Abstract
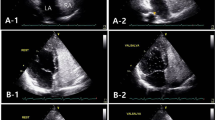
Previous investigations have suggested that patients with patent foramen ovale (PFO) often have an atrial dysfunction, like to that observed in patients with atrial fibrillation (AF) which may concur to an increased risk of cryptogenic stroke. The aim of the study is to compare the atrial resident time (Rt) of PFO patients to those with sinus rhythm (SR) and AF using patient-specific 3D computational fluid dynamics (CFD) analysis. Models of left atrium (LA) hemodynamics were obtained from time-resolved CT scans and transthoracic echocardiography (TTE). Enrolled patients were divided into three groups: 30 healthy subjects with SR, 30 with PFO, and 30 with AF without PFO. Blood stasis was evaluated by determining the blood residence time (Rt) distribution in the LA and left atrial appendage (LAA). Overall, 90 patients (mean age 47.4 ± 7.5 years, 51 males) were included into the analysis. PFO patients exhibit higher mean Rt values compared to healthy subjects (2.65 ± 0.2 vs 1.5 ± 0.2 s). Conversely, AF patients presented higher Rt when compared to PFO patients (2.9 ± 0.3 vs 2.3 ± 0.2 s). Moreover, PFO patients presenting cerebral lesions at magnetic resonance imaging have a higher Rt compared to those without (2.9 ± 0.3 vs 2.3 ± 0.2 s, respectively, p < 0.001). PFO patients have a higher degree of atrial Rt compared to healthy subjects similar to that observed in AF patients. The higher mean LA Rt values offer an insight into the pathophysiological mechanism linking PFO with cryptogenic stroke and might be a marker of high-risk PFOs.



Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Rigatelli G, Zuin M. Leonardo da Vinci and patent foramen ovale: an historical perspective. Int J Cardiol. 2016;222:826. https://doi.org/10.1016/j.ijcard.2016.08.079.
Lethen H, Flachskampf FA, Schneider R, Sliwka U, Köhn G, Noth J, Hanrath P. Frequency of deep vein thrombosis in patients with patent foramen ovale and ischemic stroke or transient ischemic attack. Am J Cardiol. 1997;80:1066–9. https://doi.org/10.1016/s0002-9149(97)00604-8.
Samuel S, Reddy ST, Parsha KN, Nguyen T, Reddy Indupuru HK, Sharrief AZ, Zhu L, McCullough LD, Savitz SI. Routine surveillance of pelvic and lower extremity deep vein thrombosis in stroke patients with patent foramen ovale. J Thromb Thrombolysis. 2021;51:1150–6. https://doi.org/10.1007/s11239-020-02262-w.
Rigatelli G, Aggio S, Cardaioli P, Braggion G, Giordan M, Dell’avvocata F, Chinaglia M, Rigatelli G, Roncon L, Chen JP. Left atrial dysfunction in patients with patent foramen ovale and atrial septal aneurysm: an alternative concurrent mechanism for arterial embolism? JACC Cardiovasc Interv. 2009;2:655–62. https://doi.org/10.1016/j.jcin.2009.05.010.
Rigatelli G, Zuin M, Adami A, Aggio S, Lanza D, d’Elia K, Braggion G, Russo M, Mazza A, Roncon L. Left atrial enlargement as a maker of significant high-risk patent foramen ovale. Int J Cardiovasc Imaging. 2019;35:2049–56. https://doi.org/10.1007/s10554-019-01666-x.
Rigatelli G, Zuin M, Fong A. Computational flow dynamic analysis of right and left atria in patent foramen ovale: potential links with atrial fibrillation. J Atr Fibrillation. 2018;10:1852. https://doi.org/10.4022/jafib.1852.
Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, Flachskampf FA, Foster E, Goldstein SA, Kuznetsova T, Lancellotti P, Muraru D, Picard MH, Rietzschel ER, Rudski L, Spencer KT, Tsang W, Voigt JU. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2015;28:1-39.e14. https://doi.org/10.1016/j.echo.2014.10.003.
Silvestry FE, Cohen MS, Armsby LB, Burkule NJ, Fleishman CE, Hijazi ZM, Lang RM, Rome JJ, Wang Y, American Society of Echocardiography; Society for Cardiac Angiography and Interventions. Guidelines for the echocardiographic assessment of atrial septal defect and patent foramen ovale: from the American Society of Echocardiography and Society for Cardiac Angiography and Interventions. J Am Soc Echocardiogr. 2015;28:910–58.
Sanatkhani S, Nedios S, Menon PG, Bollmann A, Hindricks G, Shroff SG. Subject-specific calculation of left atrial appendage blood-borne particle residence time distribution in atrial fibrillation. Front Physiol. 2021;12: 633135. https://doi.org/10.3389/fphys.2021.633135.
Bieging ET, Morris A, Chang L, Dagher L, Marrouche NF, Cates J. Statistical shape analysis of the left atrial appendage predicts stroke in atrial fibrillation. Int J Cardiovasc Imaging. 2021;37:2521–7. https://doi.org/10.1007/s10554-021-02262-8.
Jensen JA. Estimation of blood velocities using ultrasound: a signal processing approach. New York, NY, USA: Cambridge University Press; 1996.
Zhang LT, Gay M. Characterizing left atrial appendage functions in sinus rhythm and atrial fibrillation using computational models. J Biomech. 2008;41:2515–23. https://doi.org/10.1016/j.jbiomech.2008.05.012.
Feng L, Gao H, Qi N, Danton M, Hill NA, Luo X. Fluid-structure interaction in a fully coupled three-dimensional mitral-atrium-pulmonary model. Biomech Model Mechanobiol. 2021;20:1267–95. https://doi.org/10.1007/s10237-021-01444-6.
Gao H, Wang H, Berry C, Luo X, Griffith BE. Quasi-static image-based immersed boundary-finite element model of left ventricle under diastolic loading. Int J Numer Methods Biomed Eng. 2014;30:1199–222.
Griffith BE, Luo X. Hybrid finite difference/finite element immersed boundary method. Int J Numer Methods Biomed Eng. 2017;33: e2888.
Morales Ferez X, Mill J, Juhl KA, Acebes C, Iriart X, Legghe B, Cochet H, De Backer O, Paulsen RR, Camara O. Deep learning framework for real-time estimation of in-silico thrombotic risk indices in the left atrial appendage. Front Physiol. 2021;12: 694945. https://doi.org/10.3389/fphys.2021.694945.
Vedula V, George R, Younes L, Mittal R. Hemodynamics in the left atrium and its effect on ventricular flow patterns. J Biomech Eng. 2015;137: 111003.
Dueñas-Pamplona J, García JG, Sierra-Pallares J, Ferrera C, Agujetas R, López-Mínguez JR. A comprehensive comparison of various patient-specific CFD models of the left atrium for atrial fibrillation patients. Comput Biol Med. 2021;133: 104423. https://doi.org/10.1016/j.compbiomed.2021.104423.
Habibi M, Lima JA, Khurram IM, Zimmerman SL, Zipunnikov V, Fukumoto K, Spragg D, Ashikaga H, Rickard J, Marine JE, Calkins H, Nazarian S. Association of left atrial function and left atrial enhancement in patients with atrial fibrillation: cardiac magnetic resonance study. Circ Cardiovasc Imaging. 2015;8: e002769. https://doi.org/10.1161/CIRCIMAGING.114.002769.
Garcia-Villalba M, Rossini L, Gonzalo A, Vigneault D, Kahn AM, Flores O, McVeigh E, del Alamo de Pedro JC. Abstract 15017: Patient-specific mapping of left atrial thrombosis risk by computational fluid dynamics. Circulation. 2018;138:A15017.
Aune E, Baekkevar M, Roislien J, Rodevand O, Otterstad JR. Normal reference ranges for left and right atrial volume indexes and ejection fractions obtained with real-time three-dimensional echocardiography. Eur J Echocardiogr. 2009;10:738–44.
Cotter PE, Martin PJ, Pugh PJ, Warburton EA, Cheriyan J, Belham M. Increased incidence of interatrial block in younger adults with cryptogenic stroke and patent foramen ovale. Cerebrovasc Dis Extra. 2011;1:36–43.
Kim TW, Jung SW, Song IU, Koo J, Choi HS, Lee KS, Park JW, Park HJ, Kim JS. Left atrial dilatation is associated with severe ischemic stroke in men with non-valvular atrial fibrillation. J Neurol Sci. 2015;354:97–102.
Lee MJ, Park SJ, Yoon CH, Hwang JW, Ryoo S, Kim SJ, Kim GM, Chung CS, Lee KH, Bang OY. Association of left atrial enlargement with cortical infarction in subjects with patent foramen ovale. J Stroke. 2016;18:304–11.
Rigatelli G, Dell’Avvocata F, Cardaioli P, Giordan M, Braggion G, Aggio S, Chinaglia M, Mandapaka S, Kuruvilla J, Chen JP, Nanjundappa A. Permanent right-to-left shunt is the key factor in managing patent foramen ovale. J Am Coll Cardiol. 2011;58:2257–61.
Hashemi J, Rai S, Ghafghazi S, Berson RE. Blood residence time to assess significance of coronary artery stenosis. Sci Rep. 2020;10:11658. https://doi.org/10.1038/s41598-020-68292-9.
Rossini L, Martinez-Legazpi P, Vu V, Fernandez-Friera L, Perez Del Villar C, Rodriguez-Lopez S, et al. A clinical method for mapping and quantifying blood stasis in the left ventricle. J Biomech. 2016;49:2152–61. https://doi.org/10.1016/j.jbiomech.2015.11.049.
Author information
Authors and Affiliations
Contributions
Gianluca Rigatelli and Marco Zuin: conceptualization, draft the manuscript, performed the analysis. Loris Roncon: collected the data and supervised the manuscript. All authors read the final version and approved the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Ethical approval was waived due to the retrospective nature of the manuscripts and use of anonymized data.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rigatelli, G., Zuin, M. & Roncon, L. Increased Blood Residence Time as Markers of High-Risk Patent Foramen Ovale. Transl. Stroke Res. 14, 304–310 (2023). https://doi.org/10.1007/s12975-022-01045-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-022-01045-0