenase (nifH) gene diversity. Appl Environ Microbiol 70(3):1455–1465
Suzuki MT, Giovannoni SJ (1996) Bias caused by template annealing in the amplification of mixtures of 16S rRNA genes by PCR. Appl Environ Microbiol 62(2):625–630
Suzuki M, Hayakawa T, Shaw JP, Rekik M, Harayama S (1991) Primary structure of xylene monooxygenase: similarities to and differences from the alkane hydroxylation system. J Bacteriol 173(5):1690–1695
Taniguchi M, Miura K, Iwao H, Yamanaka S (2001) Quantitative assessment of DNA microarrays—comparison with Northern blot analyses. Genomics 71(1):34–39
Taroncher-Oldenburg G, Griner EM, Francis CA, Ward BB (2003) Oligonucleotide microarray for the study of functional gene diversity in the nitrogen cycle in the environment. Appl Environ Microbiol 69(2):1159–1171
Urakawa H, El Fantroussi S, Smidt H et al (2003) Optimization of single-base-pair mismatch discrimination in oligonucleotide microarrays. Appl Environ Microbiol 69(5):2848–2856
Weber A, Jung K (2002) Profiling early osmostress-dependent gene expression in Escherichia coli using DNA macroarrays. J Bacteriol 184(19):5502–5507
Whited GM, Gibson DT (1991) Toluene-4-monooxygenase, a three-component enzyme system that catalyzes the oxidation of toluene to p-cresol in Pseudomonas mendocina KR1. J Bacteriol 173(9):3010–3016
Whyte LG, Greer CW, Inniss WE (1996) Assessment of the biodegradation potential of psychrotrophic microorganisms. Can J Microbiol 42(2):99–106
Wilson KH, Wilson WJ, Radosevich JL et al (2002) High-density microarray of small-subunit ribosomal DNA probes. Appl Environ Microbiol 68(5):2535–2541
Wu L, Thompson DK, Li G, Hurt RA, Tiedje JM, Zhou J (2001) Development and evaluation of functional gene arrays for detection of selected genes in the environment. Appl Environ Microbiol 67(12):5780–5790
Yen KM, Serdar CM (1988) Genetics of naphthalene catabolism in pseudomonads. Crit Rev Microbiol 15(3):247–268
Zukowski MM, Gaffney DF, Speck D et al (1983) Chromogenic identification of genetic regulatory signals in Bacillus subtilis based on expression of a cloned Pseudomonas gene. Proc Natl Acad Sci USA 80(4):1101–1105
Zylstra GJ, Wackett LP, Gibson DT (1989) Trichloroethylene degradation by Escherichia coli containing the cloned Pseudomonas putida F1 toluene dioxygenase genes. Appl Environ Microbiol 55(12):3162–3166
Acknowledgements
Funding for the research was provided by the Neste Foundation, the Ekokem Foundation, the Finnish Funding Agency for Technology and Innovation (TEKES) and VTT’s Strategic Clean World Tecnology Theme, T2 In situ Project. Phil.Lic. Tiina Rajamäki is acknowledged for developing the gas chromatography method for analysing toluene and naphthalene. Mrs. Anna-Liisa Ruskeepää is thanked for performing the toluene and naphthalene analyses. Dr. Jussi Jäntti is thanked for providing the E. coli strains carrying recombinant plasmids, from which the negative control genes were isolated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Piskonen, R., Nyyssönen, M. & Itävaara, M. Evaluating the biodegradation of aromatic hydrocarbons by monitoring of several functional genes. Biodegradation 19, 883–895 (2008). https://doi.org/10.1007/s10532-008-9190-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-008-9190-6