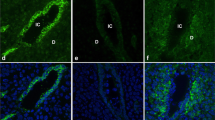
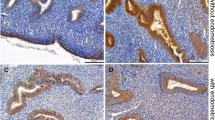
Abstract
The non-receptive uterine luminal epithelium forms an intact polarised epithelial barrier that is refractory to blastocyst invasion. During implantation, organised dismantling of this barrier leads to a receptive state promoting blastocyst attachment. Claudins are tight junction proteins that increase in the uterine epithelium at the time of implantation. Claudin 7 is a member of this family but demonstrates a basolateral localisation pattern that is distinct from other claudins. The present study investigated the localisation, abundance and hormonal regulation of claudin 7 to elucidate a role for the protein during implantation. The results showed that claudin 7 demonstrates a distinct basal and lateral localisation in the uterine luminal and glandular epithelium throughout early pregnancy. On day 1, claudin 7 is abundantly present in response to ovarian estrogen. At the time of implantation, claudin 7 decreases in abundance. This decrease is not dependent on blastocyst presence, as shown by results in pseudopregnant animals. We propose that claudin 7 mediates intercellular adhesions in the uterine epithelium and also may be responsible for stabilising adhesion proteins at the basolateral cell surface. Thus, claudin 7 may function under the maintenance of the uterine luminal epithelial barrier, in the non-receptive state preventing implantation from occurring.






Similar content being viewed by others
References
Achache H, Revel A (2006) Endometrial receptivity markers, the journey to successful embryo implantation. Hum Reprod Update 12:731–746
Amasheh S, Fromm M, Günzel D (2011) Claudins of intestine and nephron—a correlation of molecular tight junction structure and barrier function. Acta Physiol 201:133–140
Anderson JM, Van Itallie CM (2009) Physiology and function of the tight junction. Cold Spring Harb Perspect Biol 1:a002584
Angelow S, Ahlstrom R, Yu AS (2008) Biology of claudins. Am J Physiol Renal Physiol 295:F867–F876
Carp HJ, Serr DM, Mashiach S, Nebel L (1984) Influence of insemination on the implantation of transferred rat blastocysts. Gynecol Obstet Invest 18:194–198
Carson DD, Bagchi I, Dey SK, Enders AC, Fazleabas AT, Lessey BA, Yoshinaga K (2000) Embryo implantation. Dev Biol 223:217–237
Ding L, Lu Z, Foreman O, Tatum R, Lu Q, Renegar R, Cao J, Chen YH (2012) Inflammation and disruption of the mucosal architecture in claudin-7-deficient mice. Gastroenterology 142:305–315
Fain JN, Psychoyos S, Czernik AJ, Frost S, Cash WD (1973) Indomethacin, lipolysis, and cyclic AMP accumulation in white fat cells. Endocrinology 93:632–639
Fanning AS, Mitic LL, Anderson JM (1999) Transmembrane proteins in the tight junction barrier. J Am Soc Nephrol 10:1337–1345
Fazleabas AT, Strakova Z (2002) Endometrial function: cell specific changes in the uterine environment. Mol Cell Endocrinol 186:143–147
Fujita H, Chiba H, Yokozaki H, Sakai N, Sugimoto K, Wada T, Kojima T, Yamashita T, Sawada N (2006) Differential expression and subcellular localization of claudin-7, -8, -12, -13, and -15 along the mouse intestine. J Histochem Cytochem 54:933–944
Gonzalez-Mariscal L, Namorado Mdel C, Martin D, Sierra G, Reyes JL (2006) The tight junction proteins claudin-7 and -8 display a different subcellular localization at Henle’s loops and collecting ducts of rabbit kidney. Nephrol Dial Transplant 21:2391–2398
Haughian JM, Jackson TA, Koterwas DM, Bradford AP (2006) Endometrial cancer cell survival and apoptosis is regulated by protein kinase C alpha and delta. Endocr Relat Cancer 13:1251–1267
Holmes JL, Van Itallie CM, Rasmussen JE, Anderson JM (2006) Claudin profiling in the mouse during postnatal intestinal development and along the gastrointestinal tract reveals complex expression patterns. Gene Expr Patterns 6:581–588
Illingworth IM, Kiszka I, Bagley S, Ireland GW, Garrod DR, Kimber SJ (2000) Desmosomes are reduced in the mouse uterine luminal epithelium during the preimplantation period of pregnancy: a mechanism for facilitation of implantation. Biol Reprod 63:1764–1773
Inai T, Sengoku A, Hirose E, Iida H, Shibata Y (2007) Claudin-7 expressed on lateral membrane of rat epididymal epithelium does not form aberrant tight junction strands. Anat Rec (Hoboken) 290:1431–1438
Johnson AH, Frierson HF, Zaika A, Powell SM, Roche J, Crowe S, Moskaluk CA, El-Rifai W (2005) Expression of tight-junction protein claudin-7 is an early event in gastric tumorigenesis. Am J Pathol 167:577–584
Kaneko Y, Lindsay LA, Murphy CR (2008) Focal adhesions disassemble during early pregnancy in rat uterine epithelial cells. Reprod Fertil Dev 20:892–899
Kaneko Y, Lecce L, Murphy CR (2009) Ovarian hormones regulate expression of the focal adhesion proteins, talin and paxillin, in rat uterine luminal but not glandular epithelial cells. Histochem Cell Biol 132:613–622
Kaneko Y, Lecce L, Day ML, Murphy CR (2011) Beta(1) and beta(3) integrins disassemble from basal focal adhesions and beta(3) integrin is later localised to the apical plasma membrane of rat uterine luminal epithelial cells at the time of implantation. Reprod Fertil Dev 23:481–495
Kennedy TG, Gillio-Meina C, Phang SH (2007) Prostaglandins and the initiation of blastocyst implantation and decidualization. Reproduction 134:635–643
Li Q, Wang J, Armant DR, Bagchi MK, Bagchi IC (2002) Calcitonin down-regulates E-cadherin expression in rodent uterine epithelium during implantation. J Biol Chem 277:46447–46455
Li WY, Huey CL, Yu AS (2004) Expression of claudin-7 and -8 along the mouse nephron. Am J Physiol Renal Physiol 286:F1063–F1071
Lopata A (1996) Blastocyst-endometrial interaction: an appraisal of some old and new ideas. Mol Hum Reprod 2:519–525
Markov AG, Veshnyakova A, Fromm M, Amasheh M, Amasheh S (2010) Segmental expression of claudin proteins correlates with tight junction barrier properties in rat intestine. J Comp Physiol [B] 180:591–598
Matter K, Balda MS (2003) Signalling to and from tight junctions. Nat Rev Mol Cell Biol 4:225–236
Mendoza-Rodriguez CA, Gonzalez-Mariscal L, Cerbon M (2005) Changes in the distribution of ZO-1, occludin, and claudins in the rat uterine epithelium during the estrous cycle. Cell Tissue Res 319:315–330
Murphy CR (2000a) Junctional barrier complexes undergo major alterations during the plasma membrane transformation of uterine epithelial cells. Hum Reprod 15(Suppl 3):182–188
Murphy CR (2000b) The plasma membrane transformation of uterine epithelial cells during pregnancy. J Reprod Fertil Suppl 55:23–28
Murphy CR (2004) Uterine receptivity and the plasma membrane transformation. Cell Res 14:259–267
Murphy CR, Rogers AW (1981) Effects of ovarian hormones on cell membranes in the rat uterus. III. The surface carbohydrates at the apex of the luminal epithelium. Cell Biophys 3:305–320
Murphy CR, Shaw TJ (1994) Plasma membrane transformation: a common response of uterine epithelial cells during the peri-implantation period. Cell Biol Int 18:1115–1128
Nicholson MDO, Lindsay LA, Murphy CR (2010) Ovarian hormones control the changing expression of claudins and occludin in rat uterine epithelial cells during early pregnancy. Acta Histochem 112:42–52
Preston AM, Lindsay LA, Murphy CR (2006) Desmosomes in uterine epithelial cells decrease at the time of implantation: an ultrastructural and morphometric study. J Morphol 267:103–108
Psychoyos A (1976) Hormonal control of uterine receptivity for nidation. J Reprod Fertil Suppl 25:17–28
Psychoyos A (1986) Uterine receptivity for nidation. Ann N Y Acad Sci 476:36–42
Robertson SA (2007) Seminal fluid signaling in the female reproductive tract: lessons from rodents and pigs. J Anim Sci 85:E36–E44
Short RV, Yoshinaga K (1967) Hormonal influences on tumor growth in the uterus of the rat. J Reprod Fertil 14:287–293
Swingle WW, Seay P, Perlmutt J, Collins EJ, Barlow G Jr, Fedor EJ (1951) An experimental study of pseudopregnancy in rat. Am J Physiol 167:586–592
Tatum R, Zhang Y, Salleng K, Lu Z, Lin JJ, Lu Q, Jeansonne BG, Ding L, Chen YH (2010) Renal salt wasting and chronic dehydration in claudin-7-deficient mice. Am J Physiol Renal Physiol 298:F24–F34
Tinel H, Denker HW, Thie M (2000) Calcium influx in human uterine epithelial RL95-2 cells triggers adhesiveness for trophoblast-like cells. Model studies on signalling events during embryo implantation. Mol Hum Reprod 6:1119–1130
Tsukita S, Furuse M (2000) The structure and function of claudins, cell adhesion molecules at tight junctions. Ann N Y Acad Sci 915:129–135
Turksen K, Troy TC (2004) Barriers built on claudins. J Cell Sci 117:2435–2447
Van Winkle LJ, Campione AL, Webster DP (1983) Sodium ion concentrations in uterine flushings from “implanting” and “delayed implanting” mice. J Exp Zool 226:321–324
Wang X, Matsumoto H, Zhao X, Das SK, Paria BC (2004) Embryonic signals direct the formation of tight junctional permeability barrier in the decidualizing stroma during embryo implantation. J Cell Sci 117:53–62
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Poon, C.E., Madawala, R.J., Day, M.L. et al. Claudin 7 is reduced in uterine epithelial cells during early pregnancy in the rat. Histochem Cell Biol 139, 583–593 (2013). https://doi.org/10.1007/s00418-012-1052-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-012-1052-y