Summary
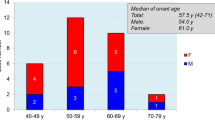
We examined 7 patients with Creutzfeldt-Jakob disease (CJD) with a methionine-to-valine change at prion protein (PrP) codon 129 (CJD129 patients). These CJD129 patients did not have either a condon 117 or 198 point mutation. For comparison, we also examined 7 patients with Gerstmann-Sträussler syndrome (GSS) with a proline-to-leucine change at PrP codon 102 (GSS102 patients) and 13 patients without any known mutations at codons 102, 117, 129, 178, or 200 (CJDwild patients). CJD129 patients had a long clinical duration and ataxia at onset, but rarely had any periodic synchronous discharge in their electroencephalogram. Unlike CJDwild patients, all CJD129 patients have typical congophilic PrP plaques in their brain. These clinicopathological findings were similar to those of GSS102. However, the distribution and morphology of PrP deposits revealed by immunohistochemistry were different between CJD129 and GSS102. In GSS102 more numerous and various types of PrP plaques are seen throughout the brain, while in CJD129 patients a unicentric core was the major feature of PrP plaques. The change in codon 129 influences the clinical course and pathological findings in CJD.
Similar content being viewed by others
References
Doh-ura K, Tateishi J, Sasaki H, Kitamoto T, Sakaki Y (1989) Pro-Leu change at position 102 of prion protein is the most common but not the sole mutation related to Gerstmann-Sträussler syndrome. Biochem Biphys Res Commun 163:974–979
Doh-ura K, Tateishi J, Kitamoto T, Sasaki H, Sakaki Y (1990) Creutzfeldt-Jakob disease patients with congophilic kuru plaques have the missense variant protein common to Gerstmann-Sträussler syndrome. Ann Neurol 27:121–126
Doh-ura K, Kitamoto T, Sakaki Y, Tateishi J (1991) CJD discrepancy. Nature 353:801–802
Goldfarb I., Brown P, Goldgaber D, Asher DM, Strass N, Graupera G, Piccardo P, Brown WT, Rubinstein R, Boellaard JW, Gajdusek DC (1989) Patients with Creutzfeldt-Jakob disease and kuru lack the mutation in the PRIP gene found in Gerstmann-Sträussler syndrome, but they show a different double-allele mutation in the same gene. Am J Hum Genet 45 [Suppl]:A189
Goldgaber D, Goldfarb LG, Brown P, Asher DM, Brown WT, Lin S, Teener JW, Feinstone SM, Rubenstein R, Kascsak RJ, Boellaard JW, Gajdusek C (1989) Mutations in familial Creutzfeldt-Jakob disease and Gerstmann-Sträussler syndrome. Exp Neurol 106:204–206
Guesdon J, Ternynck T, Avrameas S (1979) The use of avidin-biotin interaction in immunoenzymatic techniques. J Histochem Cytochem 27:1131–1139
Hsiao K, Baker HF, Crow TJ, Poutler M, Owen F, Terwillinger JD, Westaway D, Ott J, Prusiner SB (1989) Linkage of a prion protein missense variant to Gerstmann-Sträussler syndrome. Nature 338:342–345
Hsiao KK, Scott M, Foster D, Groth DF, DeArmond SJ, Prusiner SB (1990) Spontaneous neurodegeneration in transgenic mice with mutant prion protein of Gerstmann-Sträussler syndrome. Science 250:1587–1590
Kitamoto T, Tateishi J (1988) Immunohistochemical confirmation of Creutzfeldt-Jakob disease with a long clinical course with amyloid plaque core antibodies. Am J Pathol 131:435–443
Kitamoto T, Tateishi J, Tashima T, Takeshita I, Barry RA, DeArmond SJ, Prusiner SB (1986) Amyloid plaques in Creutzfeldt-Jakob disease stain with prion protein antibodies. Ann Neurol 20:204–208
Kitamoto T, Ogomori K, Tateishi J, Prusiner SB (1987) Formic acid pretreatment enhances immunostaining of cerebral and systemic amyloids. Lab Invest 57:230–236
Kitamoto T, Muramoto T, Mohri S, Doh-ura K, Tateishi J (1991) Abnormal isoform of prion protein accumulates in follicular dendritic cells in mice with Creutzfeldt-Jakob disease. J Virol 65:6292–6295
Kitamoto T, Muramoto T, Hilbich C, Beyreuther K, Tateishi J (1991) N-Terminal sequence of prion protein is also integrated into kuru plaques in patients with Gerstmann-Sträussler syndrome. Brain Res 545:319–321
Kitamoto T, Yamaguchi K, Doh-ura K, Tateishi J (1991) A prion protein missense variant is integrated in kuru plaque cores in patients with Gerstmann-Sträussler syndrome. Neurology 41:306–310
Kitamoto T, Shin R-W, Doh-ura K, Tomokane N, Miyazono M, Muramoto T, Tateishi J (1992) Abnormal isoform of prion protein accumulates in the synaptic structures of the central nervous system in patients with Creutzfeldt-Jakob disease. Am J Pathol 140:1285–1294
Kretzschmar HA, Stowring LE, Westaway D, Stubblebin WH, Prusiner SB, DeArmond SJ (1986) Molecular cloning of a human prion protein cDNA. DNA 5:315–324
Masters C, Gajdusek DC, Gibbs CJ Jr (1981) Creutzfeldt-Jakob disease virus isolations from the Gerstmann-Sträussler syndrome: with an analysis of the various forms of amyloid plaque deposition in the virus-induced spongiform encephalopathy. Brain 104:559–588
Miyazono M, Iwaki T, Kitamoto T, Kaneko Y, Doh-ura K, Tateishi J (1991) A comparative immunohistochemical study of kuru and senile plaques with a special reference to glial reactions at various stages of amyloid plaque formation. Am J Pathol 139:589–598
Owen F, Poulter M, Collinge J, Crow TJ (1990) Codon 129 changes in the prion protein gene in Caucasians. Am J Hum Genet 46:1215–1216
Palmer MS, Dryden AJ, Hughes JT, Collinge J (1991) Homozygous prion protein genotype predisposes to sporadic Creutzfeldt-Jakob disease. Nature 352:340–342
Prusiner SB (1982) Novel proteinaceous infectious particles cause scrapie. Science 216:136–144
Prusiner SB (1991) Molecular biology of prion diseases. Science 252:1515–1522
Tateishi J, Kitamoto T, Hashiguchi H, Shii H (1988) Gerstmann-Sträussler-Scheinker disease: immunohistological and experimental studies. Ann Neurol 24:35–40
Author information
Authors and Affiliations
Additional information
Supported in part by a Grant-in-Aid for Scientific Research (02454245, 03454171) from the Ministry of Education, Science and Culture of Japan, and Foundation for Advancement of Clinical Medicine
Rights and permissions
About this article
Cite this article
Miyazono, M., Kitamoto, T., Doh-ura, K. et al. Creutzfeldt-Jakob disease with codon 129 polymorphism (Valine): a comparative study of patients with codon 102 point mutation or without mutations. Acta Neuropathol 84, 349–354 (1992). https://doi.org/10.1007/BF00227660
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00227660