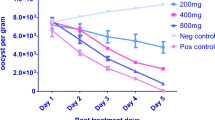
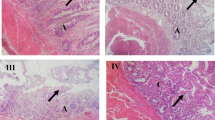
Abstract
Phytochemical compounds, plumbagin and thymol were evaluated for their efficacy against Theileria annulata using MTT cell viability assay. Plumbagin and thymol were found to be effective in preventing the proliferation of Theileria annulata infected bovine lymphocytes. The IC50 values of plumbagin and thymol were 0.019 µM and 0.009 µM, respectively. Plumbagin and thymol were found to be non-cytotoxic to the bovine peripheral blood mononuclear cells. However, both the compounds were found to have inhibitory effect on vero cell proliferation. Plumbagin had primarily anti-theilerial activity but thymol had primarily anti-mitotic activity. The in vitro efficacy and cell toxicity studies indicate the potential application of plumbagin, purified from Plumbago indica as a lead therapeutic molecule against T. annulata infection in cattle.
Graphical Abstract

Similar content being viewed by others
References
Arab HA, Fathi M, Mortezai E, Hosseinimehr SJ (2015) Chemoprotective effect of thymol against genotoxicity induced by bleomycin in human lymphocytes. Pharm Biomed Res 1(1):26–31. https://doi.org/10.18869/acadpub.pbr.1.1.26
Azhahianambi P, Madhan Mohan M, Madan N, Kumaran D, Latha Mala Priyadharshini M, Bharathi R, Senthilkumar TMA, Manoharan S (2021) Successful treatment of severe form of bovine tropical theileriosis in dairy cattle and genotyping of Theileria annulata isolates of Tamil Nadu, India. Vet Parasitol Reg Stud Reports 26:100628. https://doi.org/10.1016/j.vprsr.2021.100628
Bera AK, Bhattacharya D, Pan D, Dhara A, Kumar S, Ghosh SD (2010) Evaluation of economic Losses due to coccidiosis in the poultry industry in India. Agri Eco Res Rev 23(1):91–96. https://doi.org/10.22004/ag.econ.92156
Bilgic HB, Karagenç T, Shiels B, Tait A, Eren H, Weir W (2010) Evaluation of cytochrome b as a sensitive target for PCR based detection of T. annulata carrier animals. Vet Parasitol 174(3):341–347. https://doi.org/10.1016/j.vetpar.2010.08.025
Bothiraja C, Joshi PP, Dama GY, Pawar AP (2011) Rapid method for isolation of plumbagin, an alternative medicine from roots of Plumbago zeylanica. Eur J Integr Med 3:39–42. https://doi.org/10.1016/j.eujim.2011.02.008
Buvanesvaragurunathan K, Ganesh J, Nagul Kumar S, Porchezhiyan V, Radha A, Azhahianambi P, Pandikumar P, Ignacimuthu S (2022) In vitro growth inhibitory effect of selected 18β-glycyrrhetinic acid esters on Theileria annulata. Exp Parasitol. https://doi.org/10.1016/j.exppara.2022.108258
Campbel JDM, Spooner RL (1999) Macrophages behaving badly: infected cells and subversion of immune responses to Theileria annulata. Parasitol Today 15(1):10–16. https://doi.org/10.1016/S0169-4758%2898%2901359-3
Chatanga E, Mosssad E, Abubaker HA, Alnour SA, Katakura K, Nakao R, Salim B (2019) Evidence of multiple point mutations in Theileria annulata cytochrome b gene incriminated in buparvaquone treatment failure. Acta Trop 191:128–132. https://doi.org/10.1016/j.actatropica.2018.12.041
Chauhan M (2014) A review on morphology, phytochemistry and pharmacological activities of medicinal herb Plumbago zeylanica Linn. J Pharmacog Phytochem 3(2):95–118
Chen X, Reynolds CH (2002) Performance of similarity measures in 2D fragment-based similarity searching: comparison of structural descriptors and similarity coefficients. J Chem Inf Comput Sci 42(6):1407–1414. https://doi.org/10.1021/ci025531g
Cicek H, Eser M, Tandogan M (2009) Current status of ruminant theileriosis and its economical impact in Turkey. Turkiye Parazitol Derg 33(4):273–279
Deb DD, Parimala G, Saravana Devi S, Chakraborty T (2011) Effect of thymol on peripheral blood mononuclear cell PBMC and acute promyelocytic cancer cell line HL-60. Chem Biol Interact 193(1):97–106. https://doi.org/10.1016/j.cbi.2011.05.009
Devi PU, Solomon FE, Sharada AC (1994) In vivo tumor inhibitory and radiosensitizing effects of an Indian medicinal plant, Plumbago rosea on experimental mouse tumors. Ind J Exp Biol 32(8):523–528
Dominguez-Uscanga A, Aycart DF, Li K, Witola WH, Andrade Laborde JE (2021) Antiprotozoal activity of thymol and a thymol ester against Cryptosporidium parvum in cell culture. Int J Parasitol Drugs Drug Resist. https://doi.org/10.1016/j.ijpddr.2021.02.003
Farah HM, El Amin TH, El Hussein ARM, Khalid HE (2015) Assessment of anti-theilerial activity of the aqueous extract of Kigelia africana Unripe fruits against Theileria lestoquardi. Eur J Med Plants 5(1):101. https://doi.org/10.9734/EJMP/2015/10213
Farah HM, El Amin TH, Khalid HS, Hassan SM, El Hussein ARM (2012) In vitro activity of the aqueous extract of Gardenia ternifolia fruits against Theileria lestoquardi. J Med Plants Res 6(41):5447–5451. https://doi.org/10.5897/JMPR12.621
Fesharki RH (1991) Chemotherapeutic value of parvaquone and buparvaquone against Theileria annulata infection of cattle. Res Vet Sci 50(2):204–207. https://doi.org/10.1016/0034-5288(91)90107-y
Finney John D (1962) Probit analysis: a statistical treatment of the sigmoid response curve. Cambridge University Press, Cambridge
Grewal AS, Singh A, Kumar S, Kondal JK, Kanwar JR, Kapur J, Sawhney SMS (1997) Lowered vaccine dose for immunisation of calves against tropical theileriosis using Theileria annulata lymphoblasts. Trop Anim Health Prod 29(4):104S-108S. https://doi.org/10.1007/bf02632946
Gupta AC, Mohanty S, Saxena A, Maurya AK, Bawankule DU (2018) Plumbagin, a vitamin K3 analogue ameliorates malaria pathogenesis by inhibiting oxidative stress and inflammation. Inflammo Pharmacol 26(4):983–991. https://doi.org/10.1007/s10787-018-0465-1
Hazra B, Sarkar R, Bhattacharyya S, Ghosh PK, Chel G, Dinda B (2002) Synthesis of plumbagin derivatives and their inhibitory activities against Ehrlich ascites carcinoma in vivo and Leishmania donovani promastigotes in vitro. Phytother Res 16:133–137. https://doi.org/10.1002/ptr.867
Hoferi M, Buchbauer G, Jirovetz L (2009) Correlation of antimicrobial activities of various essential oils and their main aromatic volatile constituents. J Essent Oil Res 21:459–463. https://doi.org/10.1080/10412905.2009.9700218
Islam MT, Khalipha ABR, Bagchi R, Mondal M, Smrity SZ, Uddin SJ, Shilpi JA, Rouf R (2019) Anticancer activity of thymol: a literature-based review and docking study with emphasis on its anticancer mechanisms. IUBMB Life 71(1):9–19. https://doi.org/10.1002/iub.1935
Krishnamoorthy P, Akshata LG, Jacob SS, Suresh KP, Roy P (2021) Theileriosis prevalence status in cattle and buffaloes in India established by systematic review and meta-analysis. Ind J Anim Sci 91(4):269–279
Kumar S, Mohmad A, Parthasarathi BC, Fular A, Gupta S (2018) Current epidemiological status of bovine theileriosis in Indian scenario. Ann Clin Cytol Pathol 4(1):1090
Kumar S, Mina PR, Kumar R, Pal A, Ahmad A, Tandon S, Darokar MP (2021) 4-chlorothymol exerts antiplasmodial activity impeding redox defense system in Plasmodium falciparum. Front Pharmacol 12:628970. https://doi.org/10.3389/fphar.2021.628970
Lin LC, Yang LL, Chou CJ (2003) Cytotoxic naphthoquinones and plumbagic acid glucosides from Plumbago zeylanica. Phytochem 62(4):619–622. https://doi.org/10.1016/s0031-9422(02)00519-8
Metwaly EA, Saedia A, Ahl S, Soheir SM, Ahmed DA (2020) Evaluation of plumbagin as a potential therapeutic agent for murine Schistosomiasis mansoni. J Egypt Soc Parasitol 50(1):1–9. https://doi.org/10.21608/jesp.2020.88743
Mhadhbi M, Naouach A, Boumiza A, Chaabani MF, BenAbderrazik S, Darghouth MA (2010) In vivo evidence for the resistance of Theileria annulata to buparvaquone. Vet Parasitol 169(3–4):241–247. https://doi.org/10.1016/j.vetpar.2010.01.013
Mhadhbi M, Chaouch M, Ajroud K, Darghouth MA, BenAbderrazak S (2015) Sequence polymorphism of cytochrome b gene in Theileria annulata Tunisian isolates and its association with Buparvaquone treatment failure. PLoS ONE 10(6):e0129678. https://doi.org/10.1371/journal.pone.0129678
Minjauw B, McLeod A (2003) Tick-borne diseases and poverty. The impact of ticks and tick-borne diseases on the livelihood and marginal livestock owners in India and Eastern and Southern Africa. Research report, DFID Animal Health Programme, Centre of Tropical Veterinary Medicine, University of Edinburgh
Muleya W, Namangala B, Simuunza M (2012) Population genetic analysis and sub-structuring of Theileria parva in the northern and eastern parts of Zambia. Parasite Vector 5:255. https://doi.org/10.1186/1756-3305-5-255
NagoorMeeran MF, Javed H, Al Taee H, Azimullah S, Ojha SK (2017) Pharmacological properties and molecular mechanisms of thymol: prospects for its therapeutic potential and pharmaceutical development. Front Pharmacol 8:380. https://doi.org/10.3389/fphar.2017.00380
Narladkar BW (2018) Projected economic losses due to vector and vector-borne parasitic diseases in livestock of India and its significance in implementing the concept of integrated practices for vector management. Vet World 11:151–160. https://doi.org/10.14202/vetworld.2018.151-160
Oliveira CB, Meurer YS, Medeiros TL, Pohlit AM, Silva MV, Mineo TW, Andrade-Neto VF (2016) Anti-toxoplasma activity of estragole and thymol in murine models of congenital and non congenital toxoplasmosis. J Parasitol 102(3):369–376. https://doi.org/10.1645/15-848
Pradeepa V, Senthil-Nathan S, Sathish-Narayanan S, Selin-Rani S, Vasantha-Srinivasan P, Thanigaivel A, Ponsankar A, Edwin ES, Sakthi-Bagavathy M, Kalaivani K, Murugan K, Duraipandiyan V, Al-Dhabi NA (2016) Potential mode of action of a novel plumbagin as a mosquito repellent against the malarial vector Anopheles stephensi (Culicidae: Diptera). PesticBiochem Physiol 134:84–93. https://doi.org/10.1016/j.pestbp.2016.04.001
Purnell RE (1978) Theileria annulata as a hazard to cattle in countries on the northern Mediterranean littoral. Vet Res Commun 2:3–10. https://doi.org/10.1007/BF0229142
Raghuvanshi DS, Verma N, Singh SV, Khare S, Pal A, Negi AS (2019) Synthesis of thymol-based pyrazolines: an effort to perceive novel potent-antimalarials. Bioorg Chem 88:102933. https://doi.org/10.1016/j.bioorg.2019.102933
Rizk MA, El-Sayed SAE, Al-Araby M, Igarashi I (2022) Effect of methanolic extract from Capsicum annuum against the multiplication of several Babesia species and Theileria equi on in vitro cultures, and Babesia microti in mice. Vet World 15(1):76–82. https://doi.org/10.14202/vetworld.2022.76-82
Rizk MA, El-Sayed SAES, Igarashi I (2021) Effects of Methanolic extract from turmeric (Curcuma longa) against the in vitro multiplication of several Babesia species and Theileria equi. Parasitologia 1:188–196. https://doi.org/10.3390/parasitologia1040020
Saravanan BC, Sreekumar C, Bansal GC, Ray D, Rao JR, Mishra AK (2003) A rapid MTT colorimetric assay to assess the proliferative index of two Indian strains of Theileria annulata. Vet Parasitol 113(3–4):211–216. https://doi.org/10.1016/s0304-4017(03)00062-1
Sharifiyazdi H, Namazi F, Oryan A, Shahriari R, Razavi M (2012) Point mutations in the Theileria annulata cytochrome b gene is associated with buparvaquone treatment failure. Vet Parasitol 187(3–4):431–435. https://doi.org/10.1016/j.vetpar.2012.01.016
Sharma N, Shukla AK, Das M, Dubey VK (2012) Evaluation of plumbagin and its derivative as potential modulators of redox thiol metabolism of Leishmania parasite. Parasitol Res 110(1):341–8. https://doi.org/10.1007/s00436-011-2498-x
Siddiqui MFMF, Waghmare SP, Mode SG, Rekhate DH, Kolte SW, Hajare SW, Ali SS (2017) Phyto-pharmacological study and therapeutic efficacy of Calotropis Procera (flower) against theileriosis in cattle. J Anim Res 7(4):757–762. https://doi.org/10.5958/2277-940X.2017.00116.4
Silva ARST, Scher R, Santos FC, Ferreira SR, Cavalcanti SCH, Correa CB, Bueno LL, Alves RJ, Souza DP, Fujiwara RT, Dolabella SS (2017) Leishmanicidal activity and structure-activity relationships of essential oil constituents. Molecule 22(5):815. https://doi.org/10.3390/molecules22050815
Subramaniya BR, Srinivasan G, Sadullah SS, Davis N, Subhadra LB, Halagowder D, Sivasitambaram ND (2011) Apoptosis inducing effect of plumbagin on colonic cancer cells depends on expression of COX-2. PLoS One 6(4):e18695. https://doi.org/10.1371/journal.pone.0018695
Suchaichit N, Suchaichit NP, Kanokmedhakul K, Boottanun P, Sermswan RW, Moosophon P, Kanokmedhakul S (2021) A new cytotoxic plumbagin derivative from roots of Diospyros undulata. Nat Prod Res 35(10):1605–1612. https://doi.org/10.1080/14786419.2019.1630120
Sumsakul W, Plengsuriyakarn T, Chaijaroenkul W, Viyanant V, Karbwang J, Na-Bangchang K (2014) Antimalarial activity of plumbagin in vitro and in animal models. BMC Complement Altern Med 12(14):15. https://doi.org/10.1186/1472-6882-14-15
Tasdemir D, Kaiser M, Demirci F, Baser KHC (2006) Essential oil of Turkish Origanum onites L. and its main components, carvacrol and thymol show potent antiprotozoal activity without cytotoxicity. Planta Med 72(11):1006. https://doi.org/10.1055/s-2006-949877
Uc-Cachón AH, Borges-Argáez R, Said-Fernández S, Vargas-Villarreal J, González-Salazar F, Méndez-González M, Cáceres-Farfán M, Molina-Salinas GM (2014) Naphthoquinones isolated from Diospyros anisandra exhibit potent activity against pan-resistant first-line drugs Mycobacterium tuberculosis strains. Pulm Pharmacol Ther 27(1):114–20. https://doi.org/10.1016/j.pupt.2013.08.001
Viseras J, Hueli LE, Adroher FJ, García-Fernández P (1999) Studies on the transmission of Theileria annulata to cattle by the tick Hyalomma lusitanicum. ZentralblVeterinarmed B 46(8):505–509. https://doi.org/10.1111/j.1439-0450.1999.tb01242.x
Wilkie GM, Brown CGD, Kirvar E, Thomas M, Williamson SM, Bell-Sakyi LJ, Sparagano O (1998) Chemoprophylaxis of Theileria annulata and Theileria parva infections of calves with buparvaquone. Vet Parasitol 78(1):1–12. https://doi.org/10.1016/s0304-4017(98)00126-5
Youssefi MR, Moghaddas E, Tabari MA, Moghadamnia AA, Hosseini SM, Farash BRH, Ebrahimi MA, Mousavi NN, Fata A, Maggi F, Petrelli R, Dall’Acqua S, Benelli G, Sut S (2019) In vitro and in vivo effectiveness of carvacrol, thymol and linalool against Leishmania infantum. Molecule 24(11):2072. https://doi.org/10.3390/molecules24112072
Funding
This work was supported by Department of Biotechnology (DBT), Ministry of Science and Technology, Govt. of India, for their financial assistance (BT/PR10526/ADV/90/161/2014).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, writing first draft of manuscript were performed by ET and AP. Data analysis, manuscript review and editing were performed by AP, PP, SMR. Material preparation, manuscript review and editing SSD, NKS and GJ: Methodology. Funding acquisition and Supervision were done by AP and RM. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
No experimental animals were used in this study and no invasive and painful methods were carried out on animals in this study.
Consent to participate
Not Applicable.
Consent for publication
All the authors have approved this manuscript and given consent to publish this data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Titus, E.E., Palavesam, A., Rajaram, S.M. et al. In vitro efficacy of plumbagin and thymol against Theileria annulata. J Parasit Dis 47, 152–160 (2023). https://doi.org/10.1007/s12639-022-01550-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-022-01550-x