Abstract
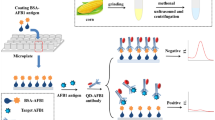
A polyvinylidene fluoride membrane-based dot immunoassay using nanobody (Nb) for rapid, qualitative, and visual detection of ochratoxin A (OTA) in cereals was developed. On the basis of optimal assay conditions, the cut-off level of this method assessed visually was 5 μg/kg for OTA, and the final results were obtained within 20 min. This method was simple with no time-consuming cleanup procedure. Good accuracy and reproducibility were obtained in recovery experiments. Results of the Nb-based dot ELISA are in agreement with the ELISA kit, except for samples that were negative for OTA presence assessed through Nb-based dot ELISA but found positive through the ELISA kit. These results indicated that the developed method could be a useful on-site screening tool for rapid detection of OTA in cereals without special instrument.

Flow chart of nanobody-based dot ELISA for OTA visual screening





Similar content being viewed by others
References
Ahn S, Lee S, Lee J, Kim B (2016) Accurate determination of ochratoxin A in Korean fermented soybean paste by isotope dilution-liquid chromatography tandem mass spectrometry. Food Chem 190:368–373
Anfossi L, Di Nardo F, Giovannoli C, Passini C, Baggiani C (2013) Increased sensitivity of lateral flow immunoassay for ochratoxin A through silver enhancement. Anal Bioanal Chem 405(30):9859–9867
Arola HO, Tullila A, Kiljunen H, Campbell K, Siitari H, Nevanen TK (2016) Specific noncompetitive immunoassay for HT-2 mycotoxin detection. Anal Chem 88(4):2446–2452
Bever CR, Majkova Z, Radhakrishnan R, Suni I, McCoy M, Wang Y, Dechant J, Gee S, Hammock BD (2014) Development and utilization of camelid VHH antibodies from alpaca for 2, 2′, 4, 4′-tetrabrominated diphenyl ether detection. Anal Chem 86(15):7875–7882
Duarte SC, Pena A, Lino CM (2010) A review on ochratoxin A occurrence and effects of processing of cereal and cereal derived food products. Food Microbiol 27(2):187–198
Hamers-Casterman C, Atarhouch T, Muyldermans S, Robinson G, Hammers C, Songa EB, Bendahman N, Hammers R (1993) Naturally occurring antibodies devoid of light chains. Nature 363(6428):446–448
He Z, He Q, Xu Y, Li Y, Liu X, Chen B, Lei D, Sun C (2013) Ochratoxin A mimotope from second-generation peptide library and its application in immunoassay. Anal Chem 85(21):10304–10311
He T, Wang Y, Li P, Zhang Q, Lei J, Zhang Z, Ding X, Zhou H, Zhang W (2014) Nanobody-based enzyme immunoassay for aflatoxin in agro-products with high tolerance to cosolvent methanol. Anal Chem 86(17):8873–8880
Hu ZQ, Li HP, Wu P, Li YB, Zhou ZQ, Zhang JB, Liu JL, Liao YC (2015) An affinity improved single-chain antibody from phage display of a library derived from monoclonal antibodies detects fumonisins by immunoassay. Anal Chim Acta 867:74–82
Lee HA, Morgan MRA (1993) Food immunoassays: applications of polyclonal, monoclonal and recombinant antibodies. Trends Food Sci Technol 4(5):129–134
Liu X, Xu Y, Xiong YH, Tu Z, Li YP, He ZY, Qiu YL, Fu JH, Gee SJ, Hammock BD (2014) VHH phage-based competitive real-time immuno-polymerase chain reaction for ultrasensitive detection of ochratoxin A in cereal. Anal Chem 86(15):7471–7477
Liu X, Xu Y, Wan DB, Xiong YH, He ZY, Wang XX, Gee SJ, Hammock BD (2015) Development of a nanobody-alkaline phosphatase fusion protein and its application in a highly sensitive direct competitive fluorescence enzyme immunoassay for detection of ochratoxin A in cereal. Anal Chem 87(2):1387–1394
Liu X, Tang Z, Duan Z, He Z, Shu M, Wang X, Gee SJ, Hammock BD, Xu Y (2017) Nanobody-based enzyme immunoassay for ochratoxin A in cereal with high resistance to matrix interference. Talanta 164:154–158
Luan C, Wang L, Chen F, Wang S, Zhao L, Shao L (2016) Determination of ochratoxin A in pig muscle using dispersive liquid-liquid microextraction combined with high-performance liquid chromatography. Food Anal Method 9(6):1490–1494
Maass DR, Sepulveda J, Pernthaner A, Shoemaker CB (2007) Alpaca (Lama pacos) as a convenient source of recombinant camelid heavy chain antibodies (VHHs). J Immunol Methods 324(1):13–25
Miller BR, Demarest SJ, Lugovskoy A, Huang F, Wu X, Snyder WB, Croner LJ, Wang N, Amatucci A, Michaelson JS, Glaser SM (2010) Stability engineering of scFvs for the development of bispecific and multivalent antibodies. Protein Eng Des Sel 23(7):549–557
Muyldermans S (2013) Nanobodies: natural single-domain antibodies. Annu Rev Biochem 82:775–797
Pal A, Dhar TK (2004) An analytical device for on-site immunoassay. Demonstration of its applicability in semiquantitative detection of aflatoxin B1 in a batch of samples with ultrahigh sensitivity. Anal Chem 76(1):98–104
Palumbo JD, O’Keeffe TL, Ho YS, Santillan CJ (2015) Occurrence of ochratoxin A contamination and detection of ochratoxigenic aspergillus species in retail samples of dried fruits and nuts. J Food Protect 78(4):836–842
Pitt JI (1987) Penicillium viridicatum, Penicillium verrucosum, and production of ochratoxin A. Appl Environ Microb 53(2):266–269
Shu M, Xu Y, Wang D, Liu X, Li Y, He Q, Tu Z, Qiu Y, Ji Y, Wang X (2015) Anti-idiotypic nanobody: a strategy for development of sensitive and green immunoassay for fumonisin B1. Talanta 143:388–393
Shu M, Xu Y, Liu X, Li Y, He Q, Tu Z, Fu JH, Gee SJ, Hammock BD (2016) Anti-idiotypic nanobody-alkaline phosphatase fusion proteins: development of a one-step competitive enzyme immunoassay for fumonisin B1 detection in cereal. Anal Chim Acta 924:53–59
Soleas GJ, Yan J, Goldberg DM (2001) Assay of ochratoxin A in wine and beer by high-pressure liquid chromatography photodiode array and gas chromatography mass selective detection. J Agri Food Chem 49(6):2733–2740
Van der Merwe KJ, Steyn PS, Fourie L, Scott DB, Theron JJ (1965) Ochratoxin A, a toxic metabolite produced by aspergillus ochraceus wilh. Nature 205(4976):1112–1113
Wang XH, Liu T, Xu N, Zhang Y, Wang S (2007) Enzyme-linked immunosorbent assay and colloidal gold immunoassay for ochratoxin A: investigation of analytical conditions and sample matrix on assay performance. Anal Bioanal Chem 389(3):903–911
Wang J, Bever CR, Majkova Z, Dechant JE, Yang J, Gee SJ, Xu T, Hammock BD (2014) Heterologous antigen selection of camelid heavy chain single domain antibodies against tetrabromobisphenol A. Anal Chem 86(16):8296–8302
Wang R, Gu X, Zhuang Z, Zhong Y, Yang H, Wang S (2016) Screening and molecular evolution of a single chain variable fragment antibody (scFv) against citreoviridin toxin. J Agri Food Chem 64(40):7640–7648
Acknowledgements
This work was financially supported by the Scientific Research Foundation of Hainan University (grant number kyqd1631) and the Academic Innovation Project for Youth Science Talents, Hainan Provincial Association for Science and Technology (grant number 201504). Partial support was provided by the Natural Science Foundation of Hainan Province (grant number 317034) and the Guangxi talent highland for preservation and deep processing research in fruits and vegetables.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of Interest
Zhichang Sun declares no conflict of interest. Zhenhua Duan declares no conflict of interest. Xing Liu declares no conflict of interest. Xing Deng declares no conflict of interest. Zongwen Tang declares no conflict of interest.
Informed Consent
Not applicable
Rights and permissions
About this article
Cite this article
Sun, Z., Duan, Z., Liu, X. et al. Development of a Nanobody-Based Competitive Dot ELISA for Visual Screening of Ochratoxin A in Cereals. Food Anal. Methods 10, 3558–3564 (2017). https://doi.org/10.1007/s12161-017-0915-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-017-0915-1