Abstract
Background
Diabetic nephropathy (DN) is a serious consequence of diabetes mellitus (DM), and it is linked to higher morbidity and mortality in diabetic patients. The quest for cheap therapeutic strategy with lesser side effects remains a major health concern. However, Dryopteris dilatata is a commonly found flavonoid-rich plant with plethora of therapeutic potentials. This study investigated the effect of methanol extract of D. dilatata (MEDd) on streptozotocin-induced diabetic nephropathy in male Wistar rat.
Methods
Animals were randomly selected into five groups (n = 5) and were treated as follows; group 1 received distilled water (10 mL/kg), group 2 received only STZ (60 mg/kg), groups 3 and 4 received STZ then 400 and 800 mg/kg of MEDd, respectively, while group 5 received STZ then pioglitazone (10 mg/kg). Following 14 days of treatment, animals were euthanized, and blood as well as pancreas and kidney tissues were collected for further studies.
Results
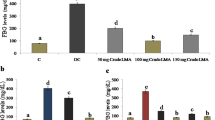
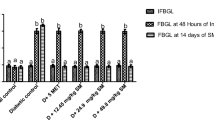
Our results revealed that MEDd significantly reduced STZ-induced hyperglycemia in diabetic rats. Markers of oxidative injury (MDA, nitrite, and GSH) were also significantly ameliorated in the pancreas and kidney of the diabetic rats following treatment with MEDd.
However, renal function markers (creatinine and urea) were significantly attenuated with marked decreased in organ weight in the diabetic rats after treatment with MEDd. Also, serum insulin and corticosterone levels were restored following MEDd treatment.
Conclusion
Methanol extract of D. dilatata demonstrated anti-diabetogenic and reno-protective potential by enhancing in vivo reno-pancreatic antioxidant defense system.














Similar content being viewed by others
Data availability
This manuscript includes all of the study’s data.
References
Valencia WM, Florez H. “How to prevent the microvascular complications of type 2 diabetes beyond glucose control. BMJ. 2017;356:i6505.
Burrows NR, Hora I, Geiss LS, Gregg EW, Albright AA. “Incidence of end-stage renal disease attributed to diabetes among persons with diagnosed diabetes—United States and Puerto Rico, 2000–2014”, MMWR. Morb Mortal Wkly Rep. 2017;66:1165–70.
Zhang L, Long J, Jiang W, et al. Trends in chronic kidney disease in China. N Engl J Med. 2016;375:905–6.
Xue R, Gui D, Zheng L, Zhai R, Wang F, Wang N. Mechanistic insight and management of diabetic nephropathy: recent progress and future perspective. J Diabetes Res. 2017.
Stenvinkel P. Chronic kidney disease: a public health priority and harbinger of premature cardiovascular disease”. J Intern Med. 2010;268:456–67.
Gheith O, Farouk N, Nampoory N, Halim MA, Al- Otaibi T. Diabetic kidney disease: worldwide difference of prevalence and risk factors. J Nephropharmacol. 2015;5:49–56.
Susztak K, Böttinger EP. Diabetic nephropathy: a frontier for personalized medicine”. J Am Soc Nephrol. 2006;17:361–7.
Fioretto P, Steffes MW, Mauer M. Glomerular structure in nonproteinuric IDDM patients with various levels of albuminuria”. Diabetes. 1994;43:1358–64.
Caramori ML, Kim Y, Huang C, et al. Cellular basis of diabetic nephropathy: 1 study design and renal structuralfunctional relationships in patients with long-standing Type 1 diabetes. Diabetes. 2002;51(2):506–13.
Perkins BA, Ficociello LH, Silva KH, Finkelstein DM, Warram JH, Krolewski AS. Regression of microalbuminuria in type 1 diabetes”. N Engl J Med. 2003;348:2285–93.
Trevisan R, Vedovato M, Mazzon C, et al. Concomitance of diabetic retinopathy and proteinuria accelerates the rate of decline of kidney function in type 2 diabetic patients”. Diabetes Care. 2002;25:2026–31.
Kramer HJ, Nguyen QD, Curhan G, Hsu C. Renal insufficiency in the absence of albuminuria and retinopathy 10 BioMed Research International among adults with type 2 diabetes mellitus”. JAMA. 2003;289:3273–7.
Chen Y, Lee K, Ni Z, He JC. Diabetic kidney disease: challenges, advances, and opportunities”. Kidney Dis. 2020;6(4):215–25.
Arora MK, Singh UK. Molecular mechanisms in the pathogenesis of diabetic nephropathy: an update”. Vascul Pharmacol. 2013;58:259–71.
Kopel J, Pena-Hernandez C, Nugent K. Evolving spectrum of diabetic nephropathy”. World J Diabetes. 2019;10:269–79.
Tavafi M. Diabetic nephropathy and antioxidants”. J Nephropathol. 2013;2:20–7.
Donate-Correa J, Luis-Rodríguez D, Martín-Núñez E, et al. Inflammatory targets in diabetic nephropathy”. J Clin Med. 2020;9(2):458.
Thallas-Bonke V, Thorpe SR, Coughlan MT, et al. Inhibition of NADPH oxidase prevents advanced glycation end product-mediated damage in diabetic nephropathy through a protein kinase C- -dependent pathway”. Diabetes. 2008;57(2):460–9.
Kao MP, Ang DS, Pall A, Struthers AD. Oxidative stress in renal dysfunction: mechanisms, clinical sequelae and therapeutic options. J Hum Hypertens. 2010;24(1):1–8.
Rünk K, Zobel M, Zobel K. Biological Flora of the British Isles: Dryopteris carthusiana, D. dilatata and D. expansa. J Ecol. 2012;100(4):1039–63.
Mordi JC, Lawrence EO, Chiedozie O. queous Leaf Extract of Dryopteris dilatata on STZ--Induced Diabetic Wistar Rats with Associated Hyperlipidemic Ameliorating Property. Journal of Dental and Medical Sciences. 2016;15(6):97–104.
Akpotu A, Celestine A, Choice N, Okorie P, Igwe U, Jide U, Adeyemo M, Nwaeme O, Obinna O. Antidiabetic and anti-hyperlipidemic effects of ethanolic extract of Dryopteris dilatata leaves. Journal of Diabetes and Endocrinology., 2018;9(3):20–7.
Alawode DI, Asiwe JN, Moke EG, Okonofua DE, Sanusi KO, Adagbada EO, Yusuf MO, Fasanmade AA. The effect of ethanol leaf extract of Cnidosculus Aconitifolius on cardiorenal functions in hypertensive and normotensive male Wistar rats. Int J Nutr Sci. 2021;6(3):155–60. https://doi.org/10.30476/IJNS.2021.92067.1145.
Asiwe JN, Anachuna KK, Moke EG, Sanusi KO, Okonofua DE, Omeru O, Fasanmade AA. High dietary salt intake alleviates fasting blood glucose in streptozotocin-induced diabetic male Wistar rats. Thai J Pharm Sci. 2021;45(3):172–7.
Akpotu EA, Ghasi SI, Ewhre LO, Adebayo OG, Asiwe JN. Anti-diabetogenic and in vivo antioxidant activity of ethanol extract of Dryopterisdilatata in alloxan-induced male Wistar rats. Biomarkers. 2021;26(8):718–25.
McCord JM, Fridovich I. Superoxide dismutase. An enzymic function for erythrocuprein (hemocuprein). J Biol Chem. 1969;244:6049–55.
Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979;95:351–8.
Reitman S, Frankel S. Glutamic-pyruvate transaminase assay by colorimetric method. Am J Clin Path. 1957;28:56.
Klil-Drori AJ, Azoulay L, Pollak MN. Cancer, obesity, diabetes, and antidiabetic drugs: is the fog clearing? Nat Rev Clin Oncol. 2017;14(2):85–99.
Anachuna KK, Oyem CJ, Nwogueze BC, Asiwe JN. Glucose lowering effects and histomorphological changes of Vernonia amygdalina on pancreatic compromised Wistar rats using alloxan monohydrate. Trop J Health Sci. 2018;25(2):27–31.
Ajayi AM, Adedapo ADA, Badaki VB, Oyagbemi AA, Adedapo AA. Chrysophyllum albidum fruit ethanol extract ameliorates hyperglycaemia and elevated blood pressure in streptozotocin-induced diabetic rats through modulation of oxidative stress. NF-κB and PPAR-γ Biomed Pharmacother. 2021;141:111879. https://doi.org/10.1016/j.biopha.2021.111879.
Asiwe JN, Kolawole TA, Anachuna KK, Ebuwe EI, Nwogueze BC, Eruotor H, Igbokwe V. Cabbage juice protect against Lead-induced liver and kidney damage in male Wistar rat. Biomarkers. 2022;27(2):151–8. https://doi.org/10.1080/1354X.2021.2022210.
Vinson JA, Dabbagh YA, Serry MM, Jang J. Plant flavonoids, especially tea flavonols, are powerful antioxidants using an in vitro oxidation model for heart disease. J Agric Food Chem. 1995;43:2800–2.
Bayili RG, Abdoul-Latif F, Kone OH, Diao M, Bassole IH, Dicko MH. Phenolic compounds and antioxidant activities in some fruits and vegetables from Burkina Faso. Afr J Biotech. 2011;10(62):13543–7.
Oboh G, Ademosun AO, Akinleye M, Omojokun OS, Boligon AA, Athayde ML. Starch composition, glycemic indices, phenolic constituents, and antioxidative and antidiabetic properties of some common tropical fruits. Journal of Ethnic Foods. 2015;2(2):64–73.
Afroz A, Ali L, Karim M, Alramadan MJ, Alam K, Magliano DJ, Billah B. Glycaemic control for people with type 2 diabetes mellitus in Bangladesh-an urgent need for optimization of management plan. Sci Rep. 2019;9(1):1–10.
David UE, Asiwe JN, Fasanmade AA. Maternal hypothyroidism prolongs gestation period and impairs glucose tolerance in offspring of Wistar rats. Horm Mol Biol Clin Invest. 2022;43(3):323–8.
Al-Badri A, Hashmath Z, Oldland GH, Miller R, Javaid K, Syed AA, Chirinos JA. Poor glycemic control is associated with increased extracellular volume fraction in diabetes. Diabetes Care. 2018;41(9):2019–25.
Christensen AS, Viggers L, Hasselström K, Gregersen S. Effect of fruit restriction on glycemic control in patients with type 2 diabetes–a randomized trial. Nutr J. 2013;12(1):1–6.
Sadiya A, Mnla R. Impact of food pattern on glycemic control among type 2 diabetic patients: a cross-sectional study in the United Arab Emirates. Diabetes, Metabolic Syndrome Obesity: Targets Therapy. 2019;12:1143–50.
Ahmadi S, Awliaei H, Haidarizadeh M, Rostamzadeh J. The effect of ethanolic extract of urtica dioica leaves on high levels of blood glucose and gene expression of glucose transporter 2 (Glut2) in liver of alloxan-induced diabetic mice. Gene Cell Tissue. 2015;2(3):e30355.
Ambika S, Saravanan R. Effect of bergenin on hepatic glucose metabolism and insulin signaling in C57BL/6 J mice with high fat-diet induced type 2 diabetes. J Appl Biomed. 2016;14(3):221–7.
Okonofua DE, Asiwe JN, Anachuna KK, Moke EG, Sanusi KO, Adagbada EO, Yusuf MO, Alawode DI, Fasanmade AA. Effect of diabetes mellitus and hypertension on osmotic fragility and hemorheological factors in male Wistar rats. Biol, Med Nat Prod Chem. 2021;10(2):73–9.
Pantoja PKD, Colmenares DAJ, Isaza MJH. New caffeic acid derivative from Tithonia diversifolia (Hemsl.) A. gray butanolic extract and its antioxidant activity. Food Chem Toxicol. 2017;109:1079–85.
Karim N, Rahman A, Chanudom L, Thongsom M, Tangpong J. Mangosteen vinegar rind from Garcinia mangostana prevents high-fat diet and streptozotocin-induced type II diabetes nephropathy and apoptosis. J Food Sci. 2019;84(5):1208–15.
Danilova IG, Bulavintceva TS, Gette IF, Medvedeva SY, Emelyanov VV, Abidov MT. Partial recovery from alloxan-induced diabetes by sodium phthalhydrazide in rats. Biomed Pharmacother. 2017;95:103–10.
Rashid K, Sinha K, Sil PC. An update on oxidative stress mediated organ pathophysiology. Food Chem Toxicol: Int J Published British Industrial Biol Res Assoc. 2013;62:584–600.
Renugadevi J, Prabu SM. Cadmium-induced hepatotoxicity in rats and the protective effect of naringenin. Exp Toxicol Pathol. 2010;62(2):171–81.
Nasiri A, Ziamajidi N, Abbasalipourkabir R, Goodarzi MT, Saidijam M, Behrouj H, et al. Beneficial effect of aqueous garlic extract on inflammation and oxidative stress status in the kidneys of type 1 diabetic rats. Indian J Clin Biochem. 2017;32:329–36.
Mobasher MA, Germoush MO, Galal El-Tantawi H, Samy El-Said K. Metformin improves biochemical and pathophysiological changes in hepatocellular carcinoma with pre-existed diabetes mellitus rats. Pathogens. 2021;10(1):59.
Acknowledgements
The technical knowledge of Divic Specialist Diagnostic Laboratory, Port Harcourt, Rivers State, Nigeria, is gratefully acknowledged by the authors.
Funding
The study was entirely self-funded by the authors.
Author information
Authors and Affiliations
Contributions
The study was conceptualized by Jerome Ndudi Asiwe; the experimental assay was managed by Emuesiri Goodies Moke, Bartholomew Chukwuebuka Nwogueze, Daubry Tarela Mellish Elias, and Godwin D Yovwin; and the statistical analysis as well as manuscript writing was done by Jerome Ndudi Asiwe and Nicholas Asiwe.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare no competing interests.
Human and animal rights
The PAMO University of Medical Science research ethics committee (PUMS-AREC/2021/067) approved the study, and the procedure closely complied with the ARRIVE recommendations as outlined in the 1996 revision of NIH Publication No. 85–23.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Asiwe, J.N., Moke, E.G., Asiwe, N. et al. Dryopteris dilatata leaf extract ameliorates streptozotocin-induced diabetic nephropathy in male Wistar rat. Nutrire 48, 1 (2023). https://doi.org/10.1186/s41110-022-00186-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41110-022-00186-4