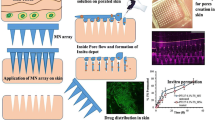
Abstract
Polymeric microneedles were prepared with Polyvinyl Pyrrolidone (PVP) K-30 using the mold casting technique. The core microneedles were coated with Eudragit E-100 by dip and spin method. The amount of 5-fluorouracil (FU) loaded in the core microneedles was 604 ± 35.4 µg. The coating thickness was 24.12 ± 1.12 µm. The objective was to deliver the 5-FU gradually in a controlled release manner at the target site in the sub-stratum corneum layer. This approach is anticipated to improve the safety and efficacy of topical melanoma treatment. The release of the drug was prolonged for up to 3 h from the polymer-coated polymeric (PCP) microneedles. The entire amount was found to release within 15 min in uncoated MNs. Likewise, the permeation of the drug from the uncoated microneedles was rapid, whereas the PCP microneedles were able to prolong the permeation up to 420 min. The PCP microneedles were subjected to stability studies at 25°C ± 2°C/60%RH, and 40°C ± 2°C/75%RH condition for 3 months. The formulations were found intact, and the release rate was not significantly different form the fresh formulation. The drug content was found to meet the acceptability criteria as well (98.12 ± 1.8% and 97.8 ± 2.1% at 25 and 40°C respectively after 3 months). Overall, this study demonstrated the feasibility of fabrication of PCP microneedles using Eudragit E100 for intraregional controlled delivery of drugs.
Graphical Abstract





Similar content being viewed by others
References
Perera E, Gnaneswaran N, Jennens R, Sinclair R. Malignant melanoma. Healthcare (Basel). 2013;2:1–19.
Slominski RM, Zmijewski MA, Slominski AT. The role of melanin pigment in melanoma. Exp Dermatol Exp Dermatol. 2015;24:258–9.
Singh AK. FRCP MB, and Loscalzo J. The Brigham Intensive Review of Internal Medicine: Elsevier; 2022.
Sboner A, Eccher C, Blanzieri E, Bauer P, Cristofolini M, Zumiani G, and Forti S. A multiple classifier system for early melanoma diagnosis. 2003;27(1):29–44.
Thompson JF, Scolyer RA, Kefford RF. Cutaneous melanoma. Lancet. 2005;365:687–701.
Beddingfield FC. The Melanoma Epidemic. Res Ipsa Loquitur The Oncologist. 2003;8(5):459–65.
Zbytek B, Carlson JA, Granese J, Ross J, Mihm M, Slominski A. Current concepts of metastasis in melanoma. Expert Rev Dermatol. 2008;3:569.
Pawlik TM, Ross MI, Johnson MM, Schacherer CW, McClain DM, Mansfield PF. Predictors and natural history of in-transit melanoma after sentinel lymphadenectomy. Ann Surg Oncol. 2005;12:587–96.
Ryan RF, Krementz ET, Litwin MS. A role for topical 5-fluorouracil therapy in melanoma. J Surg Oncol. 1988;38:250–6.
Litwin MS, Krementz ET, Mansell PW, Reed RJ. Topical chemotherapy of lentigo maligna with 5-fluorouracil. Cancer. 1975;35(3):721–33.
Kasprzak JM, and Xu YG. Diagnosis and management of lentigo maligna: a review. Drugs Context. 2015;4.
Basal & Squamous Cell Local Treatment | Skin Cancer Local Treatments. https://www.cancer.org/cancer/basal-and-squamous-cell-skin-cancer/treating/other-than-surgery.html.
Werschler WP. Considerations for use of fluorouracil cream 0.5% for the treatment of actinic keratosis in elderly patients. J Clin Aesthet Dermatol. 2008;1:22.
Tuan-Mahmood TM, McCrudden MTC, Torrisi BM, McAlister E, Garland MJ, Singh TRR. Microneedles for intradermal and transdermal delivery. Eur J Pharm Sci. 2013;50:623.
Kim YC, Park JH, Prausnitz MR. Microneedles for drug and vaccine delivery. Adv Drug Deliv Rev. 2012;64:1547.
Hong X, Wei L, Wu F, Wu Z, Chen L, Liu Z. Dissolving and biodegradable microneedle technologies for transdermal sustained delivery of drug and vaccine. Drug Des Devel Ther [Internet]. Drug Des Devel Ther. 2013;7:945–52.
Gorzelanny C, Mess C, Schneider SW, Huck V, Brandner JM. Skin barriers in dermal drug delivery: which barriers have to be overcome and how can we measure them? Pharmaceutics. 2020;12:1–31.
Shukla A, Singh AP, Maiti P. Injectable hydrogels of newly designed brush biopolymers as sustained drug-delivery vehicle for melanoma treatment. Signal Transduct Target Ther. 2021;6:63.
Maurya A, Nanjappa SH, Honnavar S, Salwa M, Murthy SN. Rapidly dissolving microneedle patches for transdermal iron replenishment therapy. J Pharm Sci. 2018;107:1642–7.
Jakka D, Matadh AV, Shankar VK, Shivakumar HN, Murthy SN. Polymer coated polymeric (PCP) microneedles for controlled delivery of drugs (dermal and intravitreal). J Pharm Sci. 2022;111(10):2867–78.
Jakka D, Matadh AV, Shivakumar HN, Maibach H, Murthy SN. Polymer Coated Polymeric (PCP) microneedles for sampling of drugs and biomarkers from tissues. Eur J Pharm Sci. 2022;175: 106203.
Hansova B, Synek S, Opatrilova R. 5-fluorouracil–characteristics and analytical determination. Curr Pharm Anal. 2011;7:1–11.
Breda M, Barattè S. A review of analytical methods for the determination of 5-fluorouracil in biological matrices. Anal Bioanal Chem. 2010;397:1191–201.
Lahiji SF, Dangol M, Jung H. A patchless dissolving microneedle delivery system enabling rapid and efficient transdermal drug delivery. Sci Reports. 2015;5:1–7.
Nitin G, Zahid Z. Formualtion and development of mucoadhesive sustained release buccal tablets and patches of 5-fluorouracil using different polymers. Asian J Pharm Clin Res. 2018;11:5.
Youssef WN, Amit K, Zeru C. The effect of microneedles on the skin permeability and antitumor activity of topical 5-fluorouracil. Acta Pharmaceutica Sinica B. 2014;4(1):94–9.
Singh P, Tyagi G, Mehrotra R, Bakhshi AK. Thermal stability studies of 5-fluorouracil using diffuse reflectance infrared spectroscopy. Drug Test Anal. 2009;1(5):240–4.
Senapati S, Mahanta AK, Kumar S, Maiti P. Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct Target Ther. 2018;3.
Meng F, Hasan A, Mahdi Nejadi Babadaei M, Hashemi Kani P, Jouya Talaei A, and Sharifi M. Polymeric-based microneedle arrays as potential platforms in the development of drugs delivery systems. J Adv Res. 2020;26:137.
Ahmed Saeed AL-JK, Mahmood S, Hamed AS, Reddy VJ, Rebhi HA, Azmana M. Current trends in polymer microneedle for transdermal drug delivery. Int J Pharm. 2020;587:119673.
Makadia HK, Siegel SJ. Poly Lactic-co-Glycolic Acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers (Basel). 2011;3:1377.
Funding
Authors declare that there were no external fundings received for the submitted research article.
Author information
Authors and Affiliations
Contributions
Conceptualization: S Narasimha Murthy, Shivakumar H N; methodology: Anusha V Matadh, Deeksha Jakka; formal analysis and investigation: Anusha V Matadh, Deeksha Jakka, Pragathi SG; writing—original draft preparation: S Narasimha Murthy, Anusha V Matadh, Deeksha Jakka; writing—review and editing: Howard Maibach, S Narasimha Murthy, Shivakumar H N, Srinath Rangappa, Reena N. M; supervision and advise: Howard Maibach.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Matadh, A.V., Jakka, D., Pragathi, S.G. et al. Polymer-Coated Polymeric (PCP) Microneedles for Controlled Dermal Delivery of 5-Fluorouracil. AAPS PharmSciTech 24, 9 (2023). https://doi.org/10.1208/s12249-022-02471-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-022-02471-x