Abstract
Background
Ovarian cancer (OV) is a gynecologic tumor with high mortality due to its drug resistance. In most cases, simple standard care is not satisfactory. Ferroptosis as a non-apoptotic form of cell death may contribute to the selective elimination of certain tumor cells. Therefore, ferroptosis has been considered to be potential therapy in anti-tumor.
Objective
Here, we report that erastin inhibits OV cell viability by inducing ferroptosis and identified the ferroptosis resistance effect of Nrf2 in OV.
Results
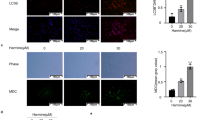
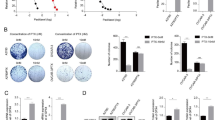
We demonstrated that erastin-induced cells release more Fe2+, malondialdehyde (MDA), and less glutathione (GSH). Nevertheless, we found that erastin-induced ferroptosis in OV cells was inhibited by overexpressed Nrf2. Mechanistically, Nrf2 promotes system xc-, increases intracellular glutamate and GSH, and thus resists tumor ferroptosis. Specifically, Nrf2 promotes system XC mainly by increasing the expression of SLC7A11.
Conclusion
This study revealed a novel cancer-promoting mechanism of Nrf2, mainly enhanced ferroptosis resistance in OV.






Similar content being viewed by others
References
Bao L et al (2017) ABCF2, an Nrf2 target gene, contributes to cisplatin resistance in ovarian cancer cells. Mol Carcinog 56(6):1543–1553
Basuli D et al (2017) Iron addiction: a novel therapeutic target in ovarian cancer. Oncogene 36(29):4089–4099
Bersuker K et al (2019) The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis. Nature 575(7784):688–692
Cao Y (2019) Adipocyte and lipid metabolism in cancer drug resistance. J Clin Invest 129(8):3006–3017
Chen X et al (2021) Cellular degradation systems in ferroptosis. Cell Death Differ 28(4):1135–1148
DeNicola GM et al (2011) Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature 475(7354):106–109
Dixon SJ et al (2012) Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149(5):1060–1072
Dong H et al (2020) Nrf2 inhibits ferroptosis and protects against acute lung injury due to intestinal ischemia reperfusion via regulating SLC7A11 and HO-1. Aging (albany NY) 12(13):12943–12959
Gao L et al (2021) Overcoming anti-cancer drug resistance via restoration of tumor suppressor gene function. Drug Resist Updat 57:100770
Grunewald T, Ledermann JA (2017) Targeted therapies for ovarian cancer. Best Pract Res Clin Obstet Gynaecol 41:139–152
He F, Ru X, Wen T (2020) NRF2, a transcription factor for stress response and beyond. Int J Mol Sci. https://doi.org/10.3390/ijms21134777
Hirschhorn T, Stockwell BR (2019) The development of the concept of ferroptosis. Free Radic Biol Med 133:130–143
Jiang L et al (2015) Ferroptosis as a p53-mediated activity during tumour suppression. Nature 520(7545):57–62
Kanlikilicer P et al (2018) Exosomal miRNA confers chemo resistance via targeting Cav1/p-gp/M2-type macrophage axis in ovarian cancer. EBioMedicine 38:100–112
Kitamura H, Motohashi H (2018) NRF2 addiction in cancer cells. Cancer Sci 109(4):900–911
Kopacz A et al (2020) Beyond repression of Nrf2: an update on Keap1. Free Radic Biol Med 157:63–74
Koppula P et al (2018) Amino acid transporter SLC7A11/xCT at the crossroads of regulating redox homeostasis and nutrient dependency of cancer. Cancer Commun (lond) 38(1):12
Koppula P, Zhuang L, Gan B (2021) Cystine transporter SLC7A11/xCT in cancer: ferroptosis, nutrient dependency, and cancer therapy. Protein Cell 12(8):599–620
Kuroki L, Guntupalli SR (2020) Treatment of epithelial ovarian cancer. BMJ 371:m3773
La Rosa P et al (2021) The Nrf2 induction prevents ferroptosis in Friedreich’s Ataxia. Redox Biol 38:101791
Lee S, Rauch J, Kolch W (2020) Targeting MAPK signaling in cancer: mechanisms of drug resistance and sensitivity. Int J Mol Sci. https://doi.org/10.3390/ijms21031102
Lei G et al (2020) The role of ferroptosis in ionizing radiation-induced cell death and tumor suppression. Cell Res 30(2):146–162
Li JH et al (2014) starBase v2.0: decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA interaction networks from large-scale CLIP-Seq data. Nucleic Acids Res 42(Database issue):D92–D97
Liu J, Xia X, Huang P (2020a) xCT: a critical molecule that links cancer metabolism to redox signaling. Mol Ther 28(11):2358–2366
Liu N, Lin X, Huang C (2020b) Activation of the reverse transsulfuration pathway through NRF2/CBS confers erastin-induced ferroptosis resistance. Br J Cancer 122(2):279–292
Liu MR, Zhu WT, Pei DS (2021) System Xc(-): a key regulatory target of ferroptosis in cancer. Invest New Drugs 39(4):1123–1131
Lo M, Wang YZ, Gout PW (2008) The x(c)- cystine/glutamate antiporter: a potential target for therapy of cancer and other diseases. J Cell Physiol 215(3):593–602
Ma Q (2013) Role of nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol 53:401–426
Ma S et al (2016) Ferroptosis is induced following siramesine and lapatinib treatment of breast cancer cells. Cell Death Dis 7:e2307
Ma L et al (2021) Targeting SLC3A2 subunit of system XC(-) is essential for m(6)A reader YTHDC2 to be an endogenous ferroptosis inducer in lung adenocarcinoma. Free Radic Biol Med 168:25–43
Orr B, Edwards RP (2018) Diagnosis and treatment of ovarian cancer. Hematol Oncol Clin North Am 32(6):943–964
Roh JL et al (2017) Nrf2 inhibition reverses the resistance of cisplatin-resistant head and neck cancer cells to artesunate-induced ferroptosis. Redox Biol 11:254–262
Sajadimajd S, Khazaei M (2018) Oxidative stress and cancer: the role of Nrf2. Curr Cancer Drug Targets 18(6):538–557
Sepand MR et al (2020) Targeting non-apoptotic cell death in cancer treatment by nanomaterials: recent advances and future outlook. Nanomedicine 29:102243
Shin D et al (2018) Nrf2 inhibition reverses resistance to GPX4 inhibitor-induced ferroptosis in head and neck cancer. Free Radic Biol Med 129:454–462
Stewart C, Ralyea C, Lockwood S (2019) Ovarian cancer: an integrated review. Semin Oncol Nurs 35(2):151–156
Su Y et al (2020) Ferroptosis, a novel pharmacological mechanism of anti-cancer drugs. Cancer Lett 483:127–136
Sun X et al (2016) Activation of the p62-Keap1-NRF2 pathway protects against ferroptosis in hepatocellular carcinoma cells. Hepatology 63(1):173–184
Tang D et al (2019) The molecular machinery of regulated cell death. Cell Res 29(5):347–364
Tang D et al (2021) Ferroptosis: molecular mechanisms and health implications. Cell Res 31(2):107–125
Torre LA et al (2018) Ovarian cancer statistics, 2018. CA Cancer J Clin 68(4):284–296
Xu T et al (2019) Molecular mechanisms of ferroptosis and its role in cancer therapy. J Cell Mol Med 23(8):4900–4912
Ye J et al (2018) Non-apoptotic cell death in malignant tumor cells and natural compounds. Cancer Lett 420:210–227
Zhang H et al (2020a) CAF secreted miR-522 suppresses ferroptosis and promotes acquired chemo-resistance in gastric cancer. Mol Cancer 19(1):43
Zhang L et al (2020b) IMCA induces ferroptosis mediated by SLC7A11 through the AMPK/mTOR pathway in colorectal cancer. Oxid Med Cell Longev 2020:1675613
Acknowledgements
We greatly appreciate the support from the Chongqing fourth people's Hospital and Chongqing General Hospital.
Author information
Authors and Affiliations
Contributions
YL performed experiments and participated in the initial draft. RY performed experiments and participated in the initial draft. ML performed experiments and analyzed the data. CC designed the study and supervised the progress. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Yongkang Li declares he has no conflict of interest. Rong Yin declares she has no conflict of interest. Miao Liang declares she has no conflict of interest. Cheng Chen declares he has no conflict of interest.
Ethical approval
The present study was approved by the Ethics Committee of Chongqing General Hospital. The research has been carried out in accordance with the World Medical Association Declaration of Helsinki.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Y., Yin, R., Liang, M. et al. Nrf2 suppresses erastin-induced ferroptosis through activating system Xc(-) in ovarian cancer. Mol. Cell. Toxicol. 20, 85–95 (2024). https://doi.org/10.1007/s13273-022-00322-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-022-00322-1