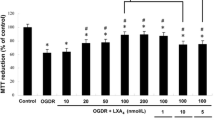
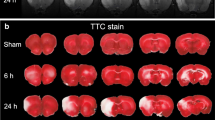
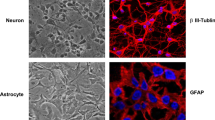
Abstract
Induction of ischemic preconditioning (IPC) represents a potential therapy against cerebral ischemia by activation of adaptive pathways and modulation of mitochondria to induce ischemic tolerance to various cells and tissues. Mitochondrial dysfunction has been ascribed to contribute to numerous neurodegenerative conditions and cerebral ischemia. Nuclear erythroid 2-related factor 2 (Nrf2) is a transcription factor that has traditionally been involved in upregulating cellular antioxidant systems to combat oxidative stress in the brain; however, the association of Nrf2 with mitochondria in the brain remains unclear. In the present study, we investigated the effects of Nrf2 on (i) IPC-induced protection of astrocytes; (ii) OXPHOS protein expression; and (iii) mitochondrial supercomplex formation.
Oxygen-glucose deprivation (OGD) was used as an in vitro model of cerebral ischemia and IPC in cultured rodent astrocytes derived from WT C57Bl/6J and Nrf2−/− mice. OXPHOS proteins were probed via western blotting, and supercomplexes were determined by blue native gel electrophoresis.
IPC-induced cytoprotection in wild-type, but not Nrf2−/− mouse astrocyte cultures following a lethal duration of OGD. In addition, our results suggest that Nrf2 localizes to the outer membrane in non-synaptic brain mitochondria, and that a lack of Nrf2 in vivo produces altered supercomplex formation in mitochondria.
Our findings support a role of Nrf2 in mediating IPC-induced protection in astrocytes, which can profoundly impact the ischemic tolerance of neurons. In addition, we provide novel evidence for the association of Nrf2 to brain mitochondria and supercomplex formation. These studies offer new targets and pathways of Nrf2, which may be heavily implicated following cerebral ischemia.







Similar content being viewed by others
References
Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74:1124–36.
Schurr A, Reid KH, Tseng MT, West C, Rigor BM. Adaptation of adult brain tissue to anoxia and hypoxia in vitro. Brain Res. 1986;374:244–8.
Dave KR, Saul I, Prado R, Busto R, Perez-Pinzon MA. Remote organ ischemic preconditioning protect brain from ischemic damage following asphyxial cardiac arrest. Neurosci Lett. 2006;404:170–5.
Della-Morte D, Dave KR, DeFazio RA, Bao YC, Raval AP, Perez-Pinzon MA. Resveratrol pretreatment protects rat brain from cerebral ischemic damage via a sirtuin 1-uncoupling protein 2 pathway. Neuroscience. 2009;159:993–1002.
Kim EJ, Raval AP, Hirsch N, Perez-Pinzon MA. Ischemic preconditioning mediates cyclooxygenase-2 expression via nuclear factor-kappa b activation in mixed cortical neuronal cultures. Transl Stroke Res. 2010;1:40–7.
Perez-Alvarez A, Araque A. Astrocyte-neuron interaction at tripartite synapses. Curr Drug Targets. 2013;14:1220–4.
Johri A, Beal MF. Mitochondrial dysfunction in neurodegenerative diseases. J Pharmacol Exp Ther. 2012;342:619–30.
Wang B, Cao W, Biswal S, Dore S. Carbon monoxide-activated nrf2 pathway leads to protection against permanent focal cerebral ischemia. Stroke. 2011;42:2605–10.
Zhao X, Sun G, Zhang J, Strong R, Dash PK, Kan YW, et al. Transcription factor nrf2 protects the brain from damage produced by intracerebral hemorrhage. Stroke. 2007;38:3280–6.
Bell KF, Fowler JH, Al-Mubarak B, Horsburgh K, Hardingham GE. Activation of nrf2-regulated glutathione pathway genes by ischemic preconditioning. Oxidative Med Cell Longev. 2011;2011:689524.
Kitteringham NR, Abdullah A, Walsh J, Randle L, Jenkins RE, Sison R, et al. Proteomic analysis of nrf2 deficient transgenic mice reveals cellular defence and lipid metabolism as primary nrf2-dependent pathways in the liver. J Proteome. 2010;73:1612–31.
Ryan MT, Hoogenraad NJ. Mitochondrial-nuclear communications. Annu Rev Biochem. 2007;76:701–22.
Lenth RV. Statistical power calculations. J Anim Sci. 2007;85:E24–9.
Kaech S, Banker G. Culturing hippocampal neurons. Nat Protoc. 2006;1:2406–15.
Kim EJ, Raval AP, Perez-Pinzon MA. Preconditioning mediated by sublethal oxygen-glucose deprivation-induced cyclooxygenase-2 expression via the signal transducers and activators of transcription 3 phosphorylation. J Cereb Blood Flow Metab. 2008;28:1329–40.
SY X, YM W, Ji Z, Gao XY, Pan SYA. modified technique for culturing primary fetal rat cortical neurons. J Biomed Biotechnol. 2012;2012:803930.
Dave KR, DeFazio RA, Raval AP, Torraco A, Saul I, Barrientos A, et al. Ischemic preconditioning targets the respiration of synaptic mitochondria via protein kinase c epsilon. J Neurosci. 2008;28:4172–82.
Degasperi A, Birtwistle MR, Volinsky N, Rauch J, Kolch W, Kholodenko BN. Evaluating strategies to normalise biological replicates of western blot data. PLoS One. 2014;9:e87293.
Sunderhaus S, Eubel H, Braun HP. Two-dimensional blue native/blue native polyacrylamide gel electrophoresis for the characterization of mitochondrial protein complexes and supercomplexes. Methods Mol Biol. 2007;372:315–24.
Shih AY, Johnson DA, Wong G, Kraft AD, Jiang L, Erb H, et al. Coordinate regulation of glutathione biosynthesis and release by nrf2-expressing glia potently protects neurons from oxidative stress. J Neurosci. 2003;23:3394–406.
Stokfisz K, Ledakowicz-Polak A, Zagorski M, Zielinska M. Ischaemic preconditioning - current knowledge and potential future applications after 30 years of experience. Adv Med Sci. 2017;62:307–16.
Narayanan SV, Dave KR, Saul I, Perez-Pinzon MA. Resveratrol preconditioning protects against cerebral ischemic injury via nuclear erythroid 2-related factor 2. Stroke. 2015;
Lo SC, Hannink M. Pgam5 tethers a ternary complex containing keap1 and nrf2 to mitochondria. Exp Cell Res. 2008;314:1789–803.
Tondera D, Czauderna F, Paulick K, Schwarzer R, Kaufmann J, Santel A. The mitochondrial protein mtp18 contributes to mitochondrial fission in mammalian cells. J Cell Sci. 2005;118:3049–59.
Dennerlein S, Wang C, Rehling P. Plasticity of mitochondrial translation. Trends Cell Biol. 2017;27:712–21.
Medeiros DM. Assessing mitochondria biogenesis. Methods. 2008;46:288–94.
Maranzana E, Barbero G, Falasca AI, Lenaz G, Genova ML. Mitochondrial respiratory supercomplex association limits production of reactive oxygen species from complex i. Antioxid Redox Signal. 2013;19:1469–80.
Genova ML, Lenaz G. Functional role of mitochondrial respiratory supercomplexes. Biochim Biophys Acta. 2014;1837:427–43.
Andrienko TN, Pasdois P, Pereira GC, Ovens MJ, Halestrap AP. The role of succinate and ros in reperfusion injury—a critical appraisal. J Mol Cell Cardiol. 2017;110:1–14.
Vanden Hoek TL, Becker LB, Shao Z, Li C, Schumacker PT. Reactive oxygen species released from mitochondria during brief hypoxia induce preconditioning in cardiomyocytes. J Biol Chem. 1998;273:18092–8.
Sun JZ, Tang XL, Park SW, Qiu Y, Turrens JF, Bolli R. Evidence for an essential role of reactive oxygen species in the genesis of late preconditioning against myocardial stunning in conscious pigs. J Clin Invest. 1996;97:562–76.
Stauch KL, Purnell PR, Fox HS. Quantitative proteomics of synaptic and nonsynaptic mitochondria: Insights for synaptic mitochondrial vulnerability. J Proteome Res. 2014;13:2620–36.
Jain A, Lamark T, Sjottem E, Larsen KB, Awuh JA, Overvatn A, et al. P62/sqstm1 is a target gene for transcription factor nrf2 and creates a positive feedback loop by inducing antioxidant response element-driven gene transcription. J Biol Chem. 2010;285:22576–91.
Inami Y, Waguri S, Sakamoto A, Kouno T, Nakada K, Hino O, et al. Persistent activation of nrf2 through p62 in hepatocellular carcinoma cells. J Cell Biol. 2011;193:275–84.
Ichimura Y, Waguri S, Sou YS, Kageyama S, Hasegawa J, Ishimura R, et al. Phosphorylation of p62 activates the keap1-nrf2 pathway during selective autophagy. Mol Cell. 2013;51:618–31.
Piantadosi CA, Carraway MS, Babiker A, Suliman HB. Heme oxygenase-1 regulates cardiac mitochondrial biogenesis via nrf2-mediated transcriptional control of nuclear respiratory factor-1. Circ Res. 2008;103:1232–40.
Tufekci KU, Civi Bayin E, Genc S, Genc K. The nrf2/are pathway: a promising target to counteract mitochondrial dysfunction in Parkinson’s disease. Parkinson’s disease. 2011;2011:314082.
Acknowledgements
None
Funding
This study was funded by grants from the National Institutes of Health, National Institute of Neurological Disease and Stroke (NINDS) NS45676, NS054147, NS34773, F31 NS080344.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
There authors declare that they have no conflicts of interest.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Electronic Supplementary Materials
ESM 1
Supplementary Fig. 1 Genotyping of Nrf2−/− and WT C57Bl/6J mice. LAC Z denotes the portion of the Nrf2 locus that has been replaced with a LAC Z and Neomycin resistance gene cassette. Presence of LAC Z indicates animals that have this gene in place of exon 4 and exon 5 of the Nrf2 gene and denotes a knockout animal. Presence of WT allele denotes presence of Nrf2 gene. Presence of both LAC Z and WT gene indicate a heterozygous knockout animal. Genotyping was performed by Transnetyx, Inc. (Cordova, TN, USA). (TIFF 306 kb)
Rights and permissions
About this article
Cite this article
Narayanan, S.V., Dave, K.R. & Perez-Pinzon, M.A. Ischemic Preconditioning Protects Astrocytes against Oxygen Glucose Deprivation Via the Nuclear Erythroid 2-Related Factor 2 Pathway. Transl. Stroke Res. 9, 99–109 (2018). https://doi.org/10.1007/s12975-017-0574-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-017-0574-y