Abstract
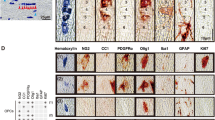
Oligodendrocytes (OLs) are glial cells that form myelin sheaths surrounding the axons in the central nervous system (CNS). Jimpy (jp) mutant mice are dysmyelinating disease models that show developmental abnormalities in myelinated OLs in the CNS. The causative gene in jp mice is the proteolipid protein (PLP) located on the X chromosome. Mutations in the jp allele result in exon 5 skipping and expression of abnormal PLP containing a C-terminal frame shift. Many lines of evidence suggest that abnormal PLP in OLs results in endoplasmic reticulum (ER) stress and cell death. To histologically detect glial responses in the jp mutant brain, we performed staining with lineage-specific markers. Using OL markers and OL progenitor cell marker staining, we identified reduced numbers of OL lineage cells in the jp mutant brain. Nuclear staining of the transcription factor Olig1 was observed in the Tabby-jp brain, whereas cytoplasmic Olig1 staining was observed in the wild-type brain at postnatal day 21, suggesting that active myelination was present in the mutant brain. Many microglial cells with activated morphology and intensive staining of CD11b microglia marker were observed in the internal capsule of the mutant brain, a region of white matter containing residual OLs. Activated astrocytes with high glial fibrillary acidic protein-immunoreactivity were also mainly observed in white matter. Finally, we performed in situ hybridization using C/EBP homologous protein (CHOP) antisense probes to detect ER stressed cells. CHOP mRNA was strongly expressed in residual OLs in the Tabby-jp mutant mice at postnatal stages. These data show that microglia and astrocytes exhibit dynamic glial activation in response to cell death of OLs during Tabby-jp pathogenesis, and that CHOP antisense probes may be a good marker for the detection of ER-stressed OLs in jp mutant mice.




Similar content being viewed by others
References
Armstrong RC, Mierzwa AJ, Sullivan GM et al (2016) Myelin and oligodendrocyte lineage cells in white matter pathology and plasticity after traumatic brain injury. Neuropharmacology 110:654–659
Arnett HA, Fancy SP, Alberta JA et al (2004) bHLH transcription factor Olig1 is required to repair demyelinated lesions in the CNS. Science 306:2111–2115
Bercury KK, Macklin WB (2015) Dynamics and mechanisms of CNS myelination. Dev Cell 32:447–458
Bhat RV, Axt KJ, Fosnaugh JS et al (1996) Expression of the APC tumor suppressor protein in oligodendroglia. Glia 17:169–174
Bin JM, Harris SN, Kennedy TE (2016) The oligodendrocyte-specific antibody “CC1” binds quaking 7. J Neurochem 139:181–186
Blecher SR, Kapalanga J, Lalonde D (1990) Induction of sweat glands by epidermal growth factor in murine X-linked anhidrotic ectodermal dysplasia. Nature 345:542–544
Brown GC, Neher JJ (2014) Microglial phagocytosis of live neurons. Nature Rev Neurosci 15:209–216
Dautigny A, Mattei MG, Morello D et al (1986) The structural gene coding for myelin-associated proteolipid protein is mutated in jimpy mice. Nature 321:867–869
Dimou L, Simon C, Kirchhoff F, Takebayashi H, Götz M (2008) Progeny of Olig2-expressing progenitors in the gray and white matter of the adult mouse cerebral cortex. J Neurosci 28:10434–10442
Duncan ID, Hammang JP, Goda S, Quarles RH (1989) Myelination in the jimpy mouse in the absence of proteolipid protein. Glia 2:148–154
Dupouey P, Lucas CV, Gomes D, Jacque C (1980) Immunohistochemical localization of the myelin basic protein and of the glial fibrillary acidic protein: comparative study in normal, quaking and jimpy mice. J Neurosci Res 5:387–398
Fannon AM, Mastronardi FG, Moscarello MA (1994) Isolation and identification of proteolipid proteins in jimpy mouse brain. Neurochem Res 19:1005–1012
Fukushima S, Nishikawa K, Furube E et al (2015) Oligodendrogenesis in the fornix of adult mouse brain; the effect of LPS-induced inflammatory stimulation. Brain Res 1627:52–69
Gotow T, Leterrier JF, Ohsawa Y et al (1999) Abnormal expression of neurofilament proteins in dysmyelinating axons located in the central nervous system of jimpy mutant mice. Eur J Neurosci 11:3893–3903
Griffiths I, Klugmann M, Anderson T et al (1998a) Axonal swellings and degeneration in mice lacking the major proteolipid of myelin. Science 280:1610–1613
Griffiths I, Klugmann M, Anderson T et al (1998b) Current concepts of PLP and its role in the nervous system. Microsc Res Tech 41:344–358
Gruenenfelder FI, Thomson G, Penderis J, Edgar JM (2011) Axon-glial interaction in the CNS: what we have learned from mouse models of Pelizaeus–Merzbacher disease. J Anat 219:33–43
He Y, Appel S, Le W (2001) Minocycline inhibits microglial activation and protects nigral cells after 6-hydroxydopamine injection into mouse striatum. Brain Res 909:187–193
Hetz C (2012) The unfolded protein response: controlling cell fate decisions under ER stress and beyond. Nat Rev Mol Cell Biol 13:89–102
Hirano A, Sax DS, Zimmerman HM (1969) The fine structure of the cerebella of jimpy mice and their “normal” litter mates. J Neuropathol Exp Neurol 28:388–400
Horie M, Watanabe K, Bepari AK et al (2014) Disruption of actin-binding domain-containing Dystonin protein causes dystonia musculorum in mice. Eur J Neurosci 40:3458–3471
Horie M, Mekada K, Sano H et al (2016) Characterization of novel dystonia musculorum mutant mice: implications for central nervous system abnormality. Neurobiol Dis 96:271–283
Ikenaka K, Kagawa T, Mikoshiba K (1992) Selective expression of DM-20, 1n alternatively spliced myelin proteolipid protein gene product, in developing nervous system and in nonglial cells. J Neurochem 58:2248–2253
Kagawa T, Ikenaka K, Inoue Y et al (1994) Glial cell degeneration and hypomyelination caused by overexpression of myelin proteolipid protein gene. Neuron 13:427–442
Kapalanga J, Blecher SR (1990) Effect of the X-linked gene Tabby (Ta) on eyelid opening and incisor eruption in neonatal mice is opposite to that of epidermal growth factor. Development 108:349–355
Klugmann M, Schwab MH, Pühlhofer A et al (1997) Assembly of CNS myelin in the absence of proteolipid protein. Neuron 18:59–70
Kobayashi H, Hoffman EP, Marks HG (1994) The rumpshaker mutation in spastic paraplegia. Nat Genet 7:351–352
Kraus-Ruppert R, Herschkowitz N, Fürst S (1973) Morphological studies on neuroglial cells in the corpus callosum of the jimpy mutant mouse. J Neuropathol Exp Neurol 32:197–202
Kudo T, Kanemoto S, Hara H et al (2008) A molecular chaperone inducer protects neurons from ER stress. Cell Death Differ 15:364–375
Lachapelle F, Lapie P, Gansmuller A, Villarroya H, Gumpel M (1990) Patchy myelination pattern in the jimpy mouse brain: immunohistochemical study. Glia 3:375–384
Lee Y, Morrison BM, Li Y et al (2012) Oligodendroglia metabolically support axons and contribute to neurodegeneration. Nature 487:443–448
Ma J, Tanaka KF, Shimizu T et al (2011) Microglial cystatin F expression is a sensitive indicator for ongoing demyelination with concurrent remyelination. J Neurosci Res 89:639–649
Macklin WB, Gardinier MV, King KD, Kampf K (1987) An AG→GG transition at a splice site in the myelin proteolipid protein gene in jimpy mice results in the removal of an exon. FEBS Lett 223:417–421
Macklin WB, Gardinier MV, Obeso ZO (1990) Structure and expression of the mouse myelin proteolipid protein gene. Ann N Y Acad Sci 605:183–193
Mayer TC, Green MC (1978) Epidermis is the site of action of tabby (Ta) in the mouse. Genetics 90:125–131
Meares GP, Mines MA, Beurel E et al (2011) Glycogen synthase kinase-3 regulates endoplasmic reticulum (ER) stress-induced CHOP expression in neuronal cells. Exp Cell Res 317:1621–1628
Menn B, Garcia-Verdugo JM, Yaschine C, Gonzalez-Perez O, Rowitch D, Alvarez-Buylla A (2006) Origin of oligodendrocytes in the subventricular zone of the adult brain. J Neurosci 26:7907–7918
Mikkola ML, Pispa J, Pekkanen M et al (1999) Ectodysplasin, a protein required for epithelial morphogenesis, is a novel TNF homologue and promotes cell-matrix adhesion. Mech Dev 88:133–146
Mori K (2009) Signalling pathways in the unfolded protein response: development from yeast to mammals. J Biochem 146:743–750
Nave KA, Lai C, Bloom FE, Milner RJ (1986) Jimpy mutant mouse: a 74-base deletion in the mRNA for myelin proteolipid protein and evidence for a primary defect in RNA splicing. Proc Natl Acad Sci USA 83:9264–9268
Nave KA, Lai C, Bloom FE, Milner RJ (1987) Splice site selection in the proteolipid protein (PLP) gene transcript and primary structure of the DM-20 protein of central nervous system myelin. Proc Natl Acad Sci USA 84:5665–5669
Nicholas R, Stevens S, Wing M, Compston A (2003) Oligodendroglial-derived stress signals recruit microglia in vitro. Neuroreport 14:1001–1005
Omlin FX, Anders JJ (1983) Abnormal cell relationships in jimpy mice: electron microscopic and immunocytochemical findings. J Neurocytol 12:767–784
Oyadomari S, Mori M (2004) Roles of CHOP/GADD153 in endoplasmic reticulum stress. Cell Death Differ 11:381–389
Phillips RJ (1954) Jimpy, a new totally sexlinked gene in the house mouse. Z Indukt Abstamm Vererbungsl 86:322–326
Readhead C, Schneider A, Griffiths I, Nave KA (1994) Premature arrest of myelin formation in transgenic mice with increased proteolipid protein gene dosage. Neuron 12:583–595
Richardson WD, Pringle NP, Yu WP, Hall AC (1997) Origins of spinal cord oligodendrocytes: possible developmental and evolutionary relationships with motor neurons. Dev Neurosci 19:58–68
Rivers LE, Young KM, Rizzi M et al (2008) PDGFRA/NG2 glia generate myelinating oligodendrocytes and piriform projection neurons in adult mice. Nat Neurosci 11:1392–1401
Roussel G, Neskovic NM, Trifilieff E, Artault JC, Nussbaum JL (1987) Arrest of proteolipid transport through the Golgi apparatus in jimpy brain. J Neurocytol 16:195–204
Rowitch DH, Lu QR, Kessaris N, Richardson WD (2002) An ‘oligarchy’ rules neural development. Trends Neurosci 25:417–422
Saab AS, Tzvetanova ID, Nave KA (2013) The role of myelin and oligodendrocytes in axonal energy metabolism. Curr Opin Neurobiol 23:1065–1072
Salzer JL (2003) Polarized domains of myelinated axons. Neuron 40:297–318
Schindler P, Luu B, Sorokine O, Trifilieff E, Van Dorsselaer A (1990) Developmental study of proteolipids in bovine brain: a novel proteolipid and DM-20 appear before proteolipid protein (PLP) during myelination. J Neurochem 55:2079–2085
Schneider A, Montague P, Griffiths IR, Fanarraga ML, Kennedy PGE, Brophy PJ, Nave K-A (1992) Uncoupling of hypomyelination and glial cell death by a mutation in the proteolipid protein gene. Nature 358:758–761
Sheridan GK, Murphy KJ (2013) Neuron–glia crosstalk in health and disease: fractalkine and CX3CR1 take centre stage. Open Biol 3:130181
Silva RM, Ries V, Oo TF et al (2005) CHOP/GADD153 is a mediator of apoptotic death in substantia nigra dopamine neurons in an in vivo neurotoxin model of parkinsonism. J Neurochem 95(4):974–986
Southwood CM, Garbern J, Jiang W, Gow A (2002) The unfolded protein response modulates disease severity in Pelizaeus–Merzbacher disease. Neuron 36:585–596
Southwood CM, Fykkolodziej B, Maheras KJ, Garshott DM, Estill M, Fribley AM, Gow A (2016) Overexpression of CHOP in myelinating cells does not confer a significant phenotype under normal or metabolic stress conditions. J Neurosci 36:6803–6819
Srivastava AK, Durmowicz MC, Hartung AJ et al (2001) Ectodysplasin-A1 is sufficient to rescue both hair growth and sweat glands in Tabby mice. Hum Mol Genet 10:2973–2981
Tajiri S, Oyadomari S, Yano S et al (2004) Ischemia-induced neuronal cell death is mediated by the endoplasmic reticulum stress pathway involving CHOP. Cell Death Differ 11:403–415
Takebayashi H, Ikenaka K (2015) Oligodendrocyte generation during mouse development. Glia 63:1350–1356
Takebayashi H, Yoshida S, Sugimori M et al (2000) Dynamic expression of basic helix-loop-helix Olig family members: implication of Olig2 in neuron and oligodendrocyte differentiation and identification of a new member, Olig3. Mech Dev 99:143–148
Timsit SG, Bally-Cuif L, Colman DR, Zalc B (1992) DM-20 mRNA is expressed during the embryonic development of the nervous system of the mouse. J Neurochem 58:1172–1175
Torii T, Miyamoto Y, Yamauchi J, Tanoue A (2014) Pelizaeus–Merzbacher disease: cellular pathogenesis and pharmacologic therapy. Pediatr Int 56:659–666
Watanabe K, Tamamaki N, Furuta T, Ackerman SL, Ikenaka K, Ono K (2006) Dorsally derived netrin 1 provides an inhibitory cue and elaborates the ‘waiting period’ for primary sensory axons in the developing spinal cord. Development 133:1379–1387
Yamamura T, Konola JT, Wekerle H, Lees MB (1991) Monoclonal antibodies against myelin proteolipid protein: identification and characterization of two major determinants. J Neurochem 57:1671–1680
Acknowledgements
This study was supported by research grants from the Ministry of Education, Culture, Sports, Science, and Technology of Japan; Grant-in-Aid for Scientific Research (B) (15H04667, H.T.), Grant-in-Aid for Scientific Research on Innovative Areas, “Glial assembly” (25117007, H.T.) and Grant-in-Aid for Exploratory Research (16K15168, H.T.); and a Grant from the Niigata University Kyowakai Society (M.I.). We thank Dr. Tetsushi Kagawa for PLP plasmid; Dr. Yukiko Mori and Ms. Satoko Yamagiwa for technical assistance; Dr. Yoshihide Yamaguchi, Dr. Mari Tada, Dr. Akiyoshi Kakita, Dr. Kazunori Imaizumi, Dr. Takashi Kudo for discussion; all Takebayashi laboratory members, especially Dr. Norihisa Bizen and Dr. Masato Yano, for providing input and advice, and Dr. Sugata Takahashi for continual support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12565_2016_383_MOESM1_ESM.pptx
Fig. S1a,b Ta-jp mutant mice. a Ta-jp mutant mouse (Ta-jp/Y, right) and control wild-type mouse (X/Y, left) at P14. b Female heterozygous mouse (Ta-jp/X, right) and wild-type mouse (X/X, left) at P21. Female heterozygous mouse showed hair color phenotype, stripes in the back (PPTX 247 kb)
12565_2016_383_MOESM2_ESM.pptx
Fig. S2a,b Increased expression of microglia marker CD11b in the white matter. a, b CD11b immunostaining in the coronal sections of wild-type mouse (a) and Ta-jp mutant mouse (b) at P21. Corpus callosum (a′, b′) and internal capsule (a″,b″) were shown in higher magnifications. Lots of CD11b-positive microglia were observed in the internal capsule of Ta-jp mutant (b″). Counterstaining was performed with Mayer’s Hematoxylin. Bars: a, b 1 mm; a′, b′, a′′, b′′ 50 μm (PPTX 1465 kb)
12565_2016_383_MOESM3_ESM.pptx
Fig. S3a–f Characterization of neurons and astrocytes in the brain of Ta-jp mutant. a, b NF-H immunostaining in the coronal sections of wild-type brain (a) and Ta-jp mutant brain (b) at P21. Cerebral cortex (a′, b′) and internal capsule (a″, b″) are shown at higher magnification. Strong NF-H signals were observed in the axons of cortex and internal capsule of Ta-jp mutant. c, d NF-M immunostaining in the coronal sections of wild-type brain (c) and Ta-jp mutant brain (d) at P21. Cerebral cortex (c′, d′) and internal capsule (c″, d″) are shown at higher magnification. Strong NF-M signals were observed in the axons of internal capsule of Ta-jp mutant. (e, f) GFAP immunostaining in the coronal sections of wild-type brain (e) and Ta-jp mutant brain (f) at P21. Activated astrocytes with strong GFAP imuunoreactivity were observed in the white matter of Ta-jp mutant brain (f). Pictures of corpus callosum (e′, f′) and internal capsule (e″, f″) are shown at higher magnification. Insets High magnification picture of dot-lined rectangle area. Counterstaining was performed with Mayer’s hematoxylin. Bars a–f 1 mm; a′–f′, a″–f″ 50 μm; insets of a′–d′, e″, f″ 20 µm; insets of a″–d″ 10 µm (PPTX 5167 kb)
12565_2016_383_MOESM4_ESM.pptx
Fig. S4a–d Double staining of CHOP ISH and IHC of glial markers. a Double staining experiment using CHOP probe (purple) and Olig2 antibody (brown) in the coronal sections of Ta-jp mutant brain at P21. b Double staining experiment using CHOP probe (purple) and CC-1 antibody (brown) in the coronal sections of Ta-jp mutant brain at P21. c Double staining experiment using CHOP probe (purple) and GFAP antibody (brown) in the coronal sections of Ta-jp mutant brain at P21. d Double staining experiment using CHOP probe (purple) and Iba1 antibody (brown) in the coronal sections of Ta-jp mutant brain at P21. Note that CHOP signal co-localizes with OL makers, Olig2 (a″) and CC-1 (b″) in internal capsule. Bars a–d 500 μm, a′–d′ 100 μm, a″–d″ 40 μm, insets 20 μm (PPTX 3776 kb)
12565_2016_383_MOESM5_ESM.pptx
Fig. S5a–c Apoptotic marker staining. a, b Immunostaining using anti-cleaved caspase-3 antibody in the coronal sections of wild-type brain (a) and Ta-jp mutant brain (b) at P21. Positive cells are indicated by arrowheads. The insets show cleaved caspase-3 positive cells noted by small rectangles. c Quantification of anti-cleaved caspase-3 positive cells in the cerebral cortex, corpus callosum and internal capsule at P21 (n = 3). More number of cleaved caspase-3 positive cells were observed in the white matters of Ta-jp mutant brain (arrowheads). Bars a, b 1 mm; a′, b′, a″, b″ 50 μm; insets 10 μm (PPTX 1541 kb)
Movie S1 Ta-jp mutant mouse (2 weeks old). One Ta-jp mutant (Ta-jp/Y) and two control littermates (+/Y) at P14. The Ta-jp mutant has smaller body than wild-type mice and shows a shivering phenotype and ataxic gait (MP4 15735 kb)
Movie S2 Ta-jp mutant mouse (3 weeks old). One Ta-jp mutant (Ta-jp/Y) and one control littermate (+/Y) at P21. The Ta-jp mutant has smaller body than wild-type mice and shows shivering phenotype with extended and spastic hindlimbs (MP4 6848 kb)
Rights and permissions
About this article
Cite this article
Ikeda, M., Hossain, M.I., Zhou, L. et al. Histological detection of dynamic glial responses in the dysmyelinating Tabby-jimpy mutant brain. Anat Sci Int 93, 119–127 (2018). https://doi.org/10.1007/s12565-016-0383-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12565-016-0383-5