Abstract
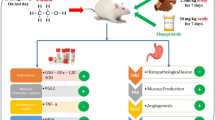
Tea polyphenols are known to prevent various ailments like cancer, atherosclerosis, hypertension and diabetes. Our study aimed at to decipher the gastroprotective effect of aqueous black tea extract (BTE) against ethanol-induced gastric damage and the role of BTE in modulating MMP-9 activity and expression, both in vivo and in vitro. The protective role of BTE was assessed in Sprague–Dawley rats after inducing damage with 70% ethanol. Human gastric adenocarcinoma cells (AGS) were treated with ethanol in ex vivo experiment. MMP-9 activity and expression were investigated through gelatin zymography and western blotting. Reactive oxygen species (ROS) generation was also studied by fluorescence spectroscopy and confocal microscopy, with or without treatment of BTE both in vivo and in vitro experiments. In addition, the effect of citric acid treated BTE (cBTE), which mimics lemon tea, was examined on ethanol-induced gastropathy. BTE exhibited antiulcer activity through reduction of glutathione depletion, lipid peroxidation, protein oxidation, ROS production and inflammatory cell infiltration in rat gastric tissues. In addition, BTE significantly inhibited synthesis and secretion of proMMP-9 both in vivo and in vitro. The mitochondrial enzymes succinate dehydrogenase and NADH oxidase in rat gastric tissues were downregulated by BTE while protecting gastric ulcer. Citric acid addition to BTE was observer to enrich the lead compound, catechin. Interestingly, cBTE showed higher anti-ulcer activity than the untreated one. BTE shows protective role against ethanol-induced gastric ulcer in rats through scavenging ROS and downregulating proMMP-9 activity. While cBTE shows better protection due to enrichment of catechin and removal of tannins in tea extract leading to enhanced inhibitory role on proMMP-9 activity and ROS production.
Graphical Abstract







Similar content being viewed by others
Abbreviations
- BTE:
-
Black tea extract
- cBTE:
-
Citric acid treated black tea extract
- ROS:
-
Reactive oxygen species
- MMP:
-
Matrix metalloproteinase
- ECM:
-
Extracellular matrix
- SDH:
-
Succinate dehydrogenase
- TIMP:
-
Tissue inhibitor of metalloproteinases
References
El-Moselhy MA, Abdel-Hamid NM, Abdel-Raheim SR. Gastroprotective effect of nicorandil in indomethacin and alcohol-induced acute ulcers. Appl Biochem Biotechnol. 2009;152(3):449–59.
Shahin M, Konturek JW, Pohle T, Schuppan D, Herbst H, Domschke W. Remodeling of extracellular matrix in gastric ulceration. Microsc Res Tech. 2001;53(6):396–408.
Chakraborty S, Stalin S, Das N, Choudhury ST, Ghosh S, Swarnakar S. The use of nano-quercetin to arrest mitochondrial damage and MMP-9 upregulation during prevention of gastric inflammation induced by ethanol in rats. Biomaterials. 2011;33(10):2991–3001.
Swarnakar S, Mishra A, Ganguly K, Sharma AV. Matrix metalloproteinase-9 activity and expression is reduced by melatonin during prevention of ethanol-induced gastric ulcer in mice. J Pineal Res. 2007;43(1):56–64.
Ganguly K, Kundu P, Banerjee A, Reiter RJ, Swarnakar S. Hydrogen peroxide-mediated downregulation of matrix metalloprotease-2 in indomethacin-induced acute gastric ulceration is blocked by melatonin and other antioxidants. Free Rad Biol Med. 2006;41(6):911–25.
Robbins SL, Coltran RS, Healing Kumar V. Pathologic bases of disease. Philadelphia: WB Saunders; 1984. p. 816–20.
Page-McCaw A, Ewald AJ, Werb Z. Matrix metalloproteinases and the regulation of tissue remodeling. Nat Rev Mol Cell Biol. 2007;8(3):221–33.
Bonnans C, Chou J, Werb Z. Remodelling the extracellular matrix in development and disease. Nat Rev Mol Cell Biol. 2014;15(12):786–801.
Guedez L, Lim MS, Stetler-Stevenson WG. The role of metalloproteinases and their inhibitors in hematological disorders. Crit Rev Oncog. 1996;7(3–4):205–25.
Zhang Y, McCluskey K, Fujii K, Wahl LM. Differential regulation of monocyte matrix metalloproteinase and TIMP-1 production by TNF-alpha, granulocyte-macrophage CSF, and IL-1beta through prostaglandin-dependent and -independent mechanisms. J Immunol. 1998;161(6):3071–6.
Gordon GM, Ledee DR, Feuer W, Fini ME. Cytokines and signaling pathways regulating matrix metalloproteinase-9 (MMP-9) expression in corneal epithelial cells. J Cell Physiol. 2009;221(2):402–11.
Lempinen M, Inkinen K, Wolff H, Ahonen J. Matrix Metalloproteinases 2 and 9 in Indomethacin-Induced Rat Gastric Ulcer. Eur Surg Res. 2000;32(3):169–76.
Singh LP, Mishra A, Saha D, Swarnakar S. Doxycycline blocks gastric ulcer by regulating matrix metalloproteinase-2 activity and oxidative stress. World J Gastroenterol. 2011;17(28):3310–21.
Greenlee KJ, Werb Z, Kheradmand F. Matrix metalloproteinases in lung: multiple, multifarious, and multifaceted. Physiol Rev. 2007;87(1):69–98.
Furuichi K, Hisada Y, Shimizu M, Okumura T, Kitagawa K, Yoshimoto K, et al. Matrix metalloproteinase-2 (MMP-2) and membrane-type 1 MMP (MT1-MMP) affect the remodeling of glomerulosclerosis in diabetic OLETF rats. Nephrol Dial Transplant. 2011;26(10):3124–31.
Matsui H, Shimokawa O, Kaneko T, Nagano Y, Rai K, Hyodo I. The pathophysiology of non-steroidal anti-inflammatory drug (NSAID)-induced mucosal injuries in stomach and small intestine. J Clin Biochem Nutr. 2011;48(2):107–11.
Wiseman SA, Balentine DA, Frei B. Antioxidants in tea. Crit Rev Food Sci Nutr. 1997;37(8):705–18.
Rietveld A, Wiseman S. Antioxidant effects of tea: evidence from human clinical trials. J Nutr. 2003;133(10):3285–92.
Frei B, Higdon JV. Antioxidant activity of tea polyphenols in vivo: evidence from animal studies. J Nutr. 2003;133(10):3275S–84S.
Leung LK, Su Y, Chen R, Huang Y, Chen ZY. Theaflavins in black tea and catechins in green tea are equally effective antioxidants. J Nutr. 2001;131(9):2248–51.
Ghosh MN. Fundamentals of experimental pharmacology. 3rd ed. Tysons Corner: Hilton and Co; 2005. p. 190–7.
Swarnakar S, Ganguly K, Kundu P, Banerjee A, Maity P, Sharma AV. Curcumin regulates expression and activity of matrix metalloproteinase 9 and 2 during prevention and healing of indomethacin-induced gastric ulcer. J Biol Chem. 2005;280(10):9409–15.
Twentyman PR, Luscombe MA. Study of some variables in a tetrazolium dye (MTT) based assay for cell growth and chemosensitivity. Br J Cancer. 1987;56(3):279–85.
Nishitani E, Sagesaka YM. Simultaneous determination of catechins, caffeine and other phenolic compounds in tea using new HPLC method. J Food Compos Anal. 2004;17(5):675–775.
Ghosh S, Chakraborty R, Majumdar S, Raychaudhuri U. Identification of ultraviolet radiation induced gallic acid and caffeic acid formation in palm juice (Borassus flabellifer) by HPLC & mass spectra technique. J Food Meas Charact. 2014;8(3):218–24.
Ho Row K, Jin Y. Recovery of catechin compounds from Korean tea by solvent extraction. Bioresour Technol. 2006;97(5):790–3.
Recknagel RO, Glende EA Jr. Spectrophotometric detection of lipid conjugated dienes. Methods Enzymol. 1984;105:331–7.
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with folin phenol reagent. J Biol Chem. 1951;193:265–75.
Singh LP, Kundu P, Ganguly K, Mishra A, Swarnakar S. Novel role of famotidine in downregulation of matrix metalloproteinase-9 during protection of ethanol-induced acute gastric ulcer. Free Rad Biol Med. 2007;43(2):289–99.
Davila JC, Davis PS, Acosta D. Changes in glutathione and cellular energy as potential mechanisms of papaverine-induced hepatotoxicity in vitro. Toxicol Appl Pharmacol. 1991;108(1):28–36.
Navarro A, Boveris A. Rat brain and liver mitochondria develop oxidative stress and lose enzymatic activities on aging. Am J Physiol Regul Integr Comp Physiol. 2004;287(5):1244–9.
Hamaishi K, Kojima R, Hahm KB. Anti-ulcer effect of tea catechin in rats. Biol Pharm Bull. 2006;29(11):2206–13.
Lee SY, Shin YW, Hahm KB. Phytoceuticals: mighty but ignored weapons against Helicobacter pylori infection. J Dig Dis. 2008;9(3):129–39.
Galvin GB, Szabo S. Experimental gastric mucosal injury: laboratory models reveal mechanisms of pathogenesis and new therapeutic strategies. FASEB J. 1992;6(3):825–31.
Loguercio C, Nardi G, Romano M. Glutathione prevents ethanol induced gastric mucosal damage and depletion of sulfydryl compounds in humans. Gut. 1993;34(2):161–5.
Yoshikawa T, Naito Y, Kishi A, Tomii T, Kaneko T, Linuma S, et al. Role of active oxygen, lipid peroxidation, and antioxidants in the pathogenesis of gastric mucosal injury induced by indomethacin in rats. Gut. 1993;34(6):732–7.
Nicoue EE, Savard S, Belkacemi K. Anthocyanins in wild blueberries of Quebec: extraction and identification. J Agric Food Chem. 2007;55(14):5626–35.
Muzolf M, Szymusiak H, Gliszczyn A, Rietjens I, Tyrakowska B. pH-dependent radical scavenging capacity of green tea catechins. J Agric Food Chem. 2008;56(3):816–23.
Acknowledgements
This work was financed by the CSIR Network Projects HUM (BSC 0119) and INDEPTH (BSC 0111). Mr. Diptadeep Sarkar and Mr. T. Muruganandan are thankfully acknowledged for their assistance in confocal microscopy and scanning electron microscopy (SEM).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Raychaudhuri, S., Ghosh, S., Roy, A. et al. Protective Role of Black Tea Flavonoids Against Ethanol-Induced Gastropathy via Matrix Metalloproteinase Pathway. Ind J Clin Biochem 34, 379–394 (2019). https://doi.org/10.1007/s12291-018-0762-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12291-018-0762-x