Abstract
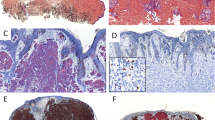
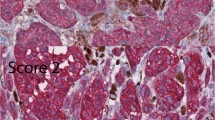
Upon the introduction of extensive sampling protocols of sentinel node biopsies, pathologists are increasingly confronted with small melanoma metastases. Using conventional histology, it proves sometimes difficult or impossible to differentiate small melanoma metastases from lymph-node nevi. Loss of the tumour suppressor gene p16 has been shown to be associated with tumour progression of melanoma. We investigated nevus and melanoma cells for the presence of the product of the gene p16, using immunohistochemistry. All nevus cells, independent of their location (nodal or skin) displayed an extensive nuclear and cytoplasmic staining for p16. In contrast, all cells of melanoma metastases, except one skin metastasis, lacked nuclear staining for p16. These findings indicate that p16 is a reliable marker to distinguish lymph-node nevi from melanoma metastasis.




Similar content being viewed by others
References
Andreola S, Clemente C (1985) Nevus cells in axillary lymphnodes from radical mastectomy specimens. Pathol Res Pract 179:616–618
Carson KF, Wen DR, Li PX, Lana AM, Bailly C, Morton DL, Cochran AJ (1996) Nodal nevi and cutaneous melanomas. Am J Surg Pathol 20:834–840
Castellano M, Pollock PM, Walters MK, Sparrow LE, Down LM, Gabrielle BG, Parson PG, Hayward NK (1997) CDKN2A/p16 is inactivated in most melanoma cell lines. Cancer Res 57:4868–4875
de Vries TJ, Smeets M, de Graaf R, Hou-Jensen K, Brocker EB, Renar N, Eggermont AM, van Muijen GN, Ruiter D (2001) Expression of gp100, MART-1, tyrosinase, and S100 in paraffin-embedded primary melanomas and locoregional, lymph node, and visceral metastases: implications for diagnosis and immunotherapy. A study conducted by the EORTC Melanoma Cooperative Group. J Pathol 193:13–20
Fisher CJ, Hill S, Millis RR (1994) Benign lymph node inclusions mimicking metastatic carcinoma. J Clin Pathol 47:245–247
Fontaine D, Parkhill W, Greer W, Walsh N (2002) Nevus cells in lymph nodes: an association with congenital cutaneous nevi. Am J Dermatopathol 24:1–5
Fujimoto A, Morita R, Hatta N, Takehara K, Takata M (1999) p16INK4a inactivation is not frequent in uncultured sporadic primary cutaneous melanoma. Oncogene 18:2527–2532
Funk JO, Schiller PI, Barret MT, Wong DJ, Kind P, Sander CA (1998) p16INK4a expression is frequently decreased and associated with 9p21 loss of heterozygosity in sporadic melanoma. J Cutan Pathol 25:291–296
Hilton DA, Penney M, Evans B, Sanders H, Love S (2002) Evaluation of molecular markers in low-grade diffuse astrocytomas: loss of p16 and retinoblastoma protein expression is associated with short survival. Am J Surg Pathol 26:472–478
Kamb A, Grius NA, Weaver-Feldhaus J, Liu Q, Harshman K, Tavtigian SV, Stockert E, Day RS 3rd, Johnson BE, Skolnick MH (1994) A cell cycle regulator potentially involved in genesis of many tumor types. Science 264:436–440
Kossard S, Wilkinson B (1997) Small cell (naevoid) melanoma: a clinicopathologic study of 131 cases. Australas J Dermatol 38[Suppl 1]:S54–S58
Lambert WC, Brodkin RH (1984) Nodal and subcutaneous cellular blue nevi. A pseudometastasizing pseudomelanoma. Arch Dermatol 120:367–370
Lohmann CM, Iversen K, Jungbluth AA, Berwick M, Busam KJ (2002) Expression of melanocyte differentiation antigens and ki-67 in nodal nevi and comparison of ki-67 expression with metastatic melanoma. Am J Surg Pathol 26:1351–1357
McCarthy SW, Palmer AA, Bale PM, Hirst E (1974) Naevus cells in lymph nodes. Pathology 6:351–358
Nobori T, Miura K, Wu DJ, Lois A, Takabayashi K, Carson DA (1994) Deletions of the cyclin-dependent kinase-4 inhibitor gene in multiple human cancers. Nature 368:753–756
Reed JA, Loganzo F Jr, Shea CR, Waker GJ, Flores FJ, Glendening JM, Bogdany JK, Shiel MJ, Haluska FG, Fountain JW (1995) Loss of expression of the p16/cyclin-dependent kinase inhibitor 2 tumor suppressor gene in melanocytic lesions correlates with invasive stage of tumor progression. Cancer Res 55:2713–2718
Ridolfi RL, Rosen PP, Thaler H (1977) Nevus cell aggregates associated with lymph nodes: estimated frequency and clinical significance. Cancer 39:164–171
Serrano M, Hannon GJ, Beach D (1993) A new regulatory motif in cell-cycle control causing specific inhibition of cyclin D/CDK4. Nature 366:704–707
Sterchi JM, Muss HB, Weidner N (1987) Cellular blue nevus simulating metastatic melanoma: report of an unusually large lesion associated with nevus-cell aggregates in regional lymph nodes. J Surg Oncol 36:71–75
Subramony C, Lewin JR (1985) Nevus cells within lymph nodes. Possible metastases from a benign intradermal nevus. Am J Clin Pathol 84:220–223
Zembowicz A, McCusker M, Chiarelli C, Dei Tos AP, Granter SR, Calonje E, McKee PH (2001) Morphological analysis of nevoid melanoma: a study of 20 cases with a review of the literature. Am J Dermatopathol 23:167–175
Acknowledgements
We thank N. Wey for photographic and computer-assisted reproductions and Dr. W. Jochum, Department of Pathology, University Hospital Zürich, for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mihic-Probst, D., Saremaslani, P., Komminoth, P. et al. Immunostaining for the tumour suppressor gene p16 product is a useful marker to differentiate melanoma metastasis from lymph-node nevus. Virchows Arch 443, 745–751 (2003). https://doi.org/10.1007/s00428-003-0897-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-003-0897-9