Abstract
Purpose
To evaluate the efficacy and safety profile of a triplet regimen consisting of gemcitabine, oxaliplatin, and infusional fluorouracil and leucovorin (LV) in advanced pancreatic carcinoma (APC).
Patients and methods
Chemotherapy-naïve patients with histo-/cytologically proven unresectable APC, and bi-dimensionally measurable diseases were eligible. Treatment consisted of fixed-dose rate (10 mg/m2/min) infusion of 800 mg/m2 gemcitabine followed by 2-h infusion of 85 mg/m2 oxaliplatin and then 48-h infusion of fluorouracil and LV (3,000 and 300 mg/m2, respectively) every 2 weeks (the GOFL regimen). The primary end-point was objective response rate.
Results
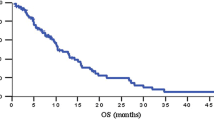
Forty-five patients were enrolled and received a median of seven [95% confidence interval (CI) 6.4–8.8] cycles of treatment. On intent-to-treat analysis, the overall response and disease-control rates were 33.3% (95% CI 21.4–48.0%) and 68.9% (95% CI 54.8–83.0%), respectively. Clinical benefit response was observed in 46.2% of initially symptomatic patients. The median time-to-tumor progression and overall survival were 5.1 (95% CI 4.0–6.3) months and 8.7 (95% CI, 6.1–11.3) months, respectively. Major grade 3–4 toxicities were neutropenia (28.9%, with 4.4% complicated with fever), peripheral sensory neuropathy (15.6%), nausea/vomiting (13.3%), and diarrhea (6.7%).
Conclusions
The triplet regimen is feasible and exhibits promising activity against APC, deserving further exploration.


Similar content being viewed by others
References
Palmer KR, Kerr M, Knowles G et al (1994) Chemotherapy prolongs survival in inoperable pancreatic carcinoma. Br J Surg 81(6):882–885
Glimelius B, Hoffman K, Sjoden PO et al (1996) Chemotherapy improves survival and quality of life in advanced pancreatic and biliary cancer. Ann Oncol 7(6):593–600
Burris HA, Moore MJ, Andersen J et al (1997) Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol 15(6):2403–2413
Heinemann V, Hinke A, Bock S et al (2007) Gemcitabine-based combinations (gem + x) vs Gemcitabine (gem) alone in the treatment of advanced pancreatic cancer: meta-analysis of sixteen randomized trials. J Clin Oncol 25(18S):4515
Shiah HS, Cheng AL, Hsu C et al (2006) Phase I–II trial of weekly gemcitabine plus high-dose 5-fluorouracil and leucovorin in advanced pancreatic cancer. J Gastroenterol Hepatol 21(3):531–536
Berlin JD, Adak S, Vaughn DJ et al (2000) A phase II study of gemcitabine and 5-fluorouracil in metastatic pancreatic cancer: an Eastern Cooperative Oncology Group Study (E3296). Oncology 58(3):215–218
Oettle H, Arning M, Pelzer U et al (2000) A phase II trial of gemcitabine in combination with 5-fluorouracil (24-hour) and folinic acid in patients with chemonaive advanced pancreatic cancer. Ann Oncol 11(10):1267–1272
Andre T, Noirclerc M, Hammel P et al (2004) Phase II study of leucovorin, 5-fluorouracil and gemcitabine for locally advanced and metastatic pancreatic cancer (FOLFUGEM 2). Gastroenterol Clin Biol 28(8–9):645–650
Raymond E, Buquet-Fagot C, Djelloul S et al (1997) Antitumor activity of oxaliplatin in combination with 5-fluorouracil and the thymidylate synthase inhibitor AG337 in human colon, breast and ovarian cancers. Anticancer Drugs 8(9):876–885
Faivre S, Raymond E, Woynarowski JM et al (1999) Supraadditive effect of 2′, 2′-difluorodeoxycytidine (gemcitabine) in combination with oxaliplatin in human cancer cell lines. Cancer Chemother Pharmacol 44:117–123
Louvet C, Labianca R, Hammel P et al (2005) Gemcitabine in combination with oxaliplatin compared with gemcitabine alone in locally advanced or metastatic pancreatic cancer: results of a GERCOR and GISCAD phase III trial. J Clin Oncol 23(15):3509–3516
Demols A, Peeters M, Polus M et al (2006) Gemcitabine and oxaliplatin (GEMOX) in gemcitabine refractory advanced pancreatic adenocarcinoma: a phase II study. Br J Cancer 94(4):481–485
Ghosn M, Farhat F, Kattan J et al (2007) FOLFOX-6 combination as the first-line treatment of locally advanced and/or metastatic pancreatic cancer. Am J Clin Oncol 30(1):15–20
Gebbia V, Maiello E, Giuliani F et al (2007) Second-line chemotherapy in advanced pancreatic carcinoma: a multicenter survey of the Gruppo Oncologico Italia Meridionale on the activity and safety of the FOLFOX4 regimen in clinical practice. Ann Oncol 18(6S):vi124–127
Oettle H, Pelezer U, Stieler J (2005) Oxaliplatin/folinic acid/5-fluorouracil [24 h] (OFF) plus best supportive care versus best supportive care alone (BSC) in second-line therapy of gemcitabine-refractory advanced pancreatic cancer (CONKO 003). J Clin Oncol 23(16S):4031
Ch’ang HJ, Wang CC, Cheng AL et al (2006) Phase I study of biweekly gemcitabine followed by oxaliplatin and simplified 48-h infusion of fluorouracil/leucovorin for advanced pancreatic cancer. J Gastroenterol Hepatol 21(5):874–879
Trotti A, Byhardt R, Stetz J et al (2000) Common toxicity criteria: version 2.0. An improved reference for grading the acute effects of cancer treatment: impact on radiotherapy. Int J Radiat Oncol Biol Phys 47(1):13–47
Miller AB, Hoogstraten B, Staquet M et al (1981) Reporting results of cancer treatment. Cancer 47(1):207–214
Kaplan EL, Meier P (1958) Nonparametric estimation from incomplete observation. J Am Stat Assoc 53:457–481
Riess H, Helm A, Niedergethmann M et al (2005) A randomized, prospective, multicenter, phase II trial of gemcitabine, 5-fluorouracil (5-FU), folinic acid vs. gemcitabine alone in patients with advanced pancreatic cancer. J Clin Oncol 23(16S):4009
Poplin E, Levy DE, Berlin J et al (2006) Phase III trial of gemcitabine (30-minute infusion) versus gemcitabine (fixed-dose-rate infusion [FDR]) versus gemcitabine + oxaliplatin (GEMOX) in patients with advanced pancreatic cancer (E6201). J Clin Oncol 24(18S):4004
Ducreux M, Mitry E, Ould-Kaci M et al (2004) Randomized phase II study evaluating oxaliplatin alone, oxaliplatin combined with infusional 5-FU, and infusional 5-FU alone in advanced pancreatic carcinoma patients. Ann Oncol 15(3):467–473
Pelzer U, Kubica K, Stieler J et al (2008) A randomized trial in patients with gemcitabine refractory pancreatic cancer. Final results of the CONKO003 study. J Clin Oncol 26(15S):4508
Garnier C, Rebischung C, Chirpaz E et al (2001) Phase II study of a combination with leucovorin (LV), 5 FU bolus and infusion (FU), gemcitabine (GEM) and oxaliplatin (LOHP) (FOLFU GEMOX regimen) in locally advanced (LA) and metastatic (M) pancreatic carcinoma (APC). Proc Am Soc Clin Oncol 20:620
Conroy T, Paillot B, Francois E et al (2005) Irinotecan plus oxaliplatin and leucovorin-modulated fluorouracil in advanced pancreatic cancer—a Groupe Tumeurs Digestives of the Federation Nationale des Centres de Lutte Contre le Cancer study. J Clin Oncol 23(6):1228–1236
Ychou M, Desserigne F, Guimbaud R et al (2007) Randomized phase II trial comparing folfirinox (5FU/leucovorin [LV], irinotecan [I] and oxaliplatin [O]) vs gemcitabine (G) as first-line treatment for metastatic pancreatic adenocarcinoma (MPA). First results of the ACCORD 11 trial. J Clin Oncol 25(18S):4516
Verslype C, Van Cutsem E, Dicato M et al (2006) The management of pancreatic cancer. Current expert opinion and recommendations derived from the 8th World Congress on Gastrointestinal Cancer, Barcelona, 2006. Ann Oncol 18(7S):vii1–10
Huguet F, Andre T, Hammel P et al (2007) Impact of chemoradiotherapy after disease control with chemotherapy in locally advanced pancreatic adenocarcinoma in GERCOR phase II and III studies. J Clin Oncol 25(3):326–331
Rana V, Krishnan S, Abbruzzese JL et al (2006) Neoadjuvant chemotherapy improves outcomes of chemoradiation therapy for locally advanced pancreatic cancer. J Clin Oncol 24(18S):4036
Chen LT, Ch’ang HJ, Huang CL et al (2008) Safety-profile of gemcitabine-based chemoradiotherapy (CRT) after triplet induction chemotherapy (CT) in locally advanced pancreatic cancer (LAPC): preliminary data from Taiwan Cooperative Oncology Group (TCOG) T1204 study. In: ASCO Gastrointestinal Cancers Symposium, Orlando; abstract 156
Acknowledgment
Supported by grants (CA094-PP-04 and CA095-CP-06) from the National Health Research Institutes, Zhunan, Taiwan.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article describes the triplet regimen, GOFL (Gemcitabine, Oxaliplatin, Fluorouracil, Leucovorin), being feasible and exhibiting promising activity against advanced pancreatic cancer. This phase II study revealed that GOFL might have better therapeutic efficacy and toxicity profile compared to the current standard gemcitabine monotherapy.
Employment/leadership position: None; Intellectual property rights/inventor/patent holder: None; Consultant/advisory role: None; Ownership interest: None; Expert testimony: None; Other: None.
Rights and permissions
About this article
Cite this article
Ch’ang, HJ., Huang, CL., Wang, HP. et al. Phase II study of biweekly gemcitabine followed by oxaliplatin and simplified 48-h infusion of 5-fluorouracil/leucovorin (GOFL) in advanced pancreatic cancer. Cancer Chemother Pharmacol 64, 1173–1179 (2009). https://doi.org/10.1007/s00280-009-0980-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-009-0980-2