Abstract
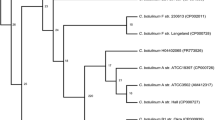
The phylogenetic interrelationships of members of theClostridium botulinum complex of species was investigated by direct sequencing of their 16S rRNA genes. Comparative analysis of the 16S rRNA sequences demonstrated the presence of four phylogenetically distinct lineages corresponding to: i) proteolyticC. botulinum types A, B, and F, andC. sporogenes, ii) saccharolytic types B, E and F, iii) types C and D andC. novyi type A, and iv) type G andC. subterminale. The phylogenetic groupings obtained from the 16S rRNA were in complete agreement with the four divisions recognised within the ‘species complex’ on the basis of phenotypic criteria.
Similar content being viewed by others
References
Aureli P, Fenicia L, Pasolini B, Gianfranceschi M, McCroskey LM & Hatheway CL (1986) Two cases of type E infant botulism in Rome caused by neurotoxigenicClostridium butyricum. J. Infect. Dis. 154: 207–211
Cato EP, George WL & Finegold SM (1986) GenusClostridium, p1141–1200.In: PHA Sneath, NS Mair, ME Sharpe & JG Holt (ed.), Bergey's manual of systematic bacteriology. The Williams & Wilkins Co., Baltimore.
Collins MD, Williams AM & Wallbanks S (1990) The phylogeny ofAerococcus andPediococcus as determined by 16S rRNA sequence analysis: description ofTetragenococcus gen.nov. FEMS Microbiol. Lett. 70: 225–262
Collins MD, Rodrigues UM, Dainty RH, Edwards RA & Roberts TA (1992) Taxonomic studies on a psychrophilicClostridium from vacuum-packed beef: Descriptionof Clostridium estertheticum sp.nov. FEMS Microbiol. Lett. 96: 235–240
Felsenstein J (1982) Numerical methods for inferring evolutionary trees. Quart. Rev. Biol. 57: 379–404
Fitch WM & Margoliash E (1967) construction of phylogenetic trees: a method based on mutation distances as estimated from cytochrome C sequences is of general applicability. Science 155: 279–284
Fox G, Wisotzkey JD & Jurtshuk Jr P (1992) How close is close: 16S rRNA sequence identity may not be sufficient to guarantee species identity. Int. J. Syst. Bacteriol. 42: 166–170
Hall JD, McCroskey LM, Pinkcomb BJ & Hatheway CL (1985) Isolation of an organism resemblingClostridium baratii which produced type F botulinal toxin from an infant with botulism. J. Clin. Microbiol. 15: 1133–1140
Holdeman LV & Brooks JB (1970) Variation among strains ofClostridium botulinum and related clostridia, p278–286.In M Herzberg (ed), Proceedings of the first U.S.-Japan Conference of Toxic Micro-organisms. U.S. Government Printing Office, Washington, D.C.
Hori H & Osawa S (1979) Evolutionary change in 5S rRNA secondary structure and phylogenetic tree of 54 5S rRNA species. Proc. Natl. Acad. Sci. USA 76: 381–385
Hutson RA, Thompson DE & Collins MD (1993) Genetic interrelationships of saccharolyticClostridium botulinum types B, E and F and related clostridia as revealed by small-subunit rRNA gene sequences. FEMS Microbiol. Lett. 108: 103–110
Johnson JL & Francis BS (1975) Taxonomy of the clostridia: Ribosomal ribonucleic acid homologies among the species. J. Gen. Microbiol. 88: 229–244
Lee WH & Riemann H (1970) The genetic relatedness of proteolyticClostridium botulinum strains. J. Gen. Microbiol. 64: 85–90
Martinez-Murcia AJ, Benlloch S & Collins MD (1992) Phylogenetic interrelationships of members of the generaAeromonas andPlesiomonas as determined by 16S ribosomal DNA sequencing: lack of congruence with results of DNA-DNA hybridizations. Int. J. Syst. Bacteriol. 42: 166–170
McCroskey LM, Hatheway CL, Fenicia L, Pasolini B & Aureli P (1986) Characterization of an organism that produces type E botulinal toxin which resemblesClostridium butyricum from the faeces of an infant with type E botulism. J. Clin. Microbiol. 23: 201–202
McCroskey LM, Hatheway CL, Woodruff BA, Greenberg JA & Jurgenson P (1991) Type F botulism due to neurotoxigenicClostridium barati from an unknown source in an adult. J. Clin. Microbiol. 29: 2618–2620
Nakamura S, Kinuira I, Yamakawa K & Nishida S (1983) Taxonomic relationships amongClostridium novyi types A and B,Clostridium haemolyticum andClostridium botulinum type C. J. Gen. Microbiol. 129: 1473–1479
Poulet S, Hauser D, Quanz M, Niemann H & Popoff MR (1992) Sequences of the botulinal neurotoxin E derived fromClostridium botulinum type E (strain beluga) andClostridium butyricum (strains ATCC43181 and ATCC43755). Biochem. Biophys. Res. Commun. 182: 107–113
Queen C & Korn LJ (1984) A comprehensive sequence analysis program for the IBM personal computer. Nucleic Acids Res. 12: 581–599
Smith LDS & Hobbs G (1974) Genus III.Clostridium Prazmowski 1880, 23.In: Bergey's Manual of Determinative Bacteriology, 8th edn, pp. 551–572. Edited by RE Buchanan & NE Gibbons. Baltimore — The Williams & Willins Co.
Suen JC, Hatheway CL, Steigerwalt AG & Brenner DJ (1988a) Genetic confirmation of identities of neurotoxigenicClostridium barati andClostridium butyricum implicated as agents of infant botulism. J. Clin. Microbiol. 26: 2191–2192
Suen JC, Hatheway CL, Steigerwalt AG & Brenner DJ (1988b)Clostridium argentinense sp.nov: A genetically homogeneous group composed of all strains ofClostridium botulinum toxin type G and some nontoxigenic strains previously identified asClostridium subterminale orclostridium hastiforme. Int. J. Syst. Bacteriol. 38: 375–381
Thompson DE, Hutson RA, East AK, Allaway D, Collins MD & Richardson PT (1993) Nucleotide sequence of the gene coding forClostridium barati type F Neurotoxin: Comparison with other Clostridial neurotoxins. FEMS Microbiol. Lett. 108: 175–182
Woese CR (1987) Bacterial evolution. Microbiol. Rev. 51: 221–271
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hutson, R.A., Thompson, D.E., Lawson, P.A. et al. Genetic interrelationships of proteolyticClostridium botulinum types A, B, and F and other members of theClostridium botulinum complex as revealed by small-subunit rRNA gene sequences. Antonie van Leeuwenhoek 64, 273–283 (1993). https://doi.org/10.1007/BF00873087
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00873087