Abstract
Purpose of Review
Metabolic comorbidities such as obesity, diabetes, hypertension, and dyslipidemia are common to multiple sclerosis (MS) and are associated with negative outcomes of the disease. Dietary intervention has the potential to improve MS co-morbidities; thus, it is a high priority for people living with MS to self-manage their disease. The present review aimed to summarize the recent evidence on the impacts of combining dietary modification with nutrition education and counseling on managing metabolic comorbidity markers in MS.
Recent Findings
Evidence suggests important roles for tailored dietary change strategies and nutrition education and counseling in managing metabolic comorbidities for MS. There is also indirect evidence suggesting a relationship between dietary fiber, the gut microbiome, and improved metabolic markers in MS, highlighting the need for more research in this area. For people living with MS, addressing both barriers and facilitators to dietary changes through behavior change techniques can help them achieve sustainable and tailored dietary behavior changes. This will support person-centered care, ultimately improving metabolic comorbidity outcomes.
Summary
Metabolic comorbidities in MS are considered modifiable diseases that can be prevented and managed by changes in dietary behavior. However, the impact of targeted dietary interventions on mitigating MS-related metabolic comorbidities remains inadequately explored. Therefore, this review has provided insights into recommendations to inform future best practices in MS. Further well-designed studies based on tailored dietary strategies applying behavior change theories are needed to address the underlying determinants of dietary practice in this population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Multiple sclerosis (MS) is an increasingly prevalent inflammatory demyelinating disease of the central nervous system (CNS) [1]. MS results in a heterogeneous array of unpredictable debilitating symptoms, including sensory disturbances (e.g., tingling, numbness, and itching), physical challenges (e.g., fatigue, spasticity, muscle weakness, and loss of balance), cognitive impairment (e.g., memory loss and poor concentration), emotional symptoms (e.g., depression and anxiety), and vision problems (e.g., blurred vision and pain in eye movement) [1, 2]. However, the manifestation and severity of the symptoms vary among individuals depending on the location of demyelination in the CNS [3].
Besides the presence of symptoms, comorbidities are more common in people with MS than in the general population [4•, 5]. Comorbidities in MS refer to the presence of additional medical conditions that coexist alongside MS and require treatment [6]. A growing body of literature highlights metabolic comorbidities such as obesity, diabetes, hypertension, and dyslipidemia are common in MS and are risk factors for cardiovascular disease (CVD) [4•, 7,8,9]. CVD, hypertension, dyslipidemia, and diabetes are grouped as “vascular comorbidities” [10]. Increasing evidence has demonstrated that vascular comorbidities in MS are linked to greater diagnostic delays [11], higher severity of common symptoms [12], an increasing rate of healthcare utilization [13], economic burden [14], and higher mortality rates [15].
A recent retrospective cohort study showed that hypertension, CVD, and type 2 diabetes (T2DM) are associated with an accelerated progression of disability in MS, as measured by the Expanded Disability Status Scale (EDSS) [8]. This is because metabolic comorbidities, particularly T2DM, are connected to reduced brain and grey matter volume [15]. Obesity, which is highly prevalent in people with MS, is related to an increased risk of additional comorbidities [16]; thus, it has been associated with worsening MS symptoms and disease progression [7, 17]. Another study has found that people with MS who have dyslipidemia and three or more comorbidities have a higher relapse incidence over 2 years compared to those without comorbidities [18]. Consequently, metabolic comorbidities result in a lower health-related quality of life (HRQoL) in people with MS [4•, 19].
Sustainable and tailored strategies are needed to delay the onset and manage metabolic comorbidities and their effects on MS outcomes. Studies have shown that metabolic comorbidities are often considered modifiable diseases, which can be preventable or modified by adopting healthy lifestyle behaviors [20,21,22]. Healthy lifestyle practices include improved eating habits, regular physical activity, maintaining a healthy weight, limited alcohol consumption, and smoking cessation. Incorporating healthy lifestyle behaviors to optimize MS outcomes such as metabolic comorbidities, is of increasing interest [23]. Dietary modification, as one element of healthy lifestyle behaviors, is a high priority for people with MS to self-manage their disease [24,25,26].
While there is emerging evidence of the role of dietary modification as a non-pharmacological treatment in MS, MS-specific nutrition guidance is lacking [27,28,29]. Dietary recommendations for people with MS are based on general population guidelines that recommend following a balanced diet to optimize overall health and decrease the risk of diet-related comorbidities [27, 28]. In the general population, changes in dietary habits are considered a first-line strategy for modulating and delaying the onset of metabolic comorbidities. For example, changes toward a healthy diet among people at risk of metabolic comorbidities have been linked with reduced risk of T2DM, CVD, and microvascular complications [30], as well as improved glucose homeostasis and blood lipid profiles [31]. Likewise, evidence from observational studies highlighted that adherence to a higher-quality dietary pattern with mild-to-moderate alcohol intake is associated with a lowered risk of metabolic comorbidities and improved HRQoL in people with MS [21, 32•] because of the potential of protecting the neurologic reserve [33]. By contrast, adherence to a less healthy diet is linked with a higher risk of obesity, an altered blood lipid profile, the presence of at least two metabolic comorbidities, and a worsened EDSS in MS [34].
In addition to early prevention, it is well-Established that already-present metabolic comorbidities can be managed by dietary interventions in the non-MS population [35]. Incorporating nutrition education and counseling by utilizing behavior change techniques can also improve markers of metabolic comorbidities [36]. Behavior change techniques include active components employed in an intervention to support and facilitate desirable changes in dietary habits [37]. However, the role of dietetic interventions in MS-related metabolic comorbidities are yet to be reviewed. Therefore, this review was conducted to summarize the available literature on dietary modification and nutrition education and counseling and their impact on metabolic comorbidity markers in MS. Metabolic markers include the body mass index (BMI), body weight (BW), waist circumference (WC), blood pressure, blood lipid profile, fasting blood glucose, hemoglobin A1c (HbA1c), and insulin.
Literature on Dietary Modification, Nutrition Education and Counseling, and Metabolic Comorbidities in MS
Dietary Intervention-Related Metabolic Comorbidity Markers in MS
Rimmer et al. [38] conducted a 9-month trial that involved a tailored, telehealth weight management program. The program included a personalized healthy diet delivered via videos, telephone consultations, and educational resources. The intervention was effective in reducing BW and BMI (p = 0.04) among 27 adults with physical disabilities (including MS). Another trial of nutrition counseling for people with MS (n = 57) incorporated education on healthy eating habits and regular messages on a WhatsApp group [39••]. The participants also received a collection of educational booklets, dietary records, and measuring tools for cooking to help them cook and choose appropriate foods. After 3 months of monthly counseling sessions, the intervention group showed significant improvements in the anthropometric measurements, including BW, WC, and BMI, as well as dietary intake assessed by a 24-h recall (all p < 0.05). Thus, the intervention led to significant weight loss among participants who were considered to be overweight or obese (p < 0.05).
These findings are consistent with another 3-month feasibility study that focused on behavioral dietary changes based on a low glycemic load (GL) diet [40••]. The diet focused on healthy dietary patterns (i.e., whole foods with low GI and minimally processed foods). The study applied a digital health approach, including education delivered via weekly modules, weekly tele-coaching calls, and the use of a mobile app to record dietary intake daily. After the intervention, participants with MS (n = 18) exhibited an improvement in cardiometabolic risk, including HbA1c, fasting blood glucose, blood pressure, and body composition. Another study by Papandreou et al. [41••] evaluated a 3-month dietitian education and counseling program that included individualized dietary interventions based on the Mediterranean diet (MedDiet) for 20 women living with MS. A significant (p < 0.001) decrease in BW, BMI, fat mass, and serum glucose was observed compared to the baseline, which was negatively correlated with cholesterol intake levels (p < 0.05). The MedDiet pattern is based on a high intake of fruits, vegetables, whole grains, olive oil, seeds, and legumes, a moderate intake of dairy products, and a low intake of animal fats [42].
Yadav et al. [43] conducted a 1-year-RCT that allocated participants into either a diet with dietitian counseling (n = 26) or a control group (n = 27). The findings revealed that a diet based on complex starchy carbohydrates without animal products or vegetable oils significantly (p < 0.001) reduced BMI. In addition, several serum metabolic biomarkers significantly decreased in the intervention group compared to the control; these biomarkers included total cholesterol (p = 0.027), low-density lipoprotein (LDL) cholesterol (p = 0.031), and fasting insulin levels (p = 0.0068). However, an inconsistent result was found in another intervention [44], where 40 mins of nutrition education once per month for 3 months, teaching 34 people with MS about a healthy diet and practical strategies to purchase, prepare, and cook food did not result in changes to BMI or WC, although the quality of the diet did improve, as assessed by the Healthy Eating Index (HEI)-2010.
To date, existing interventions have mainly focused on tailored healthy lifestyle changes with an emphasis on higher diet quality to improve markers of metabolic comorbidities in MS. Nutrition education and counseling are part of a tailored approach that is associated with increased adherence to dietary guidelines [45]. However, data on dietary adherence are limited in the reviewed interventions. Thus, exploring long-term outcomes such as the adherence to and sustainability of changes to dietary behavior are essential to inform future best practices in MS care.
Gut Microbiome, Metabolic Comorbidities, and Implications for MS
The reviewed intervention studies emphasized limiting the intake of highly processed foods and increasing the consumption of fruits, vegetables, legumes, grains, and other sources of dietary fiber. Three studies reported favorable outcomes on metabolic markers and found that fiber intake significantly increased after the intervention [38, 39••, 41••]. This is consistent with the dietary guidelines that recommend a well-balanced, diverse, and fiber-rich diet. Nutrition is a modifiable element that is able to shape the composition and characteristics of the gut microbiome [46]. A fiber-rich diet leads to gut eubiosis (a balance in the gut microbiota) through increased gut bacteria diversity and anti-inflammatory mediators associated with microbes [46, 47]. This results in reducing the risks of obesity, hyperlipidemia, hyperglycemia, T2DM, and CVD in the general population [48]. On the other hand, a low-fiber, Western diet increases the levels of pro-inflammatory mediators and promotes gut dysbiosis (an imbalance in the gut microbiota) [46, 49], which disturbs the metabolic markers [50]. Emerging evidence indicates that alterations in the composition and function of the gut microbiome may lead to the development and progression of metabolic comorbidities [50].
Compared to healthy individuals, people with MS experience gut dysbiosis more often, which may promote systemic inflammation [51]. Thus, emerging evidence leans toward the role of the gut microbiome in modulating CNS inflammation and its implications for MS pathogenesis [51, 52]. This may provide indirect evidence of the influence of dysbiosis on the pathogenesis of metabolic comorbidities in MS, though research focused on the intricate interplay between the diet, gut microbiome, and metabolic comorbidities in MS is limited. As a result, future MS dietary trials may benefit from collecting microbiota samples to elucidate the role of dietary modification on the gut microbiome and metabolic comorbidity outcomes in MS. This may lead to novel therapeutic avenues for slowing MS progression and optimizing clinical outcomes by addressing metabolic comorbidities.
Nutrition Education and Counseling: Individualized Approach for Best Practices
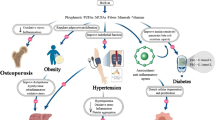
The following section discusses improving metabolic comorbidities in MS by achieving person-centered care through addressing both barriers and facilitators to dietary changes, depicted in Fig. 1.
Modifiable comorbidities in MS can be recognized in the context of chronic disease care models [10], which involve the responsibility of healthcare professionals (HCPs) to provide education in routine care. This aims to promote self-management, ultimately preventing or managing comorbidities. Nutrition education and counseling were incorporated into the reviewed interventions to empower and motivate participants to attain dietary self-management through behavioral changes. An individualized approach is required because of the complexity of MS [53]. Changes in the dietary behavior of people with MS are also complex and need ongoing support [54]. Accordingly, using appropriate behavior change theories to guide interventions is recommended to guide best practice for ensuring sustainable changes in dietary behavior [55]. Nonetheless, only one intervention reported the use of a behavior change framework, which was the Health Action Process Approach (HAPA) behavior change theory to guide implementation of the activities [40••].
The inclusion of behavior change techniques that promote personalization goals, self-regulatory skills, and social engagement while anticipating personal barriers may improve adherence to dietary changes [56]. A large body of qualitative evidence has explored barriers to achieving self-management behavioral changes, including a healthy diet in people with MS. Barriers identified were related to a lack of motivation, person-centered support, and communication from HCPs [57, 58]. Additional barriers to adopting healthy dietary choices include a lack of cooking skills and dietary knowledge [59] as well as online dietary misinformation [26]. Behavior change techniques were integrated into the reviewed interventions to overcome these barriers and promote positive changes in dietary behavior. One intervention was informed by motivational interviewing techniques to support patient-centered care [38]. Other techniques utilized were goal setting and creating action plans [38, 40••], problem-solving strategies [40••], social environment support [39••, 44], addressing food preparation and cooking [38, 39••, 44], and providing practical dietary resources [38, 39••, 40••, 44]. Self-monitoring to encourage individuals to regularly track their dietary changes were also employed in the interventions [38, 39••, 40••].
Integrating evidence-based nutrition education into the routine care of people with MS, who may face unique physical and cognitive l challenges due to the disease, is crucial. The interventions reviewed were delivered and facilitated by dietitians [40••, 41••, 43] and coaches with health education and kinesiology degrees [40••]. Ideally, nutrition educators are dietitians; however, nutrition counseling can be limited by low referral rates, the cost of dietetic services, or remote locations [60]. Hence, changes in behaviors, including nutrition, must become basic competencies for all HCPs who provide support for people living with/or at risk of nutrition-related chronic diseases [61]. Neurologists and a multidisciplinary team in MS care are needed to empower people with MS with dietary behavior self-management near the time of diagnosis and across all stages of the disease, including appropriate referrals to dietitians care. For interventions to be equitable and sustainable, dietary guidance should be personalized and tailored to the individual’s needs, barriers should be identified, and strategies to facilitate changes should be provided to this population. This will ensure tailored changes in dietary behavior and will result in person-centered care, delaying onset or early management of metabolic comorbidities, and in turn, improving clinical outcomes and quality of life for people with MS.
Conclusion and Recommendations for Research
Metabolic comorbidities such as obesity, diabetes, hypertension, and dyslipidemia are considered modifiable lifestyle diseases that can be prevented early and managed in MS by dietary modification as an adjunct to first-line therapies. Prevention and intervention of metabolic comorbidities through dietary management in people with MS should target behavioral changes for sustainable outcomes. Furthermore, dietetic management should be incorporated as part of a team-based approach in routine MS care to improve the outcome of the disease.
While current evidence suggests that dietary interventions play a role in improving metabolic comorbidity markers in MS, the impact of targeted dietary interventions on mitigating metabolic comorbidities remains inadequately explored. The current evidence is constrained by the paucity of intervention studies, small sample sizes, and the short duration of the interventions. To address this gap, further well-designed trials that include tailored dietary behavior modification strategies are needed. Interventions based on behavior change theories are also required to address the underlying determinants of dietary practices in people with MS. Together, these can improve our understanding of the role of tailored dietary behavior modification in managing and decreasing the prevalence of metabolic comorbidities in MS.
Data Availability
Not applicable.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
McGinley MP, Goldschmidt CH, Rae-Grant AD. Diagnosis and treatment of multiple sclerosis: a review. JAMA. 2021;325(8):765–79. https://doi.org/10.1001/jama.2020.26858.
Lakin L, Davis BE, Binns CC, Currie KM, Rensel MR. Comprehensive approach to management of multiple sclerosis: addressing invisible symptoms—a narrative review. Neurology and therapy. 2021;10:75–98. https://doi.org/10.1007/s40120-021-00239-2.
Prineas JW, Barnard RO, Revesz T, Kwon EE, Sharer L, Cho E-S. Multiple sclerosis: pathology of recurrent lesions. Brain. 1993;116(3):681–93. https://doi.org/10.1093/brain/116.3.681.
• Hauer L, Perneczky J, Sellner J. A global view of comorbidity in multiple sclerosis: a systematic review with a focus on regional differences, methodology, and clinical implications. J Neurol. 2021;268(11):4066–77. https://doi.org/10.1007/s00415-020-10107-y. A global systematic review shows the worldwide prevalence and patterns of comorbid conditions, including metabolic in multiple sclerosis.
Simpson RJ, McLean G, Guthrie B, Mair F, Mercer SW. Physical and mental health comorbidity is common in people with multiple sclerosis: nationally representative cross-sectional population database analysis. BMC Neurol. 2014;14(1):1–8. https://doi.org/10.1186/1471-2377-14-128.
Hall SF. A user’s guide to selecting a comorbidity index for clinical research. J Clin Epidemiol. 2006;59(8):849–55. https://doi.org/10.1016/j.jclinepi.2005.11.013.
Ciampi E, Uribe-San-Martin R, Soler B, Molnar K, Reyes D, Keller K, et al. Prevalence of comorbidities in multiple sclerosis and impact on physical disability according to disease phenotypes. Mult Scler Related Disorders. 2020;46.
Maric G, Pekmezovic T, Tamas O, Veselinovic N, Jovanovic A, Lalic K, et al. Impact of comorbidities on the disability progression in multiple sclerosis. Acta Neurol Scand. 2022;145(1):24–9. https://doi.org/10.1111/ane.13516.
Waliszewska-Prosół M, Nowakowska-Kotas M, Misiak B, Chojdak-Łukasiewicz J, Budrewicz S, Pokryszko-Dragan A. Allostatic load index in patients with multiple sclerosis: a case-control study. Psychoneuroendocrinology. 2022;142: 105788. https://doi.org/10.1016/j.psyneuen.2022.105788.
Overs S, Hughes CM, Haselkorn JK, Turner AP. Modifiable comorbidities and disability in multiple sclerosis. Curr Neurol Neurosci Rep. 2012;12:610–7. https://doi.org/10.1007/s11910-012-0293-4.
Marrie RA, Horwitz R, Cutter G, Tyry T, Campagnolo D, Vollmer T. Comorbidity delays diagnosis and increases disability at diagnosis in MS. Neurology. 2009;72(2):117–24. https://doi.org/10.1212/01.wnl.0000333252.78173.5f.
Lo LMP, Taylor BV, Winzenberg T, Palmer AJ, Blizzard L, van der Mei I. Comorbidities contribute substantially to the severity of common multiple sclerosis symptoms. J Neurol. 2021;268:559–68. https://doi.org/10.1007/s00415-020-10192-z.
Magyari M, Sorensen PS. Comorbidity in multiple sclerosis. Front Neurol. 2020;11:851. https://doi.org/10.3389/fneur.2020.00851.
Ponzio M, Monti MC, Mallucci G, Borrelli P, Fusco S, Tacchino A, et al. The economic impact of comorbidity in multiple sclerosis. Neurol Sci. 2023;44(3):999–1008. https://doi.org/10.1007/s10072-022-06517-7.
Fitzgerald KC, Damian A, Conway D, Mowry EM. Vascular comorbidity is associated with lower brain volumes and lower neuroperformance in a large multiple sclerosis cohort. Mult Scler J. 2021;27(12):1914–23. https://doi.org/10.1177/1352458520984746.
Karczewski J, Śledzińska E, Baturo A, Jończyk I, Maleszko A, Samborski P, et al. Obesity and inflammation European cytokine network. 2018;29:83–94. https://doi.org/10.1684/ecn.2018.0415.
Marrie RA. Comorbidity in multiple sclerosis: implications for patient care. Nat Rev Neurol. 2017;13(6):375–82. https://doi.org/10.1038/nrneurol.2017.33.
Kowalec K, McKay KA, Patten SB, Fisk JD, Evans C, Tremlett H, et al. Comorbidity increases the risk of relapse in multiple sclerosis: a prospective study. Neurology. 2017;89(24):2455–61. https://doi.org/10.1212/WNL.0000000000004716.
Marrie RA, Horwitz R, Cutter G, Tyry T. Cumulative impact of comorbidity on quality of life in MS. Acta Neurol Scand. 2012;125(3):180–6. https://doi.org/10.1111/j.1600-0404.2011.01526.x.
Li Y, Pan A, Wang DD, Liu X, Dhana K, Franco OH, et al. Impact of healthy lifestyle factors on life expectancies in the US population. Circulation. 2018;138(4):345–55. https://doi.org/10.1161/CIRCULATIONAHA.117.032047.
Marck CH, Neate SL, Taylor KL, Weiland TJ, Jelinek GA. Prevalence of comorbidities, overweight and obesity in an international sample of people with multiple sclerosis and associations with modifiable lifestyle factors. PLoS ONE. 2016;11(2): e0148573. https://doi.org/10.1371/journal.pone.0148573.
Ball L, Johnson C, Desbrow B, Leveritt M. General practitioners can offer effective nutrition care to patients with lifestyle-related chronic disease. J Prim Health Care. 2013;5(1):59–69. https://doi.org/10.1071/hc13059.
Moss BP, Rensel MR, Hersh CM. Wellness and the role of comorbidities in multiple sclerosis. Neurotherapeutics. 2017;14:999–1017. https://doi.org/10.1007/s13311-017-0563-6.
Probst Y, Guan V, Van Der Walt A, Rath LM, Bonney A, Kent J. Patient self-management and empowerment for multiple sclerosis: the implications of dietary lifestyle management for primary care. Aus J Gen Pract. 2022;51(4):209–12. https://doi.org/10.31128/AJGP-09-21-6179.
Marck CH, Probst Y, Chen J, Taylor B, van der Mei I. Dietary patterns and associations with health outcomes in Australian people with multiple sclerosis. Eur J Clin Nutr. 2021;75(10):1506–14. https://doi.org/10.1038/s41430-021-00864-y.
Russell RD, Black LJ, Begley A. Navigating dietary advice for multiple sclerosis. Health Expect. 2021;24(3):853–62. https://doi.org/10.1111/hex.13226.
Esposito S, Bonavita S, Sparaco M, Gallo A, Tedeschi G. The role of diet in multiple sclerosis: a review. Nutr Neurosci. 2018;21(6):377–90. https://doi.org/10.1080/1028415X.2017.1303016.
Evans E, Levasseur V, Cross AH, Piccio L. An overview of the current state of evidence for the role of specific diets in multiple sclerosis. Multiple Sclerosis and Related Disorders. 2019;36: 101393. https://doi.org/10.1016/j.msard.2019.101393.
Kirkland H, Campbell J, Reece J, Nag N, Probst Y, Neate S, et al. Higher diet quality is associated with short and long-term benefits on SF-6D health state utilities: a 5-year cohort study in an international sample of people with multiple sclerosis. Qual Life Res. 2023;32:1–14. https://doi.org/10.1007/s11136-023-03361-w.
Gong Q, Zhang P, Wang J, Ma J, An Y, Chen Y, et al. Morbidity and mortality after lifestyle intervention for people with impaired glucose tolerance: 30-year results of the Da Qing Diabetes Prevention Outcome Study. Lancet Diabetes Endocrinol. 2019;7(6):452–61. https://doi.org/10.1016/S2213-8587(19)30093-2.
Gao X, Tian Z, Zhao D, Li K, Zhao Y, Xu L, et al. Associations between adherence to four a priori dietary indexes and cardiometabolic risk factors among hyperlipidemic patients. Nutrients. 2021;13(7):2179. https://doi.org/10.3390/nu13072179.
• Suliga E, Brola W, Sobaś K, Cieśla E, Jasińska E, Gołuch K, et al. Dietary patterns and metabolic disorders in Polish adults with multiple sclerosis. Nutrients. 2022;14(9):1927. https://doi.org/10.3390/nu14091927. This study analyzed the dietary patterns of people with multiple sclerosis, and it found that an unhealthy dietary pattern was associated with the presence of some metabolic risk factors, such as abdominal obesity and low HDL-cholesterol levels.
Vollmer TL, Nair KV, Williams IM, Alvarez E. Multiple sclerosis phenotypes as a continuum: the role of neurologic reserve. Neurol Clin Pract. 2021;11(4):342–51. https://doi.org/10.1212/cpj.0000000000001045.
Dorothy V. Dietary patterns and metabolic disorders in multiple sclerosis. Neurosurg J. 2022;5(2):22–3.
Ojo O. Nutrition and chronic conditions. Nutrients. 2019;11(2):459. https://doi.org/10.3390/nu11020459.
Mitchell LJ, Ball LE, Ross LJ, Barnes KA, Williams LT. Effectiveness of dietetic consultations in primary health care: a systematic review of randomized controlled trials. J Acad Nutr Diet. 2017;117(12):1941–62. https://doi.org/10.1016/j.jand.2017.06.364.
Carey RN, Connell LE, Johnston M, Rothman AJ, De Bruin M, Kelly MP, et al. Behavior change techniques and their mechanisms of action: a synthesis of links described in published intervention literature. Ann Behav Med. 2019;53(8):693–707. https://doi.org/10.1093/abm/kay078.
Rimmer JH, Wang E, Pellegrini CA, Lullo C, Gerber BS. Telehealth weight management intervention for adults with physical disabilities: a randomized controlled trial. Am J Phys Med Rehabil. 2013;92(12):1084–94. https://doi.org/10.1097/PHM.0b013e31829e780e.
•• Afifi Z, Hassan A, Abdelrahman N, El Sayed A, Salem M. Impact of nutrition counseling on anthropometry and dietary intake of multiple sclerosis patients at Kasr Alainy Multiple Sclerosis Unit, Cairo, Egypt 2019–2020: randomized controlled clinical trial. Archives of Public Health. 2023;81(1):11. https://doi.org/10.1186/s13690-022-01013-y. This randomized controlled clinical trial examined the relationship between nutrition counselling on a healthy diet and body parameters in people with multiple sclerosis. It depicts how healthy dietary modification combined with nutrition counselling can significantly improve anthropometric measurements.
•• Wingo BC, Rinker JR, Goss AM, Green K, Wicks V, Cutter GR, et al. Feasibility of improving dietary quality using a telehealth lifestyle intervention for adults with multiple sclerosis. Mult Scler Relat Disord. 2020;46: 102504. https://doi.org/10.1016/j.msard.2020.102504. This interventional study highlights the effectiveness of healthy behavioral dietary education delivered through telehealth in improving diet quality and reducing cardiometabolic risk in people with multiple sclerosis.
•• Papandreou P, Gioxari A, Daskalou E, Vasilopoulou A, Skouroliakou M. Personalized nutritional intervention to improve Mediterranean diet adherence in female patients with multiple sclerosis: a randomized controlled study. Dietetics. 2022;1(1):25–38. https://doi.org/10.3390/dietetics1010004. This randomized controlled clinical trial showed the significant effect of dietitian counseling and individualized dietary interventions on improving body composition measurements and serum glucose in people with multiple sclerosis.
Martínez-González MA, Gea A, Ruiz-Canela M. The Mediterranean diet and cardiovascular health: a critical review. Circ Res. 2019;124(5):779–98. https://doi.org/10.1161/CIRCRESAHA.118.313348.
Yadav V, Marracci G, Kim E, Spain R, Cameron M, Overs S, et al. Low-fat, plant-based diet in multiple sclerosis: a randomized controlled trial. Mult Scler Relat Disord. 2016;9:80–90. https://doi.org/10.1016/j.msard.2016.07.001.
Uzdil Z, Türker PF, Terzi M. Effects of nutrition education given to persons with multiple sclerosis and their families on diet quality and anthropometric and biochemical measurements. Revista de Nutrição. 2022. https://doi.org/10.1590/1678-9865202235e220153.
Trestini I, Sperduti I, Caldart A, Bonaiuto C, Fiorio E, Parolin V, et al. Evidence-based tailored nutrition educational intervention improves adherence to dietary guidelines, anthropometric measures and serum metabolic biomarkers in early-stage breast cancer patients: a prospective interventional study. The Breast. 2021;60:6–14. https://doi.org/10.1016/j.breast.2021.08.008.
Xie L, Alam MJ, Marques FZ, Mackay CR. A major mechanism for immunomodulation: dietary fibres and acid metabolites. Semin Immunol. 2023;66: 101737. https://doi.org/10.1016/j.smim.2023.101737.
Cronin P, Joyce SA, O’Toole PW, O’Connor EM. Dietary fibre modulates the gut microbiota. Nutrients. 2021;13(5):1655. https://doi.org/10.3390/nu13051655.
Stephen AM, Champ MM-J, Cloran SJ, Fleith M, Van Lieshout L, Mejborn H, et al. Dietary fibre in Europe: current state of knowledge on definitions, sources, recommendations, intakes and relationships to health. Nutr Res Rev. 2017. https://doi.org/10.1017/S095442241700004X.
Riccio P, Rossano R. Diet, gut microbiota, and vitamins D+ A in multiple sclerosis. Neurotherapeutics. 2018;15(1):75–91. https://doi.org/10.1007/s13311-017-0581-4.
Wang PX, Deng XR, Zhang CH, Yuan HJ. Gut microbiota and metabolic syndrome. Chin Med J (Engl). 2020;133(7):808–16. https://doi.org/10.1097/cm9.0000000000000696.
Dopkins N, Nagarkatti PS, Nagarkatti M. The role of gut microbiome and associated metabolome in the regulation of neuroinflammation in multiple sclerosis and its implications in attenuating chronic inflammation in other inflammatory and autoimmune disorders. Immunology. 2018;154(2):178–85. https://doi.org/10.1111/imm.12903.
Yadav SK, Ito K, Dhib-Jalbut S. Interaction of the gut microbiome and immunity in multiple sclerosis: impact of diet and immune therapy. Int J Mol Sci. 2023. https://doi.org/10.3390/ijms241914756.
Manche S, Probst Y. Exploring the socio-ecological factors impacting lifestyle management of multiple sclerosis: a scoping review. Multiple Sclerosis and Related Disorders. 2023;79: 104958. https://doi.org/10.1016/j.msard.2023.104958.
Silveira SL, Richardson EV, Motl RW. Desired resources for changing diet among persons with multiple sclerosis: qualitative inquiry informing future dietary interventions. International Journal of MS Care. 2022;24(4):175–83. https://doi.org/10.7224/1537-2073.2021-052.
Baker S, Auld G, Ammerman A, Lohse B, Serrano E, Wardlaw MK. Identification of a framework for best practices in nutrition education for low-income audiences. J Nutr Educ Behav. 2020;52(5):546–52. https://doi.org/10.1016/j.jneb.2019.12.007.
Deslippe AL, Soanes A, Bouchaud CC, Beckenstein H, Slim M, Plourde H, et al. Barriers and facilitators to diet, physical activity and lifestyle behavior intervention adherence: a qualitative systematic review of the literature. Int J Behav Nutr Phys Act. 2023;20(1):1–25. https://doi.org/10.1186/s12966-023-01424-2.
Barnard E, Brown CR, Weiland TJ, Jelinek GA, Marck CH. Understanding barriers, enablers, and long-term adherence to a health behavior intervention in people with multiple sclerosis. Disabil Rehabil. 2020;42(6):822–32. https://doi.org/10.1080/09638288.2018.1510550.
Faraclas E, Merlo A, Lynn J, Lau JD. Perceived facilitators, needs, and barriers to health related quality of life in people with multiple sclerosis: a qualitative investigation. Journal of Patient-Reported Outcomes. 2022;6(1):89. https://doi.org/10.1186/s41687-022-00496-1.
Dean C, Parks S, Titcomb TJ, Arthofer A, Meirick P, Grogan N, et al. Facilitators of and barriers to adherence to dietary interventions perceived by women with multiple sclerosis and their support persons. International journal of MS care. 2022;24(5):235–41. https://doi.org/10.7224/1537-2073.2021-051.
Carter C, Harnett JE, Krass I, Gelissen IC. A review of primary healthcare practitioners’ views about nutrition: implications for medical education. Int J Med Educ. 2022;13:124–37. https://doi.org/10.5116/ijme.6271.3aa2.
Kahan S, Manson JE. Nutrition counseling in clinical practice: how clinicians can do better. JAMA. 2017;318(12):1101–2. https://doi.org/10.1001/jama.2017.10434.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions No funding was received for conducting this review. Shoroog Allogmanny holds a PhD scholarship awarded from Taibah University, Saudi Arabia; however, the funding body was not involved in writing the review or the decision to submit the review for publication.
Author information
Authors and Affiliations
Contributions
The authors’ responsibilities were as follows: S.A. was involved in the development of the research question, searching for articles of interest, and preparation of the design and original draft; YP critically revised the work and was involved in editing the original manuscript. Both authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
Yasmine Probst is a person living with MS and is supported by a fellowship funded by MS Australia. She has also received honoraria from other MS organizations. Shoroog Allogmanny has no competing interests to declare that are relevant to the content of this article.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Allogmanny, S., Probst, Y. Dietary Modification Combined with Nutrition Education and Counseling for Metabolic Comorbidities in Multiple Sclerosis: Implications for Clinical Practice and Research. Curr Nutr Rep 13, 106–112 (2024). https://doi.org/10.1007/s13668-024-00538-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13668-024-00538-8