Abstract
Purpose
Endovascular treatment for stroke patients usually requires anesthesia care, with no current consensus on the best anesthetic management strategy. Several randomized controlled trials and meta-analyses have attempted to address this. In 2022, additional evidence from three new trials was published: the GASS trial, the CANVAS II trial, and preliminary results from the AMETIS trial, prompting the execution of this updated systematic review and meta-analysis. The primary objective of this study was to evaluate the effects of general anesthesia and conscious sedation on functional outcomes measured with the modified Rankin scale (mRS) at three months.
Methods
We performed a systematic review and meta-analysis of randomized controlled trials investigating conscious sedation and general anesthesia in endovascular treatment. The following databases were examined: PubMed, Scopus, Embase, and the Cochrane Database of Randomized Controlled Trials and Systematic Reviews. The Risk of Bias 2 tool was used to assess bias. In addition, trial sequence analysis was performed on the primary outcome to estimate if the cumulative effect is significant enough to be unaffected by further studies.
Results
Nine randomized controlled trials were identified, including 1,342 patients undergoing endovascular treatment for stroke. No significant differences were detected between general anesthesia and conscious sedation with regards to mRS, functional independence (mRS, 0–2), procedure duration, onset to reperfusion, mortality, hospital length of stay, and intensive care unit length of stay. Patients treated under general anesthesia may have more frequent successful reperfusion, though the time from groin to reperfusion was slightly longer. Trial sequential analysis showed that additional trials are unlikely to show marked differences in mean mRS at three months.
Conclusions
In this updated systematic review and meta-analysis, the choice of anesthetic strategy for endovascular treatment of stroke patients did not significantly impact functional outcome as measured with the mRS at three months. Patients managed with general anesthesia may have more frequent successful reperfusion.
Trial registration
PROSPERO (CRD42022319368); registered 19 April 2022.
Résumé
Objectif
Le traitement endovasculaire pour les patient·es victimes d’un accident vasculaire cérébral (AVC) nécessite généralement des soins d’anesthésie, mais il n’existe actuellement aucun consensus sur la meilleure stratégie de prise en charge anesthésique. Plusieurs études randomisées contrôlées et méta-analyses ont tenté d’aborder cette question. En 2022, des données probantes supplémentaires provenant de trois nouvelles études ont été publiées : l’étude GASS, l’étude CANVAS II et les résultats préliminaires de l’étude AMETIS, ce qui a motivé la réalisation de cette revue systématique et méta-analyse mises à jour. L’objectif principal de cette étude était d’évaluer les effets de l’anesthésie générale et de la sédation consciente sur les devenirs fonctionnels mesurés avec l’échelle de Rankin modifiée (mRS) à trois mois.
Méthode
Nous avons réalisé une revue systématique avec méta-analyse d’études randomisées contrôlées portant sur la sédation consciente et l’anesthésie générale dans le traitement endovasculaire. Les bases de données suivantes ont été examinées : PubMed, Scopus, Embase et la base de données Cochrane des études randomisées contrôlées et des revues systématiques. L’outil Risque de biais 2 a été utilisé pour évaluer le biais. De plus, une analyse séquentielle des études a été effectuée sur le critère d’évaluation principal afin d’estimer si l’effet cumulatif était suffisamment significatif pour ne pas être affecté par d’autres études.
Résultats
Neuf études randomisées contrôlées ont été identifiées, incluant 1342 patient·es bénéficiant d’un traitement endovasculaire pour un AVC. Aucune différence significative n’a été détectée entre l’anesthésie générale et la sédation consciente en ce qui concerne la mRS, l’indépendance fonctionnelle (mRS, 0-2), la durée de l’intervention, le moment d’apparition de la reperfusion, la mortalité, la durée de séjour à l’hôpital et la durée de séjour en unité de soins intensifs. Les patient·es traité·es sous anesthésie générale pourraient avoir une reperfusion réussie plus fréquente, bien que le temps entre l’aine et la reperfusion était légèrement plus long. L’analyse séquentielle des études a montré qu’il est peu probable que d’autres études montrent des différences marquées dans la mRS moyenne à trois mois.
Conclusion
Dans cette revue systématique et méta-analyse mises à jour, le choix de la stratégie anesthésique pour le traitement endovasculaire des personnes victimes d’un AVC n’a pas eu d’impact significatif sur les devenirs fonctionnels mesurés avec la mRS à trois mois. La réussite de la reperfusion pourrait être plus fréquente chez les patient·es pris·es en charge par anesthésie générale.
Enregistrement de l’étude
PROSPERO (CRD42022319368); enregistrée le 19 avril 2022.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Cerebral ischemic stroke remains one of the leading causes of death and disability.1 For eligible patients, the available therapies include intravenous thrombolysis and endovascular treatment (EVT) with mechanical thrombectomy. Endovascular treatment continues to expand its intervention possibilities with technical improvements and a prolonged time frame of therapeutic opportunity.2 These procedures usually require anesthesia care and are generally performed under conscious sedation (CS) or general anesthesia (GA).3 Some patients require GA because of neurologic status or other comorbidities, independently of the neurovascular procedure, while most patients can potentially undergo treatment with either CS or GA. Multiple studies have investigated the difference in outcomes between these anesthesia options with mixed results. Previous meta-analyses of randomized controlled trials (RCTs) on this subject have generally concluded that GA is superior to CS in terms of successful recanalization and functional independence at three months.4,5,6,7,8,9 In 2022, additional evidence from three new trials was published: the GASS trial,10 the AMETIS trial,11 and the CANVAS II study,12 prompting the execution of this updated meta-analysis.
The primary objective of this updated systematic review and meta-analysis was to evaluate the effect of different anesthesia approaches on functional outcomes measured with the modified Rankin Scale (mRS) at three months.
Methods
This manuscript was prepared following the Preferred Reporting Items for Systematic reviews and Meta-Analysis (PRISMA) Statement Guidelines.13 The PRISMA checklist is available in Electronic Supplementary Material (ESM) eAppendix 1.
The protocol of this systematic review and meta-analysis was registered in PROSPERO (CRD42022319368; 19 April 2022).
Eligibility criteria
Published studies that met the following Population, Intervention, Comparison, Outcomes and Study (PICOS) criteria were included: adult (aged 18 yr or older) patients with ischemic stroke undergoing mechanical thrombectomy (P); GA (I); CS (C); mRS at three months (primary outcome), functional independence at three months (defined as an mRS of 0–2), mortality at three months, successful recanalization (as defined by the Thrombolysis in Cerebral Infarction [TICI] score of 2b or 3), onset to reperfusion (min), door to reperfusion (min), groin to reperfusion (min), duration of intervention (min), length of stay (LOS) in the intensive care unit (ICU) (hr), hospital LOS (days) (O); and randomized controlled trial (S).
Search strategy
We performed a systematic search of the medical literature to identify, screen, and include articles. The search strategy was elaborated and performed by F. G. and P. D. using the following databases (last updated on 13 October 2022): PubMed, Scopus, the Cochrane Database of Randomized Controlled Trials (CENTRAL), the Cochrane Database of Systematic Reviews, and Embase. The search included all available articles from database inception to the search date. The references of included studies, relevant reviews, and meta-analyses on the subject were also examined for articles meeting the inclusion criteria for this updated meta-analysis. We did not apply any restriction on the language or year of publication. For specific information regarding our search strategy, see ESM eAppendix 2.
Study selection
Two researchers (D. F. and I. L.) independently screened titles and abstracts of the identified articles to select relevant manuscripts. Each citation was reviewed in full-text form if considered potentially relevant.
Data extraction and data retrieval
After identifying those studies meeting the inclusion criteria, two authors (P. D. and T. P.) manually reviewed and assessed each of the included studies. Any disagreement on both study selection and data extraction was resolved by discussion, and, if no agreement was reached, consultation with a third author (F. G.) was used to resolve disputes.
The following information was collected for each paper: first author, year of publication, total number of patients per group (GA or CS), mRS at three months, mortality, TICI score, duration of intervention, onset to reperfusion in minutes, groin to reperfusion in minutes, ICU LOS in hr and hospital LOS in days.
If data were missing and contact details were available, a request for further information was sent by e-mail to the corresponding author of the study. If no reply was received after the initial request, a second message was sent seven days later, followed by a third and final request one week after the second one.
Quality assessment and certainty of evidence assessment
Two researchers (A. D. C. and A. B.) independently evaluated the quality of included RCTs using the Risk of Bias (RoB) 2 tool.14 Disagreements were resolved by discussion with a third researcher (N. S.). If an agreement was not reached, the final decision was taken by the third researcher (N. S.).
An overall risk of bias was expressed on a three-grade scale (“low risk of bias,” “high risk of bias,” or “some concerns”). Detailed ROB 2 assessment is available in ESM eAppendix 3.
We used the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach to assess the certainty of evidence related to each of the outcomes.15
Statistical methods
Published data with mean and standard deviation were entered as continuous variables, while continuous variables expressed as median and interquartile range or first-third quartile, mean and standard deviation were calculated with Hozo’s method.16 Data with a binary outcome were entered as a dichotomous variable.
Meta-analyses were performed with a random-effects model to account for between-study heterogeneity,17 using Hartung–Knapp adjustment to calculate the confidence interval (CI) around the pooled effect.18,19 The treatment effects for continuous outcomes were analyzed with the inverse variance method and expressed as mean differences (MDs) with their 95% confidence intervals (CIs). The treatment effects for dichotomous outcomes were analyzed with the Mantel–Haenszel method and expressed as odds ratios (ORs) with their 95% CIs. Confidence intervals for ORs within the no effect area and including values < 0.70 or > 1.43 were considered imprecise and penalized accordingly in the GRADE assessment.
The statistical analysis was performed using the “meta” package20 for R software version 4.1.2 (R Foundation for Statistical Computing, Vienna, Austria). Trial sequence analysis was performed on the main outcome with the Trial Sequential Analysis software (Copenhagen Trial Unit, Centre for Clinical Intervention Research, Copenhagen, Denmark) with a power of 90% and a type I error of 5% and arbitrarily electing to use a difference of 0.5 points in the mRS.
Inconsistency, heterogeneity, and publication bias analysis
We assessed heterogeneity using I2, considering I2 values of < 25% as low, 25% to 50% as moderate, and > 50% as high. The restricted maximum likelihood estimator and the Paule–Mandel estimator were used to calculate the heterogeneity variance (τ2) for continuous outcomes and for binary outcomes, respectively.21 Publication bias was evaluated both by a visual inspection of funnel plots (ESM eAppendix 4) and with the Egger test for any outcome with ten or more studies (P value < 0.05 as index of a possible publication bias).
Deviations from protocol
Performing trial sequential analysis (TSA) on a very small difference in mRS (0.09) was considered impractical and of limited clinical significance. We therefore chose to perform the analysis selecting a clinically relevant value, rather than the lower CI.22 No clear consensus exists regarding mRS improvement thresholds for stroke patients, though in literature a change in 1 point on the mRS scale has been described with certainty as clinically relevant.23 Specific treatment options for stroke patients offer widely ranging benefits and risk-benefit ratios. Hemicraniectomy, used in patients developing intracranial hypertension, has been reported to offer a median benefit in mRS of 2 points and a mean benefit of 1.1 points, while the use of alteplase has been reported to offer a median benefit in mRS of 1 point and a mean benefit of 0.53 points.24 After discussion among our research group, we elected to perform TSA using a pragmatic threshold of 0.5 points in MD on the mRS scale, reasoning it would certainly be of clinical significance.
We performed sensitivity analyses on the main outcome, excluding studies with an evaluation other than “low risk of bias.” We also analyzed different definitions of the selected dichotomous outcome for mRS (functional independence or mRS of 0–2). Specifically, we examined excellent functional outcome (mRS of 0–1) and positive functional outcome (mRS of 0–3).
There were insufficient data to provide new insight on the time taken from door to reperfusion. Additionally, insufficient information was obtained regarding patients whose treatment was converted from CS to GA; therefore, the planned subgroup analysis was not performed. Instead, a subgroup analysis excluding patients with posterior circulation stroke was performed to account for the different clinical and outcome characteristics of this type of stroke.12
Results
Study selection and data retrieval
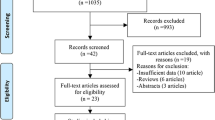
The initial search found 672 articles and the assessment for duplicates excluded 300. We screened a total of 372 abstracts, leading to the inclusion of eight full-text manuscripts10,12,25,26,27,28,29,30 and one conference abstract11 comparing EVT under CS or GA. Search results are shown in the PRISMA diagram (Fig. 1). Characteristics of the included studies are available in Table 1, while excluded articles are reported in ESM eAppendix 5.
Study characteristics
The nine included studies randomized a total of 1,342 patients, allocating 670 to GA and 672 to CS. Seven of the included studies randomized 1,116 patients with large vessel occlusion (LVO) of the anterior circulation, whereas two studies randomized 226 patients treated for stroke of the posterior circulation. Forest plots for primary and secondary outcomes are available in Fig. 2 and ESM eAppendix 6.
According to the risk of bias evaluation, six studies10,12, 25,26,27,28 were judged to be at low risk of bias, while some concerns arose for three studies11,29,30 (Fig. 3). Details about the risk of bias judgements are available as ESM eAppendix 3. The detailed GRADE assessment for individual outcomes is available in Table 2.
Outcomes
Modified Rankin scale score at three months
Functional outcome (mRS) at three months was evaluated in eight studies.10,12, 25,26,27,28,29,30 No significant difference was detected with regards to the mRS (MD, 0.12; 95% CI, -0.09 to 0.34) (Fig. 2) and heterogeneity was low (I2 = 0%). The subgroup analysis evaluating patients with anterior circulation stroke10,25,26,27,28,29 also did not show a significant difference between the two anesthetic management strategies (MD, 0.10; 95% CI, -0.11 to 0.31) (ESM eAppendix 6).
The risk of bias was reported as low in six of the included studies in this analysis, while two were evaluated as having some concerns of bias (Fig. 3). The sensitivity analysis excluding the trials with some concerns29,30 showed similar results (MD, 0.13; 95% CI, -0.15 to 0.42) (ESM eAppendix 6). The overall certainty of evidence assessed with GRADE was rated as moderate.
The eight included trials were analyzed with TSA to estimate if the effect was large enough to be unaffected by further studies: the cumulative z-score reached the required sample size at the seventh trial, showing no effect overall among the examined treatments (Fig. 4).
Trial sequential analysis on main outcome (power of 90% and a type I error of 5%). The z-curve, representing the cumulative z-score of the included studies, remains below the conventional test boundaries (−1.96 and +1.96), showing the null hypothesis of no difference between intervention groups is accepted. The boundary for futility is reached at the fifth trial while the required information size, represented by the dotted vertical line, is reached with the seventh trial.
Functional independence at three months
Functional independence (defined as an mRS of 0–2) was analyzed in eight studies.10,11,12, 25,26,27,28, 30 There was no difference between GA and CS with regards to functional independence at three months (OR, 1.23; 95% CI, 0.88 to 1.72) (Fig. 2). Heterogeneity for this outcome was moderate (I2 = 28%).
The sensitivity analysis performed on studies reporting excellent functional outcomes (mRS, 0–1) (OR, 1.12; 95% CI, 0.82 to 1.55) and positive functional outcomes (mRS, 0–3) (OR, 1.21; 95% CI, 0.90 to 1.62) yielded similar results. The subgroup analysis excluding patients with posterior circulation stroke12,30 also did not show marked differences in terms of functional independence (OR, 1.27; 95% CI, 0.81 to 2.01) (ESM eAppendix 6).
The risk of bias was reported as low in six of the included studies in this analysis while two were evaluated as having some concerns (Fig. 3). The sensitivity analysis excluding the trials with some concerns showed similar results (OR, 1.35; 95% CI, 0.89 to 2.05) (ESM eAppendix 6). The overall certainty of evidence assessed with GRADE was rated as low (Table 2).
Mortality
Mortality at three months was analyzed in seven studies.10,12,25,26,27,28,29 No significant difference in mortality was found between patients managed with CS and GA (OR, 1.10; 95% CI, 0.74 to 1.65) (Fig. 2) and heterogeneity was low (I2 = 0%).
Risk of bias was reported as low in six of the included studies in this analysis, while one was evaluated as having some concerns of bias (Fig. 3). The overall certainty of evidence assessed with GRADE was rated as very low (Table 2).
Successful reperfusion
Successful reperfusion, defined as a TICI score of 2b or 3, was analyzed in eight studies.10,12, 25,26,27,28,29,30 General anesthesia showed a greater proportion of patients with successful reperfusion than CS did (OR, 1.86; 95% CI, 1.12 to 3.10) with a number needed to treat of 11 (Fig. 2). Heterogeneity was moderate (I2 = 26%). A sensitivity analysis using 99.29% CIs showed a small chance of uncertainty in this result (OR, 1.86; 95% CI, 0.83 to 4.19) (ESM eAppendix 6).
The risk of bias was reported as low in six of the included studies in this analysis, while two were evaluated as having some concerns of bias (Fig. 3). The overall certainty of evidence assessed with GRADE was rated as moderate (Table 2).
Duration of intervention
Procedure duration (min) was analyzed in four studies.12, 26,29, 30 There was no significant difference between CS and GA (MD, 2.3; 95% CI, -23.1 to 27.6) (ESM eAppendix 6). Heterogeneity for this outcome was high (I2 = 71%).
The risk of bias was low in two of the included studies, while the remaining two were evaluated as having some concerns of bias (Fig. 3). The overall certainty of evidence assessed with GRADE was rated as low (Table 2).
Onset to reperfusion
Time from stroke onset to reperfusion (min) was analyzed in five studies.10,12,25, 27,28 There was no significant difference between CS and GA (MD, -7.7; 95% CI, -24.4 to 9.0) (Fig. 2) and heterogeneity was low (I2 = 0%).
The risk of bias was low for all five studies (Fig. 3) and the overall certainty of evidence assessed with GRADE was rated as high (Table 2).
Groin to reperfusion
Time from groin puncture to reperfusion (min) was analyzed in five studies.12,25,27,28,29 Time from groin puncture to reperfusion was shorter in the CS group than in the GA group (MD, -6.7; 95% CI, -11.3 to -2.1) (Fig. 2). Heterogeneity was low (I2 = 0%).
The risk of bias was reported as low in four of the included studies in this analysis, while one was evaluated as having some concerns of bias (Fig. 3). The overall certainty of evidence assessed with GRADE was rated as high (Table 2).
Intensive care unit length of stay
Intensive care unit LOS (hr) was measured in four studies.10,12,26,28 There was no significant difference between CS and GA (MD, -14.1; 95% CI, -51.1 to 22.9) (ESM eAppendix 6) and heterogeneity for this outcome was moderate (I2 = 45%).
The risk of bias was reported as low in all four studies (Fig. 3) and the overall certainty of evidence assessed with GRADE was rated as low (Table 2).
Hospital length of stay
Hospital LOS (days) was measured in three studies.12,26,30 There was no significant difference between CS and GA (MD, -0.8; 95% CI, -2.4 to 0.8) and heterogeneity was low (I2 = 0%) (ESM eAppendix 6).
The risk of bias was low in two of the included studies, while one was evaluated as having some concerns of bias (Fig. 3). The overall certainty of evidence assessed with GRADE was rated as very low (Table 2).
Discussion
The optimal anesthetic technique during EVT for cerebral ischemic stroke remains a controversial and debated topic, with current guidelines advising a decision based on the patient’s characteristics without providing specific recommendations.31 The main result of this updated systematic review and meta-analysis is that the mRS three months after EVT is not influenced by the chosen anesthetic strategy. Nevertheless, successful reperfusion may have occurred more often in patients managed with GA. The outcome was also unchanged when patients with ischemic stroke of the posterior circulation were excluded. Our analysis also shows no significant difference between GA and CS when considering the time from onset to reperfusion and procedure duration, while the time from groin to reperfusion was slightly longer in patients undergoing EVT with GA. Furthermore, no difference was detected in terms of hospital and ICU LOS. Finally, TSA was performed on the primary outcome, showing that it is improbable that further trials will show a mean improvement in mRS of greater than 0.5 points for either anesthetic strategy at three months.
Previous meta-analyses of RCTs have reached similar conclusions regarding rates of successful reperfusion while also reporting that patients managed under GA had improved functional outcome scores.4,5,6,7 More recent meta-analyses, including the GASS trial, have shown alternating results, with some showing a benefit for GA8,9 and one showing no benefit in terms of long-term outcome.32 Compared with the latest meta-analyses, we have updated the current evidence by including results from the latest trials.11,12 We have also elected to differentiate between anterior and posterior circulation strokes as the involvement of the brainstem has been associated with diminished consciousness and poorer outcomes compared with anterior circulation strokes.12,30 Additionally, in terms of treatment options, the effectiveness of EVT in posterior circulation strokes is currently being established in the literature and may warrant further investigation.12 Furthermore, this meta-analysis employed TSA to evaluate if the obtained results can be considered sufficient to form clinically relevant conclusions.
While meta-analyses of RCTs have often shown that patients managed with GA had better functional outcomes, meta-analyses using observational trials and large naturalistic cohorts33,34,35,36,37,38,39,40,41,42,43 have not found marked differences in outcome between the two anesthetic strategies. In fact, several large observational trials have shown a benefit for cohorts managed with local anesthesia or CS.44,45,46 The more favourable outcome reported in patients managed with CS or local anesthesia in observational trials could be related to the inherent “selection bias” present in nonrandomized patients, which may occur despite there being no specific criteria to prefer one method over the other in most patients.
The planned subgroup analysis on outcomes in patients requiring emergent conversion to GA was not performed. This event was relatively rare in the included trials and outcomes for these patients were not explicitly reported. The available evidence on the subject comes from a post hoc analysis of the data from the AnStroke, Goliath, and Siesta trials showing that patients emergently converted to GA after initiating the procedure under CS had significantly worse outcomes.47 No clear criteria have been established to predict the risk of conversion to GA. Procedure duration, higher Sequential Organ Failure Assessment scores, number of pharmacological agents used, and tandem strokes have been reported as potential risk factors for emergent conversion to GA in a small retrospective trial,48 while another study found procedure duration was longer in patients who were emergently converted to GA.49
With the mRS at three months was not markedly different between anesthetic strategies, it is debatable if the apparent increased rate of successful reperfusion in patients managed with GA represents a clinically relevant advantage. Successful reperfusion has been associated with improved functional outcomes in previous meta-analyses.50 It is therefore reasonable that obtaining a greater rate of successful reperfusion could be beneficial for patients undergoing EVT. On the other hand, given the apparent lack of benefit in terms of clinical outcome in this study, it is also plausible that successful reperfusion obtained under GA may have been occasionally futile.51 Furthermore, while procedure duration and time from onset to reperfusion were unaffected by the anesthetic strategy, groin puncture to reperfusion was slightly longer in patients managed with GA. With recanalization apparently being more successful under GA, this could represent a potential source of bias. We can only speculate that patients managed under CS are either reperfused more quickly or that the procedure is suspended earlier than when the patients are managed with GA. It has been stated that procedural conditions are improved under GA,5 and this may play a role in the decision to continue or suspend EVT. Accordingly, as some authors have previously suggested, GA could be favoured when there are reasons to expect a prolonged procedure or when facing patient-related factors that may complicate procedural management with CS (i.e., severe agitation, predicted difficult airway, poor baseline oxygen saturation, dysphagia, and inability to follow commands).52
Future studies should establish if the more frequent successful reperfusion with GA also carries a benefit in terms of functional outcomes in specific subgroups of patients. Additionally, future trials should strive to confirm emergent conversion to GA as a risk factor for the worse functional outcome, as this knowledge could potentially influence the decision on the initial anesthetic strategy.
This study has some limitations. First, we could not analyze all prespecified outcomes and subgroups because of the lack of new data compared with previous studies. Second, the GASS trial10 reported outcomes between two and six months rather than the prespecified three months. Third, the decision to perform TSA using a 0.5 point on the mRS scale was arbitrary; we recognize that other thresholds could have been chosen. Fourth, GA and CS protocols were not uniform among different studies. Fifth, the wide CIs for some outcomes may limit the strength of our results and the conclusions which can be drawn.
Conclusion
Anesthetic management did not significantly impact functional outcomes at three months in patients with LVO undergoing EVT. Patients managed with GA may have improved chances of successful reperfusion. Trial sequential analysis showed that further trials are unlikely to show a MD in mRS of more than 0.5 points for either anesthetic strategy.
References
World Health Organization. Global health estimates: life expectancy and leading causes of death and disability, 2022. Available from URL: https://www.who.int/data/gho/data/themes/mortality-and-global-health-estimates (accessed December 2022).
Dornbos D 3rd, Arthur AS. Current state of the art in endovascular stroke treatment. Neurol Clin 2022; 40: 309–19. https://doi.org/10.1016/j.ncl.2021.11.008
American Society of Anesthesiologists’. Continuum of depth of sedation: definition of general anesthesia and levels of sedation/analgesia, 2019. Available from URL: https://www.asahq.org/standards-and-guidelines/continuum-of-depth-of-sedation-definition-of-general-anesthesia-and-levels-of-sedationanalgesia (accessed December 2022).
Bai X, Zhang X, Wang T, et al. General anesthesia versus conscious sedation for endovascular therapy in acute ischemic stroke: a systematic review and meta-analysis. J Clin Neurosci 2021; 86: 10–7. https://doi.org/10.1016/j.jocn.2021.01.012
Campbell D, Diprose WK, Deng C, Barber PA. General anesthesia versus conscious sedation in endovascular thrombectomy for stroke: a meta-analysis of 4 randomized controlled trials. J Neurosurg Anesthesiol 2021; 33: 21–7. https://doi.org/10.1097/ana.0000000000000646
Schönenberger S, Hendén PL, Simonsen CZ, et al. Association of general anesthesia vs procedural sedation with functional outcome among patients with acute ischemic stroke undergoing thrombectomy: a systematic review and meta-analysis. JAMA 2019; 322: 1283–93. https://doi.org/10.1001/jama.2019.11455
Zhang Y, Jia L, Fang F, Ma L, Cai B, Faramand A. General anesthesia versus conscious sedation for intracranial mechanical thrombectomy: a systematic review and meta-analysis of randomized clinical trials. J Am Heart Assoc 2019; 8: e011754. https://doi.org/10.1161/jaha.118.011754
Lee CW, Chang YP, Huang YT, et al. General anesthesia but not conscious sedation improves functional outcome in patients receiving endovascular thrombectomy for acute ischemic stroke: a meta-analysis of randomized clinical trials and trial sequence analysis. Front Neurol 2022; 13: 1017098. https://doi.org/10.3389/fneur.2022.1017098
Campbell D, Butler E, Barber PA. End the confusion: general anaesthesia improves patient outcomes in endovascular thrombectomy. Br J Anaesth 2022; 129: 461–4. https://doi.org/10.1016/j.bja.2022.06.018
Maurice A, Eugène F, Ronzière T, et al. General anesthesia versus sedation, both with hemodynamic control, during intraarterial treatment for stroke: the GASS randomized trial. Anesthesiology 2022; 136: 567–76. https://doi.org/10.1097/aln.0000000000004142
Chabanne R, Geeraerts T, Begard M, et al. Procedural sedation versus general anesthesia in anterior circulation large vessel occlusion thrombectomy: the Anesthesia Management in Endovascular Therapy for Ischemic Stroke (AMETIS) randomized controlled trial (conference abstract). Eur Stroke J 2022; 7: 3–545. https://doi.org/10.1177/23969873221087559
Liang F, Wu Y, Wang X, et al. General anesthesia vs conscious sedation for endovascular treatment in patients with posterior circulation acute ischemic stroke: an exploratory randomized clinical trial. JAMA Neurol 2022; e223018. https://doi.org/10.1001/jamaneurol.2022.3018
Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 2009; 339: b2700. https://doi.org/10.1136/bmj.b2700
Sterne JA, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 2019; 366: l4898. https://doi.org/10.1136/bmj.l4898
Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008; 336: 924–6. https://doi.org/10.1136/bmj.39489.470347.ad
Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol 2005; 5: 13. https://doi.org/10.1186/1471-2288-5-13
Cumpston M, Li T, Page MJ, et al. Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst Rev 2019; 10: ED000142. https://doi.org/10.1002/14651858.ed000142
Röver C, Knapp G, Friede T. Hartung-Knapp-Sidik-Jonkman approach and its modification for random-effects meta-analysis with few studies. BMC Med Res Methodol 2015; 15: 99. https://doi.org/10.1186/s12874-015-0091-1
IntHout J, Ioannidis JP, Borm GF. The Hartung-Knapp-Sidik-Jonkman method for random effects meta-analysis is straightforward and considerably outperforms the standard DerSimonian-Laird method. BMC Med Res Methodol 2014; 14: 25. https://doi.org/10.1186/1471-2288-14-25
Schwarzer G, Carpenter JR, Rucker G. Meta-Analysis with R, 1st ed. Cham: Springer International Publishing; 2015.
Veroniki AA, Jackson D, Viechtbauer W, et al. Methods to estimate the between‐study variance and its uncertainty in meta‐analysis. Res Synth Methods 2016; 7: 55–79. https://doi.org/10.1002/jrsm.1164
De Cassai A, Pasin L, Boscolo A, Salvago M, Navalesi P. Trial sequential analysis: plain and simple. Korean J Anesthesiol 2021; 74: 363–5. https://doi.org/10.4097/kja.20637
Broderick JP, Adeoye O, Elm J. Evolution of the modified Rankin scale and its use in future stroke trials. Stroke 2017; 48: 2007–12. https://doi.org/10.1161/strokeaha.117.017866
Bath PM, Lees KR, Schellinger PD, et al. Statistical analysis of the primary outcome in acute stroke trials. Stroke 2012; 43: 1171–8. https://doi.org/10.1161/strokeaha.111.641456
Löwhagen Hendén P, Rentzos A, Karlsson JE, et al. General anesthesia versus conscious sedation for endovascular treatment of acute ischemic stroke: the AnStroke trial (anesthesia during stroke). Stroke 2017; 48: 1601–7. https://doi.org/10.1161/strokeaha.117.016554
Schönenberger S, Uhlmann L, Hacke W, et al. Effect of conscious sedation vs general anesthesia on early neurological improvement among patients with ischemic stroke undergoing endovascular thrombectomy: a randomized clinical trial. JAMA 2016; 316: 1986–96. https://doi.org/10.1001/jama.2016.16623
Simonsen CZ, Yoo AJ, Sørensen LH, et al. Effect of general anesthesia and conscious sedation during endovascular therapy on infarct growth and clinical outcomes in acute ischemic stroke: a randomized clinical trial. JAMA Neurol 2018; 75: 470–7. https://doi.org/10.1001/jamaneurol.2017.4474
Sun J, Liang F, Wu Y, et al. Choice of Anesthesia for EndoVAScular treatment of acute ischemic stroke (CANVAS): results of the CANVAS pilot randomized controlled trial. J Neurosurg Anesthesiol 2020; 32: 41–7. https://doi.org/10.1097/ana.0000000000000567
Ren C, Xu G, Liu Y, Liu G, Wang J, Gao J. Effect of conscious sedation vs. general anesthesia on outcomes in patients undergoing mechanical thrombectomy for acute ischemic stroke: a prospective randomized clinical trial. Front Neurol 2020; 11: 170. https://doi.org/10.3389/fneur.2020.00170
Hu G, Shi Z, Li B, Shao W, Xu B. General anesthesia versus monitored anesthesia care during endovascular therapy for vertebrobasilar stroke. Am J Transl Res 2021; 13: 1558–67.
Powers WJ, Rabinstein AA, Ackerson T, et al. Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2019; 50: e344–418. https://doi.org/10.1161/str.0000000000000211
Tosello R, Riera R, Tosello G, et al. Type of anaesthesia for acute ischaemic stroke endovascular treatment. Cochrane Database Syst Rev 2022; 7: CD013690. https://doi.org/10.1002/14651858.cd013690.pub2
Brinjikji W, Murad MH, Rabinstein AA, Cloft HJ, Lanzino G, Kallmes DF. Conscious sedation versus general anesthesia during endovascular acute ischemic stroke treatment: a systematic review and meta-analysis. AJNR Am J Neuroradiol 2015; 36: 525–9. https://doi.org/10.3174/ajnr.a4159
Brinjikji W, Pasternak J, Murad MH, et al. Anesthesia-related outcomes for endovascular stroke revascularization: a systematic review and meta-analysis. Stroke 2017; 48: 2784–91. https://doi.org/10.1161/strokeaha.117.017786
Butt W, Dhillon PS, Podlasek A, et al. Local anesthesia as a distinct comparator versus conscious sedation and general anesthesia in endovascular stroke treatment: a systematic review and meta-analysis. J Neurointerv Surg 2022; 14: 221–6. https://doi.org/10.1136/neurintsurg-2021-017360
Ilyas A, Chen C-J, Ding D, et al. Endovascular mechanical thrombectomy for acute ischemic stroke under general anesthesia versus conscious sedation: a systematic review and meta-analysis. World Neurosurg 2018; 112: e355–67. https://doi.org/10.1016/j.wneu.2018.01.049
Jing R, Dai HJ, Lin F, Ge WY, Pan LH. Conscious sedation versus general anesthesia for patients with acute ischemic stroke undergoing endovascular therapy: a systematic review and meta-analysis. Biomed Res Int 2018; 2018: 1–9. https://doi.org/10.1155/2018/2318489
Wan TF, Zhang JR, Liu L. Effect of general anesthesia vs. conscious sedation on the outcomes of acute ischemic stroke patients after endovascular therapy: a meta-analysis of randomized clinical trials. Front Neurol 2019; 10: 1131. https://doi.org/10.3389/fneur.2019.01131
Gravel G, Boulouis G, Benhassen W, et al. Anaesthetic management during intracranial mechanical thrombectomy: systematic review and meta-analysis of current data. J Neurol Neurosurg Psychiatry 2019; 90: 68–74. https://doi.org/10.1136/jnnp-2018-318549
Kim C, Kim SE, Jeon JP. Influence of anesthesia type on outcomes after endovascular treatment in acute ischemic stroke: meta-analysis. Neurointervention 2019; 14: 17–26. https://doi.org/10.5469/neuroint.2019.00045
Li X, Hu Z, Li Q, et al. Anesthesia for endovascular treatment in anterior circulation stroke: a systematic review and meta-analysis. Brain Behav 2019; 9: e01178. https://doi.org/10.1002/brb3.1178
Shen H, Ma X, Wu Z, et al. Conscious sedation compared to general anesthesia for intracranial mechanical thrombectomy: a meta-analysis. Brain Behav 2021; 11: e02161. https://doi.org/10.1002/brb3.2161
Feil K, Herzberg M, Dorn F, et al. General anesthesia versus conscious sedation in mechanical thrombectomy. J Stroke 2021; 23: 103–12. https://doi.org/10.5853/jos.2020.02404
Cappellari M, Pracucci G, Forlivesi S, et al. General anesthesia versus conscious sedation and local anesthesia during thrombectomy for acute ischemic stroke. Stroke 2020; 51: 2036–44. https://doi.org/10.1161/strokeaha.120.028963
Dhillon PS, Butt W, Podlasek A, et al. Association between anesthesia modality and clinical outcomes following endovascular stroke treatment in the extended time window. J Neurointerv Surg 2022; https://doi.org/10.1136/neurintsurg-2022-018846
Han H, Wang Y, Wang H, et al. General anesthesia vs. local anesthesia during endovascular treatment for acute large vessel occlusion: a propensity score-matched analysis. Front Neurol 2022; 12: 801024. https://doi.org/10.3389/fneur.2021.801024
Simonsen CZ, Schönenberger S, Hendén PL, et al. Patients requiring conversion to general anesthesia during endovascular therapy have worse outcomes: a post hoc analysis of data from the SAGA collaboration. AJNR Am J Neuroradiol 2020; 41: 2298–302. https://doi.org/10.3174/ajnr.a6823
Geraldini F, De Cassai A, Napoli M, et al. Risk factors for general anesthesia conversion in anterior circulation stroke patients undergoing endovascular treatment. Cerebrovasc Dis 2021; 51: 481–7. https://doi.org/10.1159/000520929
Chen M, Kronsteiner D, Pfaff JA, et al. Emergency intubation during thrombectomy for acute ischemic stroke in patients under primary procedural sedation. Neurol Res Pract 2021; 3: 27. https://doi.org/10.1186/s42466-021-00125-0
Manning NW, Warne CD, Meyers PM. Reperfusion and clinical outcomes in acute ischemic stroke: systematic review and meta-analysis of the Stent-retriever-based, early window endovascular stroke trials. Front Neurol 2018; 9: 301. https://doi.org/10.3389/fneur.2018.00301
Lee SH, Kim BJ, Han MK, et al. Futile reperfusion and predicted therapeutic benefits after successful endovascular treatment according to initial stroke severity. BMC Neurol 2019; 19: 11. https://doi.org/10.1186/s12883-019-1237-2
Hindman BJ, Dexter F. Anesthetic management of emergency endovascular thrombectomy for acute ischemic stroke, part 2: integrating and applying observational reports and randomized clinical trials. Anesth Analg 2019; 128: 706–17.
Author contributions
Federico Geraldini conceived the idea, performed the initial search, contacted corresponding authors for missing data, performed the statistical analysis, and wrote and edited the manuscript. Paolo Diana conceived the idea, manually reviewed and assessed each of the included studies, and wrote and edited the manuscript. Davide Fregolent reviewed titles, abstracts, and full text manuscripts for potential inclusion and wrote and edited the manuscript. Alessandro De Cassai helped with the statistical evaluation, performed the ROB2 evaluation, and wrote and edited the manuscript. Annalisa Boscolo performed the ROB 2 evaluation and wrote and edited the manuscript. Tommaso Pettenuzzo performed the GRADE assessment, manually reviewed and assessed each of the included studies, and wrote and edited the manuscript. Nicolò Sella screened for duplicates, supervised the ROB 2 evaluation, resolving disputes, and helped with the revision and editing of the manuscript. Irene Lupelli reviewed titles, abstracts, and full text manuscripts for potential inclusion and critically revised and edited the manuscript. Paolo Navalesi supervised the project, helped with the interpretation and contextualization of the results, and critically assessed and edited the finalized manuscript. Marina Munari helped plan the research, assessed it for overall merit, supervised the project, helped with the interpretation and contextualization of the results, and critically assessed and edited the finalized manuscript.
Disclosures
All authors have no conflict of interest to report.
Funding statement
No funding to declare.
Editorial responsibility
This submission was handled by Dr. Stephan K. W. Schwarz, Editor-in-Chief, Canadian Journal of Anesthesia/Journal canadien d’anesthésie.
Author information
Authors and Affiliations
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Geraldini, F., Diana, P., Fregolent, D. et al. General anesthesia or conscious sedation for thrombectomy in stroke patients: an updated systematic review and meta-analysis. Can J Anesth/J Can Anesth 70, 1167–1181 (2023). https://doi.org/10.1007/s12630-023-02481-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-023-02481-8