Abstract
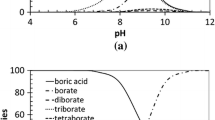
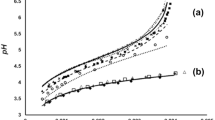
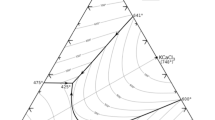
Buffer capacity studies, carried out using pH titrations, demonstrated that electrolytes containing NiSO4 + H3BO3 + Na2SO4 buffer more effectively than in the presence of H3BO3 + Na2SO4. The increased buffer capacity is dependent on the concentration of both Ni2+ and H3BO3. Thermodynamic analysis of the pH titration data suggested the formation of a weak complex, Ni(H2BO3)2, (logK ∼ 3.8 to 4.9) between Ni2+ and H3BO3 in mixed chloride-sulphate electrolytes at 55° C.
Similar content being viewed by others
References
A. L. Zhurin and M. G. Shoikhet,J. Appl. Chem. (U. S. S. R.) 29 (1958) 641.
N. Tanaka, Y. Saito and H. Ogino,Bull. Chem. Soc. Japan 36 (1963) 794.
J. A. Bolzan, E. A. Jauregui and A. J. Arvia,Electrochim. Acta 8 (1963) 841.
H. S. Harned and B. B. Owen, ‘The Physical Chemistry of Electrolyte Solutions’, Reinhold Publishing Corporation, New York (1950).
I. M. Kolthoff and W. Bosch,Rec. Trav. chim. 46 (1927) 180.
B. B. Owen and E. J. King,J. Amer. Chem. Soc. 65 (1943) 1612.
B. V. K. S. R. A. Tilak, S. R. Rajagopalan and A. K. N. Reddy,Trans. Faraday Soc. 58 (1962) 795.
D. M. Chizhikov and L. V. Pliginskaya,Soviet Electrochemistry 3 (1961) 69.
I. Epelboin, 21st Meeting of the International Committee on Thermodynamics and Electrochemical Kinetics, Prague (1970).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tilak, B.V., Gendron, A.S. & Mosoiu, M.A. Borate buffer equilibria in nickel refining electrolytes. J Appl Electrochem 7, 495–500 (1977). https://doi.org/10.1007/BF00616760
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00616760