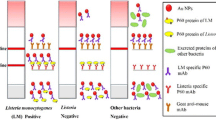
Abstract
Listeria monocytogenes is a food-borne bacterium that causes listeriosis upon the ingestion of contaminated food. Traditional methods to detect L. monocytogenes require pre-enrichment broths to increase its concentration. To improve the screening of contaminated food and prevent listeriosis outbreaks, rapid, specific and sensitive assays are needed to detect L. monocytogenes. This study developed a prototype lateral flow immunochromatographic assay (LFIA) employing antibodies against L. monocytogenes Internalin A (InlA) and Internalin B (InlB) proteins, that are involved in non-phagocytic cell invasion. The following antibodies were used to capture L. monocytogenes antigenic targets: mouse anti-Internalin A monoclonal antibody (MAb-2D12) conjugated to colloidal gold nanoparticles and a mouse anti-Internalin B polyclonal antibody. This test was able to detect pure L. monocytogenes from culture with a limit of detection (LOD) ranging from 5.9 × 103 to 1.5 × 104 CFU/mL. In milk artificially contaminated with L. monocytogenes, the LOD was 1 × 105 CFU/mL. This prototype test discriminated L. monocytogenes from other bacterial species (Listeria innocua, Enterobacter cloacae, Bacillus cereus). Results indicate that this LFIA developed using antibodies against L. monocytogenes InlA and InlB proteins is a sensitive and specific tool that can be potentially useful to rapidly detect L. monocytogenes in contaminated food.





Similar content being viewed by others
Data Availability
Not Applicable.
Code Availability
Not Applicable.
Abbreviations
- AuNP:
-
Colloidal Gold Nanoparticle
- BHI:
-
Brain Heart Infusion
- InlA:
-
Internalin A
- InlB:
-
Internalin B
- LFIA:
-
Lateral Flow Immunochromatographic Assay
- LM:
-
Listeria Monocytogenes
- LOD:
-
Limit of Detection
- MAb:
-
Monoclonal Antibody
- NC:
-
Nitrocellulose Membrane
- OD:
-
Optical Density
- PAb:
-
Polyclonal Antibody
- TSB:
-
Tryptic Soy Broth
References
Banada PP, Bhunia AK (2008) Antibodies and immunoassays for detection of bacterial pathogens. Princ bact detect biosensors recognit recept microsystems. Springer, London, pp 567–602
Bhunia AK (2008) Biosensors and bio-based methods for the separation and detection of foodborne pathogens. Adv Food Nutr Res 54:1–44. https://doi.org/10.1016/S1043-4526(07)00001-0
Bhunia AK (2014) One day to one hour: How quickly can foodborne pathogens be detected? Future Microbiol 9:935–946. https://doi.org/10.2217/fmb.14.61
Bhunia AK (2018) Foodborne microbial pathogens. Springer, New York
Bierne H, Cossart P (2007) Listeria monocytogenes Surface Proteins: from genome predictions to function. Microbiol Mol Biol Rev 71:377–397. https://doi.org/10.1128/mmbr.00039-06
Carlin CR, Liao J, Weller D et al (2021) Listeria cossartiae sp Nov, listeria immobilis sp nov, listeria portnoyi sp nov and listeria rustica sp nov, isolated from agricultural water and natural environments. Int J Syst Evol Microbiol. https://doi.org/10.1099/ijsem.0.004795
Cho I, Irudayaraj J (2013) Lateral-flow enzyme immunoconcentration for rapid detection of listeria monocytogenes. Anal Bioanal Chem 405:3313–3319. https://doi.org/10.1007/s00216-013-6742-3
Cho IH, Bhunia A, Irudayaraj J (2015) Rapid pathogen detection by lateral-flow immunochromatographic assay with gold nanoparticle-assisted enzyme signal amplification. Int J Food Microbiol 206:60–66. https://doi.org/10.1016/j.ijfoodmicro.2015.04.032
Drolia R, Bhunia AK (2019) Crossing the intestinal barrier via listeria adhesion protein and Internalin A. Trends Microbiol 27:408–425. https://doi.org/10.1016/j.tim.2018.12.007
Dwivedi HP, Jaykus LA (2011) Detection of pathogens in foods: The current state-of-the-art and future directions. Crit Rev Microbiol 37:40–63. https://doi.org/10.3109/1040841X.2010.506430
Gnanou Besse N, Barre L, Buhariwalla C et al (2010) The overgrowth of Listeria monocytogenes by other Listeria spp. in food samples undergoing enrichment cultivation has a nutritional basis. Int J Food Microbiol 136:345–351. https://doi.org/10.1016/j.ijfoodmicro.2009.10.025
Gussenhoven GC, Van Der Hoorn MAWG, Goris MGA et al (1997) LEPTO dipstick, a dipstick assay for detection of Leptospira-specific immunoglobulin M antibodies in human sera. J Clin Microbiol 35:92–97
Hoffmann S, Batz MB, Junior Morris G (2014) Annual Cost of Illness and Quality-Adjusted Life Year Losses in the United States Due to 14 Foodborne Pathogens. J Food Prot 75:1292–1302. https://doi.org/10.4315/0362-028X
Lathrop AA, Bailey TW, Kim K-P, Bhunia AK (2014) Pathogen-specific antigen target for production of antibodies produced by comparative genomics. Antib Technol J 4:13–22. https://doi.org/10.2147/anti.s54848
Li Q, Zhang S, Cai Y et al (2017) Rapid detection of Listeria monocytogenes using fluorescence immunochromatographic assay combined with immunomagnetic separation technique. Int J Food Sci Technol 52:1559–1566. https://doi.org/10.1111/ijfs.13428
Liu H, Du X, Zang Y-X et al (2017) SERS-based lateral flow strip biosensor for simultaneous detection of listeria monocytogenes and salmonella enterica serotype enteritidis. J Agric Food Chem 65:10290–10299. https://doi.org/10.1021/acs.jafc.7b03957
Lv X, Huang Y, Liu D et al (2019) Multicolor and ultrasensitive ELISA based on fluorescence hybrid chain reaction for simultaneous detection of pathogens. J Agric Food Chem 67:9390–9398. https://doi.org/10.1021/acs.jafc.9b03414
Mclauchlin J (1990) Human listeriosis in Britain, 1967–85, a summary of 722 cases: 1. Listeriosis during pregnancy and in the newborn. Epidemiol Infect 104:181–189. https://doi.org/10.1017/S0950268800059343
Mendonça M, Conrad NL, Conceição FR et al (2012) Highly specific fiber optic immunosensor coupled with immunomagnetic separation for detection of low levels of Listeria monocytogenes and L ivanovii. BMC Microbiol. https://doi.org/10.1186/1471-2180-12-275
O’Connor L, O’Leary M, Leonard N et al (2010) The characterization of Listeria spp. isolated from food products and the food-processing environment. Lett Appl Microbiol 51:490–498. https://doi.org/10.1111/j.1472-765X.2010.02928.x
Oravcová K, Trnčíková T, Kuchta T, Kaclíková E (2008) Limitation in the detection of Listeria monocytogenes in food in the presence of competing Listeria innocua. J Appl Microbiol 104:429–437. https://doi.org/10.1111/j.1365-2672.2007.03554.x
Phraephaisarn C, Khumthong R, Takahashi H et al (2017) A novel biomarker for detection of Listeria species in food processing factory. Food Control 73:1032–1038. https://doi.org/10.1016/j.foodcont.2016.10.001
Radoshevich L, Cossart P (2018) Listeria monocytogenes: towards a complete picture of its physiology and pathogenesis. Nat Rev Microbiol 16:32–46. https://doi.org/10.1038/nrmicro.2017.126
Schlech WF (2019) Epidemiology and clinical manifestations of listeria monocytogenes infection. Gram-postive pathogens. ASM press, Washington, pp 793–802
Snapir YM, Vaisbein E, Nassar F (2006) Low virulence but potentially fatal outcome-Listeria ivanovii. Eur J Intern Med 17:286–287. https://doi.org/10.1016/j.ejim.2005.12.006
Snowden K, Hommel M (1991) Antigen detection immunoassay using dipsticks and colloidal dyes. J Lmmunological Methods 140:57–65. https://doi.org/10.1016/0022-1759(91)90126-z
Tully E, Higson SP, O’Kennedy R (2008) The development of a “labeless” immunosensor for the detection of Listeria monocytogenes cell surface protein, Internalin B. Biosens Bioelectron 23:906–912. https://doi.org/10.1016/j.bios.2007.09.011
Ueda S, Iwase M, Kuwabara Y (2013) Evaluation of Immunochromatography for the rapid and specific identification of listeria monocytogenes from food. Biocontrol Sci 18:157–161. https://doi.org/10.4265/bio.18.157
Uusitalo S, Kögler M, Välimaa AL et al (2016) Detection of: Listeria innocua on roll-to-roll produced SERS substrates with gold nanoparticles. RSC Adv 6:62981–62989. https://doi.org/10.1039/c6ra08313g
Wu Z (2019) Simultaneous detection of listeria monocytogenes and salmonella typhimurium by a SERS-based lateral flow immunochromatographic assay. Food Anal Methods 12:1086–1091. https://doi.org/10.1007/s12161-019-01444-4
Acknowledgements
Students’ fellowships were granted by: the Brazilian National Research Council (grant # 133188/2016-7 2016-2018), the Brazilian Coordination for the Improvement of Higher Education Personnel (CAPES grant # 88882.385457/2007-01 2019-2023) and the Alliances Program for Education and Training (Scholarships Brazil—PAEC OAS-GCUB). AK is a research fellow from the Brazilian Research Council/CNPq (“Bolsista de Produtividade em Pesquisa, Grant # 314366/2020-2). MMAS is PVN-II research fellow from FAPEAM/Amazonas (PECTI-AM/SAÚDE Program, Grant #004/2020) and a research fellow from the Brazilian Research Council/CNPq (“Bolsista de Produtividade em Pesquisa, Grant # 311986-2019-6).
Funding
Not Applicable.
Author information
Authors and Affiliations
Contributions
Conception and design: RSM, ESF, MM, FRC, MMAS, SBS; Acquisition of data: RSM, ESF, AV, DRS, ANM, TGS; Analysis and interpretation of data: LLL, ESF, RSM, AK, MMAS, SBS; Drafting the manuscript: LLL, ESF, SBS, MMAS; Revising the manuscript critically: LLL, RSM, AK, MM, FRC, MMAS, SBS; Approval of the final manuscript: LLL, ESF, MM, ANM, AV, DRS, TGS, RSM, MMAS, FRC, AK, SBS.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Consent to participate
Not Applicable.
Consent for publication
Not Applicable.
Ethics approval
Not Applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lopes-Luz, L., Silva-Filho, E., Mendonça, M. et al. Combined antibodies against internalins A and B proteins have potential application in immunoassay for detection of Listeria monocytogenes. J Food Sci Technol 60, 123–131 (2023). https://doi.org/10.1007/s13197-022-05597-9
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-022-05597-9