Abstract
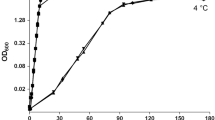
Sulfolobus solfataricus, a hyperthermophilic crenarchaeon, contains two genes encoding reverse gyrases, topR1 and topR2. The steady-state level of their transcripts were quantified during the growth phases for cells maintained either at 72, or 80°C, and after temperature changes from one to the other temperature. The transcripts of both genes are weakly expressed, but the highest level is observed in actively dividing cells, and is almost undetectable in cells in decline phase. During the temperature shift experiments, there is no significant topR2 variation. By contrast, there is a maximum 2.4-fold increase in topR1 transcripts within 30 min after the downshift. After 1 h, the transcript level reaches the level characteristic of cells adapted to the new temperature. After an upward shift, the topR1 expression pattern is inversely regulated with a transient decrease with the same time course. The topR1 expression profile is completely different from that of topR2 after temperature shift experiments; this suggests a different regulation process for the two reverse gyrase genes. The fine tuning of the topR1 transcript expression within a short interval of time after a temperature shift illustrates a rapid adaptation response to temperature change.





Similar content being viewed by others
References
Andersson AF, Lundgren M, Eriksson S, Rosenlund M, Bernander R, Nilsson P (2006) Global analysis of mRNA stability in the archaeon Sulfolobus. Genome Biol 7:R99.1–R99.10
Atomi H, Matsumi R, Imanaka T (2004) Reverse gyrase is not a prerequisite for hyperthermophilic life. J Bacteriol 186:4829–4833
Bell SD, Jaxel C, Nadal M, Kosa PF, Jackson SP (1998) Temperature, template topology, and factor requirements of archaeal transcription. Proc Natl Acad Sci USA 95:15218–15222
Champoux JJ (2001) DNA topoisomerases: structure, function, and mechanism. Annu Rev Biochem 70:369–413
Champoux JJ, Dulbecco R (1972) An activity from mammalian cells that untwists superhelical DNA. A possible swivel for DNA replication. Proc Natl Acad Sci USA 69:143–146
Confalonieri F, Elie C, Nadal M, Bouthier de la Tour CB, Forterre P, Duguet M (1993) Reverse gyrase: a helicase-like domain and a type I topoisomerase in the same polypeptide. Proc Natl Acad Sci USA 90:4753–4757
Corbett KD, Berger JM (2004) Structure, molecular mechanisms, and evolutionary relationships in DNA topoisomerases. Annu Rev Biophys Biomol Struct 33:95–118
Declais AC, Marsault J, Confalonieri F, Bouthier de laTour C, Duguet M (2000) Reverse gyrase, the two domains intimately cooperate to promote positive supercoiling. J Biol Chem 275:19498–19504
Duguet M (1993) The helical repeat of DNA at high temperature. Nucleic Acids Res 21:463–468
Forterre P (2002) A hot story from comparative genomics: reverse gyrase is the only hyperthermophile-specific protein. Trends Genet 18:236–237
Forterre P, Gribaldo S, Gadelle D, Serre MC (2007) Origin and evolution of DNA topoisomerases. Biochimie 89:427–446
Forterre P, Mirambeau G, Jaxel C, Nadal M, Duguet M (1985) High positive supercoiling in vitro catalyzed by an ATP and polyethylene glycol-stimulated topoisomerase from Sulfolobus acidocaldarius. Embo J 4:2123–2128
Jaxel C, Bouthier de la Tour C, Duguet M, Nadal M (1996) Reverse gyrase gene from Sulfolobus shibatae B12: gene structure, transcription unit and comparative sequence analysis of the two domains. Nucleic Acids Res 24:4668–4675
Kagawa HK, Yaoi T, Brocchieri L, McMillan RA, Alton T, Trent JD (2003) The composition, structure and stability of a group II chaperonin are temperature regulated in a hyperthermophilic archaeon. Mol Microbiol 48:143–156
Kikuchi A, Asai K (1984) Reverse gyrase–a topoisomerase which introduces positive superhelical turns into DNA. Nature 309:677–681
Krah R, Kozyavkin SA, Slesarev AI, Gellert M (1996) A two-subunit type I DNA topoisomerase (reverse gyrase) from an extreme hyperthermophile. Proc Natl Acad Sci USA 93:106–110
Krogh BO, Shuman S (2002) A poxvirus-like type IB topoisomerase family in bacteria. Proc Natl Acad Sci USA 99:1853–1858
Lee MH, Park H, Shim G, Lee J, Koo HS (2001) Regulation of gene expression, cellular localization, and in vivo function of Caenorhabditis elegans DNA topoisomerase I. Genes Cells 6:303–312
Lopez-Garcia P, Forterre P (1997) DNA topology in hyperthermophilic archaea: reference states and their variation with growth phase, growth temperature, and temperature stresses. Mol Microbiol 23:1267–1279
Lopez-Garcia P, Forterre P (1999) Control of DNA topology during thermal stress in hyperthermophilic archaea: DNA topoisomerase levels, activities and induced thermotolerance during heat and cold shock in Sulfolobus. Mol Microbiol 33:766–777
Lundgren M, Bernander R (2007) Genome-wide transcription map of an archaeal cell cycle. Proc Natl Acad Sci USA 104:2939–2944
Menzel R, Gellert M (1983) Regulation of the genes for E. coli DNA gyrase: homeostatic control of DNA supercoiling. Cell 34:105–113
Mudgil Y, Singh BN, Upadhyaya KC, Sopory SK, Reddy MK (2002) Cloning and characterization of a cell cycle-regulated gene encoding topoisomerase I from Nicotiana tabacum that is inducible by light, low temperature and abscisic acid. Mol Genet Genomics 267:380–390
Nadal M (2007) Reverse gyrase: an insight into the role of DNA-topoisomerases. Biochimie 89:447–455
Nadal M, Couderc E, Duguet M, Jaxel C (1994) Purification and characterization of reverse gyrase from Sulfolobus shibatae. Its proteolytic product appears as an ATP-independent topoisomerase. J Biol Chem 269:5255–5263
Nadal M, Jaxel C, Portemer C, Forterre P, Mirambeau G, Duguet M (1988) Reverse gyrase of Sulfolobus: purification to homogeneity and characterization. Biochemistry 27:9102–9108
Park EJ, Han SY, Chung IK (2001) Regulation of mouse DNA topoisomerase IIIalpha gene expression by YY1 and USF transcription factors. Biochem Biophys Res Commun 283:384–391
Peter BJ, Arsuaga J, Breier AM, Khodursky AB, Brown PO, Cozzarelli NR (2004) Genomic transcriptional response to loss of chromosomal supercoiling in Escherichia coli. Genome Biol 5:R87.1–R87.16
Richmond CS, Glasner JD, Mau R, Jin H, Blattner FR (1999) Genome-wide expression profiling in Escherichia coli K-12. Nucleic Acids Res 27:3821–3835
Rodriguez AC, Stock D (2002) Crystal structure of reverse gyrase: insights into the positive supercoiling of DNA. Embo J 21:418–426
She Q, Singh RK, Confalonieri F, Zivanovic Y, Allard G, Awayez MJ, Chan-Weiher CC, Clausen IG, Curtis BA, De Moors A, Erauso G, Fletcher C, Gordon PM, Heikamp-de Jong I, Jeffries AC, Kozera CJ, Medina N, Peng X, Thi-Ngoc HP, Redder P, Schenk ME, Theriault C, Tolstrup N, Charlebois RL, Doolittle WF, Duguet M, Gaasterland T, Garrett RA, Ragan MA, Sensen CW, Van der Oost J (2001) The complete genome of the crenarchaeon Sulfolobus solfataricus P2. Proc Natl Acad Sci USA 98:7835–7840
Spellman PT, Sherlock G, Zhang MQ, Iyer VR, Anders K, Eisen MB, Brown PO, Botstein D, Futcher B (1998) Comprehensive identification of cell cycle-regulated genes of the yeast Saccharomyces cerevisiae by microarray hybridization. Mol Biol Cell 9:3273–3297
Tosh K, Cheesman S, Horrocks P, Kilbey B (1999) Plasmodium falciparum: stage-related expression of topoisomerase I. Exp Parasitol 91:126–132
Tse-Dinh YC (1998) Bacterial and archeal type I topoisomerases. Biochim Biophys Acta 1400:19–27
Tse-Dinh YC, Qi H, Menzel R (1997) DNA supercoiling and bacterial adaptation: thermotolerance and thermoresistance. Trends Microbiol 5:323–326
Valenti A, Napoli A, Ferrara MC, Nadal M, Rossi M, Ciaramella M (2006) Selective degradation of reverse gyrase and DNA fragmentation induced by alkylating agent in the archaeon Sulfolobus solfataricus. Nucleic Acids Res 34:2098–2108
Wang JC (1971) Interaction between DNA and an Escherichia coli protein ω. J Mol Biol 55:523–533
Wang JC (2002) Cellular roles of DNA topoisomerases: a molecular perspective. Nat Rev Mol Cell Biol 3:430–440
Zechiedrich EL, Khodursky AB, Bachellier S, Schneider R, Chen D, Lilley DM, Cozzarelli NR (2000) Roles of topoisomerases in maintaining steady-state DNA supercoiling in Escherichia coli. J Biol Chem 275:8103–8113
Zillig W, Kletzin A, Schleper C, Holz I, Janekovic D, Hain J, Lanzendörfer M, Kristjansson JK (1994) Screening for sulfolobales, their plasmids and their viruses in Icelandic solfataras. Syst Appl Microbiol 16:609–628
Acknowledgments
We thank Alex Grossmann for the critical reading of the manuscript. This work was supported by Université Versailles-St-Quentin-en-Yvelines and by Centre National de la Recherche Scientifique (CNRS).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by F. Robb.
Rights and permissions
About this article
Cite this article
Garnier, F., Nadal, M. Transcriptional analysis of the two reverse gyrase encoding genes of Sulfolobus solfataricus P2 in relation to the growth phases and temperature conditions. Extremophiles 12, 799–809 (2008). https://doi.org/10.1007/s00792-008-0186-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-008-0186-2