Abstract
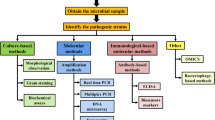
Peptide nucleic acids (PNAs) are DNA/RNA analogs in which sugar-phosphate backbone is replaced by N-2-aminoethylglycine repeating units. Since PNA contains a neutral skeleton, there is no electrostatic repulsion, resulting in significant stability of its hybrid structure with complementary oligonucleotides. At present, PNA has taken the place of DNA probe in many studies. There are several disadvantages of cellular uptake of PNA, so modifications in PNA backbone or covalent coupling with cell-penetrating peptides are necessary to improve its delivery inside the cells. In recent years, PNA has been extensively used in the rapid detection of microorganisms, such as fluorescence in situ hybridization, PCR amplification, biosensor, and gene chip. The structure and characteristics of PNA probe, the hybridization method, and the design principle of PNA probes are introduced in this review, and the application progress of PNA probes in the rapid detection of foodborne pathogens is summarized. On this basis, the advantages and disadvantages of PNA probes are analyzed, and the future development trend is prospected.











Similar content being viewed by others
References
Afshari A, Baratpour A, Ithanzade S, Jamshidi A (2018) Salmonella Enteritidis and Salmonella Typhimorium identification in poultry carcasses. Iranian J Microbiol 10:45–50
Almeida C, Azevedo NF, Santos S, Keevil CW, Vieira MJ (2011) Discriminating multi-species populations in biofilms with peptide nucleic acid fluorescence in situ hybridization (PNA FISH). PLoS One 6:13. https://doi.org/10.1371/journal.pone.0014786
Almeida C, Sousa JM, Rocha R, Cerqueira L, Fanning S, Azevedo NF, Vieira MJ (2013) Detection of Escherichia coli O157 by peptide nucleic acid fluorescence in situ hybridization (PNA-FISH) and comparison to a standard culture method. Appl Environ Microbiol 79:6293–6300. https://doi.org/10.1128/AEM.01009-13
Bertucci A, Lulf H, Septiadi D, Manicardi A, Corradini R, De Cola L (2014) Intracellular delivery of peptide nucleic acid and organic molecules using zeolite-L nanocrystals. Adv Healthc Mater 3:1812–1817. https://doi.org/10.1002/adhm.201400116
Breipohl G, Will DW, Peyman A, Uhlmann E (2010) ChemInform Abstract: novel synthetic routes to PNA monomers and PNA-DNA linker molecules. Tetrahedron 29:14671–14868. https://doi.org/10.1002/chin.199808209
Cheng F, Yi F, Jinming K, Zhiqiang G, Narayanan B (2008) Electrical detection of oligonucleotide using an aggregate of gold nanoparticles as a conductive tag. Anal Chem 80:9387–9394. https://doi.org/10.1021/ac801433z
Cherny DY, Belotserkovskii BP, Frank-Kamenetskii MD, Egholm M, Buchardt O, Berg RH, Nielsen PE (1993) DNA unwinding upon strand-displacement binding of a thymine-substituted polyamide to double-stranded DNA. Proc Natl Acad Sci U S A 90:1667–1670. https://doi.org/10.1073/pnas.90.5.1667
Chim W, Sedighi A, Brown CL, Pantophlet R, Li PCH (2018) Effect of buffer composition on PNA-RNA hybridization studied in the microfluidic microarray chip. Can J Chem 96:241–247. https://doi.org/10.1139/cjc-2017-0319
Choi JJ, Kim CH (2009) Peptide nucleic acid-based array for detecting and genotyping human papillomaviruses. J Clin Microbiol 47:1785–1790. https://doi.org/10.1128/JCM.01398-08
Chunyan Y et al (2008) Hybridization assay of hepatitis B virus by QCM peptide nucleic acid biosensor. Biosens Bioelectron 23:879–885. https://doi.org/10.1016/j.bios.2007.09.003
Debaene F, Winssinger N (2003) Azidopeptide nucleic acid. An alternative strategy for solid-phase peptide nucleic acid (PNA) synthesis. Org Lett 5:4445–4447. https://doi.org/10.1021/ol0358408
Ditmangklo B, Boonlua C, Suparpprom C, Vilaivan T (2013) Reductive alkylation and sequential reductive alkylation-click chemistry for on-solid-support modification of pyrrolidinyl peptide nucleic acid. Bioconjug Chem 24:614–625. https://doi.org/10.1021/bc3005914
Dong B, Nie K, Shi H, Li W, Liu Z (2016) Design, synthesis, properties, and applications of chiral peptide nucleic acid monomers. Curr Org Chem 20:2703–2717. https://doi.org/10.2174/1385272820666160505120014
Dueholm KL et al (1994) Synthesis of peptide nucleic acid monomers containing the four natural nucleobases: thymine, cytosine, adenine, and guanine and their oligomerization. J Organic Chem:5767–5773. https://doi.org/10.1021/jo00098a042
Egholm M, Nielsen PE, Buchardt O, Berg RH (1992) Recognition of guanine and adenine in DNA by cytosine and thymine containing peptide nucleic acids (PNA). Jamchemsoc 114:9677–9678. https://doi.org/10.1021/ja00050a068
Egholm M et al (1993) PNA hybridizes to complementary oligonucleotides obeying the Watson-crick hydrogen-bonding rules. Nature. https://doi.org/10.1038/365566a0
Enrico B et al (2009) The oncogenic kinase Bcr-Abl directly regulates splicing of BcIX through a quaternary complex coordinated by Nck-Beta and Sam-68 adapter proteins. Blood 114:854
Fabani MM, Gait MJ (2008) miR-122 targeting with LNA/2’-O-methyl oligonucleotide mixmers, peptide nucleic acids (PNA), and PNA-peptide conjugates. Rna 14:336–346. https://doi.org/10.1261/rna.844108
Ferreira AM, Cruz-Moreira D, Cerqueira L, Miranda JM, Azevedo NF (2017) Yeasts identification in microfluidic devices using peptide nucleic acid fluorescence in situ hybridization (PNA-FISH). Biomed Microdevices 19:11. https://doi.org/10.1007/s10544-017-0150-y
Gaylord BS, Massie MR, Feinstein SC, Bazan GC (2005) SNP detection using peptide nucleic acid probes and conjugated polymers: applications in neurodegenerative disease identification. Proc Natl Acad Sci U S A 102:34–39. https://doi.org/10.1073/pnas.0407578101
Golfier G, Lemoine S, Van MA, Bendjoudi A, Rossier J, Le CS, Potier MC (2009) Selection of oligonucleotides for whole-genome microarrays with semi-automatic update. Bioinformatics 25:128–129. https://doi.org/10.1093/bioinformatics/btn573
Gootenberg JS et al (2017) Nucleic acid detection with CRISPR-Cas13a/C2c2. Science 356:438–442. https://doi.org/10.1126/science.aam9321
Gupta A, Mishra A, Puri N (2017) Peptide nucleic acids: advanced tools for biomedical applications. J Biotechnol 259:148–159. https://doi.org/10.1016/j.jbiotec.2017.07.026
He XP, Zou BJ, Qi XM, Chen S, Lu Y, Huang Q, Zhou GH (2019) Methods of isothermal nucleic acid amplification-based microfluidic chips for pathogen microorganism detection. Yi chuan = Hereditas 41:611–624. https://doi.org/10.16288/j.yczz.19-051
Hejazi MS, Pournaghi-Azar MH, Alipour E, Abdolahinia ED, Arami S, Navvah H (2011) Development of a novel electrochemical biosensor for detection and discrimination of DNA sequence and single base mutation in dsDNA samples based on PNA-dsDNA hybridization—a new platform technology. Electroanalysis 23:503–511. https://doi.org/10.1002/elan.201000413
Howarth NM, Wakelin LPG (1998) α-PNA: a novel peptide nucleic acid analogue of DNA. Cheminform 29:5441–5450. https://doi.org/10.1021/jo970111p
Jang H, Kim JChoi JJ, Son Y, Park H (2010) Peptide nucleic acid array for detection of point mutations in hepatitis B virus associated with antiviral resistance. J Clin Microbiol 48:3127–3131. https://doi.org/10.1128/JCM.02058-09
Johann M, Celia R, Montserrat C, Daniel R, Jose Angel MG, Carlos B, Javier T (2008) Label-free detection of DNA hybridization based on hydration-induced tension in nucleic acid films. Nat Nanotechnol 3:301–307. https://doi.org/10.1038/nnano.2008.91
Kira A, Matsuo K, Nakajima S-I (2016) DNA hybridization activity of single-stranded DNA-conjugated gold nanoparticles used as probes for DNA detection. Jpn J Appl Phys 55:223–225. https://doi.org/10.7567/jjap.55.02be04
Krishna MS et al (2019) Sequence- and structure-specific probing of RNAs by short nucleobase-modified dsRNA-binding PNAs (dbPNAs) incorporating a fluorescent light-up uracil analog. Anal Chem:5331–5338. https://doi.org/10.1021/acs.analchem.9b00280
Kuhnast B, Dolle F, Tavitian B (2002) Fluorine-18 labeling of peptide nucleic acids. J Label Compd Radiopharm 45:1–11. https://doi.org/10.1002/jlcr.522
Kuwahara M, Arimitsu M, Sisido M (2010) ChemInform Abstract: synthesis of δ-amino acids with an ether linkage in the main chain and nucleobases on the side chain as monomer units for oxy-peptide nucleic acids. Tetrahedron 30:10067–10078. https://doi.org/10.1002/chin.199951189
Liu Z, Li W, Shi H, Nie K, Bo D (2016) Design, synthesis, properties, and applications of chiral peptide nucleic acid monomers. Curr Org Chem 20:1. https://doi.org/10.2174/1385272820666160505120014
Lowe G, Vilaivan T (1997) Amino acids bearing nucleobases for the synthesis of novel peptide nucleic acids. J Chem Soc Perkin Trans 1:555–560. https://doi.org/10.1002/chin.199729223
Machado A et al (2013) Fluorescence in situ hybridization method using a peptide nucleic acid probe for identification of Lactobacillus spp. in milk samples. Int J Food Microbiol 162:64–70. https://doi.org/10.1016/j.ijfoodmicro.2012.09.024
MacKay J, Williams A, Sofka H, Rozners E (2018) Design and synthesis of pi-extended nucleobases for sequence selective triple-helical recognition of RNA using peptide nucleic acids. Abstracts of Papers of the American Chemical Society 255
Malamgari SR, Manikandan P, Ramani P, Katta VR (2018) Synthesis of peptide nucleic acid monomers via N-alkylation of nosyl-protected amino acids with N-Boc bromoethyl amine. Chem Select 3:3948–3951. https://doi.org/10.1002/slct.201800202
Mário SJ, Rocha R , Cerqueira L , Almeida C, Azevedo NF, Bastin B, Bird P, Benzinger MJ, Agin J, Goins D, Chen Y, Brodsky M, Odumoru J (2019) Validation of biomode S.A. probe4cronobacterTM for the identification of cronobacter spp. J AOAC Int 102(3):855–864. https://doi.org/10.5740/jaoacint.18-0328
Nielsen PE, Egholm M, Berg RH, Buchardt O (1991) Sequence-selective recognition of DNA by strand displacement with a thymine-substituted polyamide. Science 254:1497–1500. https://doi.org/10.1126/science.1962210
Ogita K, Suzuki T, Enomoto R, Han D, Yoneda Y (2003) Direct identification of Staphylococcus aureus from positive blood culture bottles. J Clin Microbiol 41:889–891. https://doi.org/10.1128/JCM.41.2.889-891.2003
Ole B et al (2003) PNA microarrays for hybridisation of unlabeled DNA samples. Nucleic Acids Res 31:119e. https://doi.org/10.1093/nar/gng120
Pardee K, Green AA, Takahashi MK, Braff D, Lambert G, Lee JW, Ferrante T, Ma D, Donghia N, Fan M, Daringer NM, Bosch I, Dudley DM, O’Connor DH, Gehrke L, Collins JJ (2016) Rapid, low-cost detection of zika virus using programmable biomolecular components. Cell 165:1255–1266. https://doi.org/10.1016/j.cell.2016.04.059
Pasquer F, Pelludat C, Duffy B, Frey JE (2010) Broad spectrum microarray for fingerprint-based bacterial species identification. BMC Biotechnology 10:13–16 doi:https://doi.org/10.1186/1472-6750-10-13
Pasquier L, Chuard C (2017) Listeria monocytogenes infections. Revue Medicale Suisse 13:1737–1740
Pournaghi-Azar MH, Ahour F, Hejazi MS (2010) Direct detection and discrimination of double-stranded oligonucleotide corresponding to hepatitis C virus genotype 3a using an electrochemical DNA biosensor based on peptide nucleic acid and double-stranded DNA hybridization. Anal Bioanal Chem 397:3581–3857. https://doi.org/10.1007/s00216-010-3875-5
Ramani P, Cauteruccio S, Licandro E, Baldoli C (2018) Synthesis of luminescent 2,3-diphenylmaleimide-labeled peptide nucleic acid oligomers. Tetrahedron Lett 59:2229–2231. https://doi.org/10.1016/j.tetlet.2018.03.060
Roberta DA, Roberto C, Cristina F, Laura Z, Marcello G, Rosangela M, Giuseppe S (2010) Ultrasensitive detection of non-amplified genomic DNA by nanoparticle-enhanced surface plasmon resonance imaging. Chembiochem 9:2067–2070. https://doi.org/10.1016/j.bios.2010.02.008
Rocha R, Almeida C, Azevedo NF (2018) Influence of the fixation/permeabilization step on peptide nucleic acid fluorescence in situ hybridization (PNA-FISH) for the detection of bacteria. PLoS One 13:457–462. https://doi.org/10.1371/journal.pone.0196522
Rohde A, Hammerl JA, Appel B, Dieckmann R, Dahouk SA (2015) FISHing for bacteria in food—a promising tool for the reliable detection of pathogenic bacteria? Food Microbiol 46:395–407. https://doi.org/10.1016/j.fm.2014.09.002
Rownicki M, Wojciechowska M, Wierzba AJ, Czarnecki J, Bartosik D, Gryko D, Trylska J (2017) Vitamin B12 as a carrier of peptide nucleic acid (PNA) into bacterial cells. Sci Rep 7:7644. https://doi.org/10.1038/s41598-017-08032-8
Saarbach J, Masi D, Zambaldo C, Winssinger N (2017) Facile access to modified and functionalized PNAs through Ugi-based solid phase oligomerization. Bioorg Med Chem 25:5171–5177. https://doi.org/10.1016/j.bmc.2017.05.064
Saarbach J, Sabale PM, Winssinger N (2019) Peptide nucleic acid (PNA) and its applications in chemical biology, diagnostics, and therapeutics. Curr Opin Chem Biol 52:112–124. https://doi.org/10.1016/j.cbpa.2019.06.006
Sawata S, Kai E, Ikebukuro K, Iida T, Honda T, Karube I (1999) Application of peptide nucleic acid to the direct detection of deoxyribonucleic acid amplified by polymerase chain reaction. Biosens Bioelectron 14:397–404. https://doi.org/10.1016/s0956-5663(99)00018-4
Seitz O (2000) Solid-phase synthesis of doubly labeled peptide nucleic acids as probes for the real-time detection of hybridization. Angew Chem Int Ed 39:3249–3252. https://doi.org/10.1002/1521-3773(20000915)39:18
Shallcrossa LJ, Hayward AC (2013) The role of the Panton-Valentine leucocidin toxin in staphylococcal disease: a systematic review and meta-analysis. Lancet Infect Dis 13:43–54. https://doi.org/10.1016/S1473-3099(12)70238-4
Tantillo GM, Fontanarosa M, Pinto AD, Musti M (2010) Updated perspectives on emerging vibrios associated with human infections. Lett Appl Microbiol 39:117–126. https://doi.org/10.1111/j.1472-765X.2004.01568.x
Tatsuro E, Kagan K, Naoki N, Yuzuru T, Eiichi T (2005) Label-free detection of peptide nucleic acid-DNA hybridization using localized surface plasmon resonance based optical biosensor. Anal Chem 77:6976–6984. https://doi.org/10.1021/ac0513459
Thomson SA, Josey JA, Cadilla R, Gaul MD, Fred Hassman C, Luzzio MJ, Pipe AJ, Reed KL, Ricca DJ, Wiethe RW, Noble SA (1995) Fmoc mediated synthesis of peptide nucleic-acids. Tetrahedron 51:6179–6194. https://doi.org/10.1016/0040-4020(95)00286-h
Tirayut V (2018) Fluorogenic PNA probes. Beilstein. J Org Chem 14:253–281. https://doi.org/10.3762/bjoc.14.17
Vaijayanthi T, Bando T, Pandian GN, Sugiyama H (2012) Progress and prospects of pyrrole-imidazole polyamide-fluorophore conjugates as sequence-selective DNA probes. ChemBioChem 13:2170–2185. https://doi.org/10.1002/cbic.201200451
Walker MJ, Burns M, Quaglia M, Nixon G, Hopley CJ, Gray KM, Moore V, Singh M, Cowen S (2018) Almond or mahaleb? Orthogonal allergen analysis during a live incident investigation by ELISA, molecular biology, and protein mass spectrometry. J AOAC Int 101:162–169. https://doi.org/10.5740/jaoacint.17-0405
Wang P (2009) Recent advances in the rapid diagnosis of various infectious diseases by PNA FISH. Rev Med Microbiol 20:56–62. https://doi.org/10.1097/MRM.0b013e328331ada5
Will DW, Breipohl G, Langner D, Knolle J, Uhlmann E (1995) The synthesis of polyamide nucleic acids using a novel monomethoxytrityl protecting-group strategy. Tetrahedron 51:12069–12082. https://doi.org/10.1016/0040-4020(95)00766-2
Xu Y et al (2018) Nucleic acid biosensor synthesis of an all-in-one universal blocking linker recombinase polymerase amplification with a peptide nucleic acid-based lateral flow device for ultrasensitive detection of food pathogens. Anal Chem 90:708–715. https://doi.org/10.1021/acs.analchem.7b01912
Yang X et al (2017) PCR-free colorimetric DNA hybridization detection using a 3D DNA nanostructured reporter probe. ACS Appl Mater Interfaces 9:38281–38287. https://doi.org/10.1021/acsami.7b11994
Zhang GJ (2011) Silicon nanowire biosensor for ultrasensitive and label-free direct detection of miRNAs. Methods Mol Biol 676:111. https://doi.org/10.1021/ac801890f
Zhang Y et al (2018) Inhibiting methicillin-resistant Staphylococcus aureus by tetrahedral DNA nanostructure-enabled antisense peptide nucleic acid delivery. Nano Lett:231–233. https://doi.org/10.1021/acs.nanolett.8b02166
Zhao XH, Lin CW (2017) Rapid label-free visual detection of KRAS mutations using peptide nucleic acid and unmodified gold nanoparticles. RSC Adv 7:48554–48560. https://doi.org/10.1039/c7ra09088a
Zhao XH et al (2013) Loop-mediated isothermal amplification assay targeting the femA gene for rapid detection of Staphylococcus aureus from clinical and food samples. J Microbiol Biotechnol 23:246–250. https://doi.org/10.4014/jmb.1207.07022
Zhao X, Lin CW, Wang J, Oh DH (2014) Advances in rapid detection methods for foodborne pathogens. J Microbiol Biotechnol 24:297–312. https://doi.org/10.4014/jmb.1310.10013
Zhao XH, Chang CC, Chuang TL, Lin CW (2016) Detection of KRAS mutations of colorectal cancer with peptide-nucleic-acid-mediated real-time PCR clamping. Biotechnol Biotechnol Equip 30:1155–1162. https://doi.org/10.1080/13102818.2016.1228479
Zhao X, Zhao F, Wang J, Zhong N (2017) Biofilm formation and control strategies of foodborne pathogens: food safety perspectives. RSC Adv 7:36670–36683. https://doi.org/10.1039/c7ra02497e
Zhao XH, Xia J, Liu Y (2019a) Contrast of real-time fluorescent PCR methods for detection of Escherichia coli O157:H7 and of introducing an internal amplification control. Microorganisms 7:14. https://doi.org/10.3390/microorganisms7080230
Zhao XH, Zhao FH, Zhong NJ (2019b) Production of diacylglycerols through glycerolysis with SBA-15 supported Thermomyces lanuginosus lipase as catalyst. J Sci Food Agric. https://doi.org/10.1002/jsfa.10140
Zhichao F, Kelley SO (2009) Direct electrocatalytic mRNA detection using PNA-nanowire sensors. Anal Chem 81:612–617. https://doi.org/10.1021/ac801890f
Zhong JL, Zhao XH (2018) Isothermal amplification technologies for the detection of foodborne pathogens. Food Anal Methods 11:1543–1560. https://doi.org/10.1007/s12161-018-1177-2
Zhong J, Zhao X (2019) Transcriptomic analysis of viable but non-culturable Escherichia coli O157:H7 formation induced by low temperature. Microorganisms. https://doi.org/10.3390/microorganisms7120634
Funding
This work has been supported by the National Natural Science Foundation of China (31501582), Hubei Provincial Natural Science Foundation of China (2018CFB514), and Graduate Innovative Fund of Wuhan Institute of Technology (CX2019206).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Xihong Zhao declares that he has no conflict of interest. Chuncheng Wu declares that he has no conflict of interest.
Ethical Approval
This article does not contain any studies with human and animal subjects.
Informed Consent
Informed consent is not applicable in this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, X., Wu, C. Recent Advances in Peptide Nucleic Acids for Rapid Detection of Foodborne Pathogens. Food Anal. Methods 13, 1956–1972 (2020). https://doi.org/10.1007/s12161-020-01811-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-020-01811-6