Abstract
Purpose
Essential metals may be crucial in obesity and type 2 diabetes (T2DM); diabesity pathogenesis and consequences. This study aimed to determine the metal levels in obese and non-obese patients with and without T2DM and their relationships with fetuin-A(Fet-A) levels, insulin sensitivity, and insulin resistance.
Methods
A total of 314 participants were enrolled, with 160 newly diagnosed T2DM patients and 154 non-T2DM subjects categorized into diabetic obese (n = 57), diabetic non-obese (n = 103), non-diabetic obese (n = 48), and non-diabetic non-obese (n = 106) subgroups. Fet-A, insulin sensitivity (QUCKI)/resistance (HOMA-IR), fasting glucose, and body mass index (BMI) were assessed. The essential metals were measured using inductively coupled plasma mass spectroscopy (ICP-MS).
Results
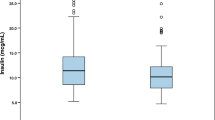
Fet-A levels were 3-fold higher (1391.4 ± 839.8 ng/ml) in T2DM patients than in non-T2DM (2165.6 ± 651.9 vs. 424.3 ± 219.1 ng/ml, p < 0.0001). Fet-A levels were 2.3-fold higher in the diabetic obese group than in the diabetic non-obese group (p < 0.0001). Fet-A levels were 2.0-fold higher in the diabetic non-obese group than in the non-diabetic obese group (p < 0.0001). Fet-A levels were positively correlated with insulin resistance (HOMA-IR) (r = 0.34, p < 0.0001) and negatively correlated with insulin sensitivity (QUIKI) (r = −0.41, p < 0.0001).
Cu, Se, Zn, and Fe levels were significantly lower in diabetic patients than in non-diabetic patients (p < 0.05). Se and Zn were significantly correlated with Fet-A (r = −0.41, p = 0.049 and r = −0.42, p = 0.001, respectively). Se and Zn were also correlated with insulin resistance (HOMA-IR) (r = −0.45, p = 0.049 and r = −0.36, p = 0.012, respectively) and insulin sensitivity (QUIKI) (r = 0.49, p = 0.042 and r = 0.30, p = 0.003, respectively). Similarly, Fe was negatively correlated with insulin levels (r = −0.33, p = 0.04) and insulin sensitivity (r = −0.34, p = 0.30). However, Mn was significantly correlated with Fet-A (r = 0.37, p = 0.001) and insulin resistance/sensitivity (r = 0.24, p = 0.026 and r = −0.24, p = 0.041) respectively in the diabetic obese group. Mg was an independent predictor of diabesity.
Conclusions
Mg play a significant role in obesity-related T2DM pathogenesis and complications via Fet-A, insulin sensitivity, and resistance modifications.

Similar content being viewed by others
References
Ozougwu JC, Obimba KC, Belonwu CD, Unakalamba CB. The pathogenesis and pathophysiology of type 1 and type 2 diabetes mellitus. J Physiol Pathophysiol. 2013;4:46–57. https://doi.org/10.5897/JPAP2013.0001.
Syed Ikmal SIQ, Zaman Huri H, Vethakkan SR, Wan Ahmad WA. Potential biomarkers of insulin resistance and atherosclerosis in type 2 diabetes mellitus patients with coronary artery disease. Int J Endocrinol. 2013:11. https://doi.org/10.1155/2013/698567.
Rochette L, Zeller M, Cottin Y, Vergely C. Diabetes, oxidative stress and therapeutic strategies. Biochim Biophys Acta. 2014;1840:2709–29. https://doi.org/10.1016/j.bbagen.2014.05.017.
VJ, Leon MA, Goustin AS. Alpha2-HSG, a specific inhibitor of insulin receptor autophosphorylation, interacts with the insulin receptor. Mol Cell Endocrinol. 2000;164:87–98.
Stefan N, Fritsche A, Weikert C, Boeing H, Joost H-G, Häring H-U, et al. Plasma fetuin-A levels and the risk of type 2 diabetes. Diabetes. 2008;57:2762–7. https://doi.org/10.2337/db08-0538.
Shidfar F, Zarrati M, Khamseh ME, Haghighat N, Rostami A, Zolfaghari H. Relationship between serum levels of fetuin-A with apo-A1, apo-B100, body composition and insulin resistance in patients with type 2 diabetes. Med J Islam Repub Iran. 2014;28:100.
Khalil H, Al-kuobaili F. Elevated fetuin - A level associated with an Atherogenic lipid profile in type 2 diabetes. Int J Pharm Sci Rev Res. 2013;21:266–9.
El-Deeb TS, Bakkar SM, Eltoony L, Zakhary MM, Kamel AA, Nafee AM, Hetta HF. The adipokine chemerin and fetuin-A serum levels in type 2 diabetes mellitus: relation to obesity and inflammatory markers. Egypt J Immunol. 2018;25(1):191–202.
Mooradian AD, Morley JE. Micronutrient status in diabetes mellitus. Am J Clin Nutr. 1987;45(5):877–95.
Evans JL, Goldfine ID, Maddux BA, Grodsky GM. Are oxidative stress-activated signaling pathways mediators of insulin resistance and -cell dysfunction? Diabetes. 2003;52(1):1–8.
Pérez-Matute P, Zulet MA, Martínez JA. Reactive species and diabetes: counteracting oxidative stress to improve health. Curr Opin Pharmacol. 2009;9(6):771–9.
Taylor C. Zinc, the pancreas, and diabetes: insights from rodent studies and future directions. Biometals. 2005;18(4):305–12.
Wijesekara N, Chimienti F, Wheeler MB. Zinc, a regulator of islet function and glucose homeostasis. Diabetes Obes Metab. 2009;11:202–14.
Simcox JA, McClain DA. Iron and diabetes risk. Cell Metab. 2013;17(3):329–41.
Hansen AF, Simić A, Åsvold BO, Romundstad PR, Midthjell K, Syversen T et al. Trace elements in early phase type 2 diabetes mellitus—a population-based study. The HUNT study in Norway. J Trace Elem Med Biol. 2017;40:46–53.
Chen H, Tan C. Prediction of type-2 diabetes based on several element levels in blood and chemometrics. Biol Trace Elem Res. 2012;147(1):67–74.
Dubey P, Thakur V, Chattopadhyay M. Role of minerals and trace elements in diabetes and insulin resistance. Nutrients. 2020;12(6):1864.
International diabetes federation. The IDF diabetes Atlas, 6th ed., 2013. Available online: http://www.idf.org/atlasmap/atlasmap. Accessed 5 May 2014.
Feller S, Boeing H, Pischon T. Body mass index, waist circumference, and the risk of type 2 diabetes mellitus: implications for routine clinical practice. Dtsch Arztebl Int. 2010;107(26):470.
Salgado AL, Carvalho L, Oliveira AC, Santos VN, Vieira JG, Parise ER. Insulin resistance index (HOMA-IR) in the differentiation of patients with non-alcoholic fatty liver disease and healthy individuals. Arq Gastroenterol. 2010;47(2):165–9. https://doi.org/10.1590/S0004-28032010000200009.
Vanhala P, Vanhala M, Kumpusalo E, Keinanen-Kiukaanniemi S. The quantitative insulin sensitivity check index QUICKI predicts the onset of type 2 diabetes better than fasting plasma insulin in obese subjects: a 5-year follow-up study. J Clin Endocrinol & Metab. 2002;87(12):5834–7. https://doi.org/10.1210/jc.2002-020591.
Misra A, Singhal N, Sivakumar B, Bhagat N, Jaiswal A, Khurana L. Nutrition transition in India: secular trends in dietary intake and their relationship to diet related non-communicable diseases. J Diabetes. 2011;3:278–92. https://doi.org/10.1111/j.1753-0407.2011.00139.x.
Misra A, Shrivastava U. Obesity and dyslipidemia in south Asians. Nutrients. 2013;5:2708–33. https://doi.org/10.3390/nu5072708.
Bourebaba L, Marycz K. Pathophysiological implication of fetuin-A glycoprotein in the development of metabolic disorders: a concise review. J Clin Med. 2019;8(12):2033.
Boren J, Taskinen MR, Olofsson SO, Levin M. Ectopic lipid storage and insulin resistance: a harmful relationship. J Intern Med. 2013;274(1):25–40.
Cai MM, Smith ER, Holt SG. The role of fetuin-A in mineral trafficking and deposition. BoneKEy reports. 2015:4.
Kovářová M, Kalbacher H, Peter A, Häring HU, Didangelos T, Stefan N, Birkenfeld A, Schleicher E, Kantartzis K. Detection and characterization of phosphorylation, glycosylation, and fatty acid bound to fetuin A in human blood. J Clin Med. 2021;10(3):411.
Sun Q, Cornelis MC, Manson JE, Hu FB. Plasma levels of fetuin-A and hepatic enzymes and risk of type 2 diabetes in women in the US. Diabetes. 2013;62:49–55. https://doi.org/10.2337/db12-0372.
Ismail NA, Ragab S, El Dayem SMA, ElBaky AA, Salah N, Hamed M. Fetuin-A levels in obesity: differences in relation to metabolic syndrome and correlation with clinical and laboratory variables. Arch Med Sci. 2012;8:826–33. https://doi.org/10.5114/aoms.2012.31616.
Chaudhary DP. Magnesium deficiency in type 2 diabetes. In Magnesium in Human Health and Disease 2013 pp. 119–126. Humana Press, Totowa. https://doi.org/10.1007/978-1-62703-044-1_7.
Hua H, Gonzales J, Rude RK. Magnesium transport induced ex vivo by a pharmacological dose of insulin is impaired in non-insulin-dependent diabetes mellitus. Magnes-Res. 1995;8(4):359–66.
Ekmekcioglu C, Prohaska C, Pomazal K, Steffan I, Schernthaner G, Marktl W. Concentrations of seven trace elements in different hematological matrices in patients with type 2 diabetes as compared to healthy controls. Biol Trace Elem Res. 2001;79(3):205–19.
Fernández-Real JM, Lopez-Bermejo A, Ricart W. Iron stores, blood donation, and insulin sensitivity and secretion. Clin Chem. 2005;51(7):1201–5. https://doi.org/10.1373/clinchem.2004.046847.
Jiang R, Manson JE, Meigs JB, Ma J, Rifai N, Hu FB. Body iron stores in relation to risk of type 2 diabetes in apparently healthy women. Jama. 2004;291(6):711–7. https://doi.org/10.1001/jama.291.6.711.
Al-Quraishy S, Dkhil MA, Moneim AE. Anti-hyperglycemic activity of selenium nanoparticles in streptozotocin-induced diabetic rats. Int J Nanomedicine. 2015;10:6741. https://doi.org/10.2147/IJN.S91377.
Meyer JA, Spence DM. A perspective on the role of metals in diabetes: past findings and possible future directions. Metallomics. 2009;1(1):32–41. https://doi.org/10.1039/b817203j.
Dewitte K, Dhondt A, Giri M, StOckl D, Rottiers R, Lameire N, et al. Differences in serum ionized and total magnesium values during chronic renal failure between non-diabetic and diabetic patients: a cross-sectional study. Diabetes Care. 2004;27(10):2503–5. https://doi.org/10.2337/diacare.27.10.2503.
De Valk HW, Verkaaik R, Van Rijn HJ, Geerdink RA, Struyvenberg A. Oral magnesium supplementation in insulin-requiring type 2 diabetic patients. Diabet Med. 1998;15(6):503–7. https://doi.org/10.1002/(SICI)1096-9136(199806)15:6<503::AID-DIA596>3.0.CO;2-M.
Rodríguez-Morán M, Guerrero-Romero F. Oral magnesium supplementation improves insulin sensitivity and metabolic control in type 2 diabetic subjects: a randomized double-blind controlled trial. Diabetes Care. 2003;26(4):1147–52. https://doi.org/10.2337/diacare.26.4.1147.
Sprietsma JE, Schuitemaker GE. Diabetes can be prevented by reducing insulin production. Med Hypotheses. 1994;42(1):15–23. https://doi.org/10.1016/0306-9877(94)90029-9.
Dzięgielewska-Gęsiak S, Wysocka E, Michalak S, Nowakowska-Zajdel E, Kokot T, Muc-Wierzgoń M. Role of lipid peroxidation products, plasma total antioxidant status, and cu-, Zn-superoxide dismutase activity as biomarkers of oxidative stress in elderly prediabetics. Oxidative Med Cell Longev 2014; 2014. https://doi.org/10.1155/2014/987303.
Beshgetoor D, Hambidge M. Clinical conditions altering copper metabolism in humans. Am J Clin Nutr. 1998;67(5):1017S–21S. https://doi.org/10.1093/ajcn/67.5.1017S.
Funding
The authors acknowledge an Intramural Research Grant (IEC-24/18) to Dr. Vandana Tiwari from Dr. Ram Manohar Lohia Institute of Medical Sciences, Lucknow, for their financial support.
Author information
Authors and Affiliations
Contributions
Anumesh Pathak- Manuscript drafting and writing, data analysis, tables and graphs preparation.
Vandana Tiwari: conceptualization, funding acquisition, execution of the experiment, validation of data, and draft editing.
Manish Raj Kulshrestha- Manuscript preparation and editing.
Shivani Singh- Sample collection & data collection, help in tables preparation.
Shefali Singh- Sample collection & enrolled patients.
Vikram Singh- Clinical history & patient details.
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pathak, A.K., Tiwari, V., Kulshrestha, M.R. et al. Impact of essential metals on insulin sensitivity and fetuin in obesity-related type 2 diabetes pathogenesis. J Diabetes Metab Disord 22, 703–712 (2023). https://doi.org/10.1007/s40200-023-01193-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-023-01193-6