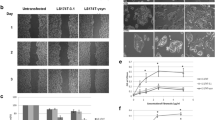
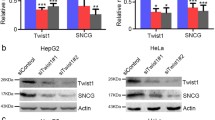
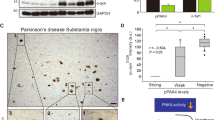
Abstract
Hyperactivation of AKT plays a critical role in the survival and proliferation of cancer cells. However, the molecular mechanisms underlying AKT activation remain elusive. Here, we tested the effect of γ-synuclein, a member of the synuclein family of proteins, on the activation of AKT. We show that the expression level of γ-synuclein is increased in non-small cell lung cancer (NSCLC) tissues. γ-Synuclein binds to the protein kinase domain of AKT and promotes its phosphorylation. Overexpression of γ-synuclein in H157 cells enhances cell proliferation and protects the cells from staurosporine-induced cytotoxicity. Knockdown of γ-synuclein attenuates AKT activation and cell proliferation induced by epidermal growth factor. The effect of γ-synuclein is abolished when AKT is depleted. Thus, γ-synuclein promotes cell survival and proliferation via activating AKT and may play a causal role in the pathogenesis of NSCLC.




Similar content being viewed by others
References
Esposito L, Conti D, Ailavajhala R, Khalil N, Giordano A. Lung cancer: are we up to the challenge? Curr genomics. 2010;11(7):513–8.
Travis WD, Travis LB, Devesa SS. Lung cancer. Cancer. 1995;75(1 Suppl):191–202.
Molina JR, Yang P, Cassivi SD, Schild SE, Adjei AA. Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clin Proc. 2008;83(5):584–94.
Soda M, Choi YL, Enomoto M, Takada S, Yamashita Y, Ishikawa S, et al. Identification of the transforming EML4–ALK fusion gene in non-small-cell lung cancer. Nature. 2007;448(7153):561–6.
Paez JG, Jänne PA, Lee JC, Tracy S, Greulich H, Gabriel S, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004;304(5676):1497–500.
Lemjabbar-Alaoui H, Hassan OU, Yang Y-W, Buchanan P. Lung cancer: biology and treatment options. Biochim Biophys Acta. 2015;1856(2):189–210.
Zhu G, Fan Z, Ding M, Zhang H, Mu L, Ding Y, et al. An EGFR/PI3K/AKT axis promotes accumulation of the Rac1-GEF Tiam1 that is critical in EGFR-driven tumorigenesis. Oncogene. 2015;34(49):5971–82.
Lastwika KJ, Wilson W, Li QK, Norris J, Xu H, Ghazarian SR, et al. Control of PD-L1 expression by oncogenic activation of the AKT–mTOR pathway in non–small cell lung cancer. Cancer Res. 2016;76(2):227–38.
Settleman J, Kurie JM. Drugging the bad “AKT-TOR” to overcome TKI-resistant lung cancer. Cancer Cell. 2007;12(1):6–8.
Zhou H, Huang S. Role of mTOR signaling in tumor cell motility, invasion and metastasis. Curr Protein Peptide Sci. 2011;12(1):30.
Franke T. PI3K/Akt: getting it right matters. Oncogene. 2008;27(50):6473–88.
Guo G, Gong K, Wohlfeld B, Hatanpaa KJ, Zhao D, Habib AA. Ligand-independent EGFR signaling. Cancer Res. 2015;75(17):3436–41.
Hill MM, Hemmings BA. Hemmings, inhibition of protein kinase B/Akt: implications for cancer therapy. Pharmacol Ther. 2002;93(2):243–51.
Balsara BR, Pei J, Mitsuuchi Y, Page R, Klein-Szanto A, Wang H, et al. Frequent activation of AKT in non-small cell lung carcinomas and preneoplastic bronchial lesions. Carcinogenesis. 2004;25(11):2053–9.
Okudela K, Suzuki M, Kageyama S, Bunai T, Nagura K, Igarashi H, et al. PIK3CA mutation and amplification in human lung cancer. Pathol Int. 2007;57(10):664–71.
Spoerke JM, O’Brien C, Huw L, Koeppen H, Fridly J, Brachmann RK, et al. Phosphoinositide 3-kinase (PI3K) pathway alterations are associated with histologic subtypes and are predictive of sensitivity to PI3K inhibitors in lung cancer preclinical models. Clin Cancer Res. 2012;18(24):6771–83.
Singh VK, Jia Z. Targeting synuclein-γ to counteract drug resistance in cancer. Expert Opin Ther Targets. 2008;12(1):59–68.
Duyckaerts C. Neurodegenerative lesions: seeding and spreading. Rev Neurol. 2013;169(10):825–33.
Lavedan C, Leroy E, Dehejia A, Buchholtz S, Dutra A, Nussbaum RL, et al. Identification, localization and characterization of the human γ-synuclein gene. Hum Genet. 1998;103(1):106–12.
Liu J, Spence MJ, Zhang YL, Jiang Y, Liu YE, Shi YE. Transcriptional suppression of synuclein γ (SNCG) expression in human breast cancer cells by the growth inhibitory cytokine oncostatin M. Breast Cancer Res Treatment. 2000;62(2):99–107.
Ahmad M, Attoub S, Singh MN, Martin FL, El-Agnaf OM. γ-synuclein and the progression of cancer. FASEB J. 2007;21(13):3419–30.
Liu H, Liu W, Wu Y, Zhou Y, Xue R, Luo C, et al. Loss of epigenetic control of synuclein-γ gene as a molecular indicator of metastasis in a wide range of human cancers. Cancer Res. 2005;65(17):7635–43.
Jiang Y, Liu YE, Lu A, Gupta A, Goldberg ID, Liu J, et al. Stimulation of estrogen receptor signaling by γ synuclein. Cancer Res. 2003;63(14):3899–903.
Jia T, Liu YE, Liu J, Shi YE. Stimulation of breast cancer invasion and metastasis by synuclein γ. Cancer Res. 1999;59(3):742–7.
Gupta A, Inaba S, Wong OK, Fang G, Liu J. Breast cancer-specific gene 1 interacts with the mitotic checkpoint kinase BubR1. Oncogene. 2003;22(48):7593–9.
Du K, Tsichlis PN. Regulation of the Akt kinase by interacting proteins. Oncogene. 2005;24(50):7401–9.
Hashimoto M, Bar-On P, Ho G, Takenouchi T, Rockenstein E, Crews L, et al. Beta-synuclein regulates Akt activity in neuronal cells. A possible mechanism for neuroprotection in Parkinson’s disease. J Biol Chem. 2004;279(22):23622–9.
Brognard J, Clark AS, Ni Y, Dennis PA. Akt/protein kinase B is constitutively active in non-small cell lung cancer cells and promotes cellular survival and resistance to chemotherapy and radiation. Cancer Res. 2001;61(10):3986–97.
Toulany M, Rodemann HP. Phosphatidylinositol 3-kinase/Akt signaling as a key mediator of tumor cell responsiveness to radiation. Semin Cancer Biol. 2015;35:180–90.
Vivanco I, Sawyers CL. The phosphatidylinositol 3-kinase–AKT pathway in human cancer. Nat Rev Cancer. 2002;2(7):489–501.
Malanga D, Scrima M, De Marco C, Fabiani F, De Rosa N, De Gisi S, et al. Activating E17K mutation in the gene encoding the protein kinase AKT in a subset of squamous cell carcinoma of the lung. Cell Cycle. 2008;7(5):665–9.
Carpten JD, Faber AL, Horn C, Donoho GP, Briggs SL, Robbins CM, et al. A transforming mutation in the pleckstrin homology domain of AKT1 in cancer. Nature. 2007;448(7152):439–44.
Beck JT, Ismail A, Tolomeo C. Targeting the phosphatidylinositol 3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) pathway: an emerging treatment strategy for squamous cell lung carcinoma. Cancer Treat Rev. 2014;40(8):980–9.
Gadgeel SM, Wozniak A. Preclinical rationale for PI3K/Akt/mTOR pathway inhibitors as therapy for epidermal growth factor receptor inhibitor-resistant non–small-cell lung cancer. Clin Lung Cancer. 2013;14(4):322–32.
Meng J, Dai B, Fang B, Bekele BN, Bornmann WG, Sun D, et al. Combination treatment with MEK and AKT inhibitors is more effective than each drug alone in human non-small cell lung cancer in vitro and in vivo. PLoS One. 2010;5(11):e14124.
Ji H, Liu YE, Jia T, Wang M, Liu J, Xiao G, et al. Identification of a breast cancer-specific gene, BCSG1, by direct differential cDNA sequencing. Cancer Res. 1997;57(4):759–64.
Buchman VL, Hunter HJ, Pinõn LG, Thompson J, Privalova EM, Ninkina NN, et al. Persyn, a member of the synuclein family, has a distinct pattern of expression in the developing nervous system. J Neurosci. 1998;18(22):9335–41.
Wu K, Quan Z, Weng Z, Li F, Zhang Y, Yao X, et al. Expression of neuronal protein synuclein γ gene as a novel marker for breast cancer prognosis. Breast Cancer Res Treatment. 2007;101(3):259–67.
Liang W, Miao S, Zhang B, He S, Shou C, Manivel P, et al. Synuclein γ protects Akt and mTOR and renders tumor resistance to Hsp90 disruption. Oncogene. 2015;34(18):2398–405.
Author information
Authors and Affiliations
Corresponding author
Additional information
Zengxia Ma and Jianyi Niu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ma, Z., Niu, J., Sun, E. et al. Gamma-synuclein binds to AKT and promotes cancer cell survival and proliferation. Tumor Biol. 37, 14999–15005 (2016). https://doi.org/10.1007/s13277-016-5371-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-016-5371-9